LAMP Assay for Rapid Viral Diagnostics: A 2024 Review of Principles, Applications, and Future Directions
This review provides a comprehensive analysis of Loop-Mediated Isothermal Amplification (LAMP) technology for rapid viral diagnostics, tailored for researchers, scientists, and drug development professionals.
LAMP Assay for Rapid Viral Diagnostics: A 2024 Review of Principles, Applications, and Future Directions
Abstract
This review provides a comprehensive analysis of Loop-Mediated Isothermal Amplification (LAMP) technology for rapid viral diagnostics, tailored for researchers, scientists, and drug development professionals. We explore the foundational molecular principles and advantages of LAMP over conventional PCR, including its isothermal nature and rapid result generation. The article details current methodological protocols, diverse viral pathogen applications (e.g., SARS-CoV-2, Influenza, Dengue, HIV, and emerging threats), and integration with point-of-care platforms. Critical troubleshooting and optimization strategies for primer design, reaction conditions, and inhibitor management are examined. Finally, we present a rigorous validation framework and comparative assessment against gold-standard methods (qRT-PCR) and other isothermal techniques (RPA, NASBA), discussing regulatory pathways and clinical performance metrics. This synthesis aims to guide the implementation and advancement of LAMP in both research and clinical settings.
Understanding LAMP Technology: Core Principles and Advantages for Viral Detection
This whitepaper, framed within a broader thesis reviewing LAMP assays for rapid viral diagnostics, provides an in-depth technical guide to the core molecular mechanisms enabling Loop-Mediated Isothermal Amplification (LAMP). We dissect the enzymatic activities, primer design logic, and complex reaction dynamics that facilitate rapid, isothermal nucleic acid amplification, making it indispensable for point-of-care diagnostics and field-deployable pathogen detection.
Unlike the thermal cycling required in Polymerase Chain Reaction (PCR), LAMP operates at a constant temperature (typically 60–65°C). This isothermal characteristic eliminates the need for precision thermocyclers, enabling instrument simplification and rapid amplification, often within 15–60 minutes. The core innovation lies in a set of specifically designed primers and the strand-displacing activity of Bst DNA polymerase.
Core Molecular Mechanism: A Stepwise Analysis
Key Enzymatic Component:BstDNA Polymerase
The reaction is powered by a DNA polymerase with high strand displacement activity, commonly derived from Geobacillus stearothermophilus (Bst). Key properties include:
- Strand Displacement: Actively unwinds double-stranded DNA without the need for heat denaturation.
- Lack of 5'→3' Exonuclease Activity: Prevents undesired degradation of primers and amplicons.
- Optimal Temperature: ~65°C, ideal for rapid DNA synthesis while maintaining primer-template complex stability.
The Primer Design: FIP, BIP, F3, B3, LF, LB
LAMP utilizes 4 to 6 primers recognizing 6 to 8 distinct regions on the target DNA. This design is fundamental to forming loop structures for self-primed amplification.
- F3 (Forward Outer) & B3 (Backward Outer): Initiate the first strand displacement events.
- FIP (Forward Inner Primer): Contains the F2 sequence (complementary to F2c) and the same sequence as F1c.
- BIP (Backward Inner Primer): Contains the B2 sequence (complementary to B2c) and the same sequence as B1c.
- LF & LB (Loop Primers): Bind to loop regions formed during amplification, accelerating the reaction by providing additional priming sites.
The Three-Stage Amplification Process
Stage 1: Strand Displacement Synthesis and Loop Formation Initiation begins with FIP binding to the F2c region. Bst polymerase extends the primer, synthesizing a complementary strand. Simultaneously, the F3 primer binds upstream to F2c, and its extension displaces the FIP-linked strand, releasing a single-stranded DNA. The complementary F1 and F1c regions on this strand self-anneal, forming a loop at one end (dumbbell-like structure). An identical process initiated by BIP and B3 on the opposite end generates a stem-loop DNA structure with complementary ends. These ends anneal to form a dumbbell structure, the starting material for cyclic amplification.
Stage 2: Cyclic Amplification Phase The dumbbell structure self-primes from the 3' end of the loop. Strand displacement synthesis elongates the strand, regenerating the original stem-loop and simultaneously displacing a new strand. This displaced strand forms a complementary structure at the opposite end, leading to a series of self-priming and strand displacement events that exponentially amplify the target, producing concatemers of alternating sense and antisense repeats.
Stage 3: Elongation and Recycling Phase The loop primers (LF, LB) bind to the single-stranded loops in the dumbbell structures, further accelerating amplification by providing additional initiation sites. The final products are a mixture of stem-loop DNAs of various lengths and long cauliflower-like structures with multiple loops, resulting from annealing between alternately inverted repeats of the target in the same strand.
Diagram Title: The Three Core Stages of LAMP Amplification
Quantitative Performance Data
Table 1: Comparative Performance of LAMP vs. qPCR for Viral Detection
| Parameter | LAMP Assay | Traditional qPCR | Notes |
|---|---|---|---|
| Amplification Temperature | Constant (60-65°C) | Cycled (95°C, 55-60°C, 72°C) | LAMP requires only a heat block. |
| Time to Result | 15 - 60 minutes | 60 - 120 minutes | LAMP speed varies with target copy number. |
| Detection Limit (LoD) | 1 - 10 copies/µL | 1 - 10 copies/µL | Comparable analytical sensitivity. |
| Amplification Efficiency | High (Exponential) | High (Exponential) | Both are highly efficient. |
| Instrument Complexity | Low (Isothermal block) | High (Precision thermocycler) | Key for POC/decentralized use. |
| Multiplexing Capability | Moderate | High | LAMP multiplexing is more challenging. |
Table 2: Key Enzymes and Reagents in a Standard LAMP Reaction Mix
| Component | Typical Concentration | Function |
|---|---|---|
| Bst DNA Polymerase (Large Fragment) | 0.08 - 0.32 U/µL | Strand-displacing DNA synthesis. |
| dNTPs | 1.0 - 1.4 mM each | Nucleotide building blocks. |
| MgSO4 | 4 - 8 mM | Essential cofactor for polymerase activity. |
| Betaine | 0.6 - 1.0 M | Reduces secondary structure in GC-rich targets; equalizes base stability. |
| FIP/BIP Primers | 1.0 - 1.6 µM each | Inner primers for loop formation. |
| F3/B3 Primers | 0.1 - 0.2 µM each | Outer primers for strand displacement initiation. |
| LF/LB Primers | 0.4 - 0.8 µM each | Loop primers to accelerate reaction. |
| Thermostable Buffer | 1X | Maintains optimal pH (8.0-8.8) at 60-65°C. |
| Target DNA | Variable | Template for amplification. |
Detailed Experimental Protocol: Validating a LAMP Assay
Objective: To establish and optimize a LAMP assay for the detection of a specific viral gene sequence.
Primer Design Protocol
- Sequence Alignment: Retrieve 20-30 target viral genome sequences from NCBI GenBank. Align using Clustal Omega to identify conserved regions (~200 bp).
- Primer Generation: Input the conserved sequence into primer design software (e.g., PrimerExplorer V5, Eiken). Specify parameters:
- Primer Length: F3/B3: 17-20 bp; F1/F2/B1/B2: 18-21 bp.
- Tm: F3/B3: ~60°C; F2/B2: ~65°C; F1/B1 (for loop): >65°C.
- GC Content: 40-65%.
- Distance: F2 to F1: 40-60 bp; F2 to F3: 0-20 bp.
- Specificity Check: Perform BLAST analysis of all primers against the host genome and relevant microbiome to ensure no significant homology.
LAMP Reaction Setup (25 µL total volume)
- Prepare a Master Mix on ice:
- 12.5 µL 2X Isothermal Amplification Buffer (commercial, contains dNTPs, Mg2+, buffer).
- 1.0 µL Bst 2.0 WarmStart DNA Polymerase (8 U/µL).
- 5.0 µL Primer Mix (containing FIP/BIP at 16 µM each, F3/B3 at 2 µM each, LF/LB at 8 µM each in nuclease-free water).
- 1.0 µL Betaine (5 M stock).
- 1.5 µL Nuclease-free water.
- Aliquot 21 µL of Master Mix into each reaction tube.
- Add 4 µL of template DNA (or nuclease-free water for no-template control).
- Incubate in a heat block or dry bath at 65°C for 30-45 minutes.
- Termination: Heat inactivation at 80°C for 5 minutes (optional).
Amplicon Detection and Analysis
- Real-Time Monitoring: Use a real-time fluorimeter with intercalating dye (e.g., SYTO 9, EvaGreen) added to the master mix. Plot fluorescence vs. time (cycle threshold).
- Endpoint Detection:
- Visual: Add 1 µL of SYBR Green I post-amplification. Positive: bright green; negative: orange.
- Gel Electrophoresis: Run 5 µL of product on a 2% agarose gel. Positive: ladder-like pattern of multiple bands.
- Turbidity: Measure precipitation of magnesium pyrophosphate at 400 nm.
Diagram Title: Standard LAMP Assay Development and Analysis Workflow
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for LAMP Development
| Item | Example Product/Supplier | Function in Research Context |
|---|---|---|
| Strand-Displacing Polymerase | Bst 2.0/3.0 WarmStart (NEB), Bst LF (Thermo) | Engineered for higher speed, stability, and hot-start capability to reduce non-specific amplification. |
| Isothermal Amplification Buffer | Commercial 2X Mix (OptiGene, NEB) | Pre-optimized buffer with Mg2+, dNTPs, and stabilizers for robust performance. |
| Fluorescent Intercalating Dye | SYTO 9, EvaGreen (Biotium) | Real-time monitoring of amplification; must be compatible with isothermal conditions. |
| Visual Detection Dye | SYBR Green I, Hydroxy Naphthol Blue (HNB) | For colorimetric endpoint readout, crucial for field applications without instruments. |
| Synthetic Gene Targets (GBlocks) | Integrated DNA Technologies (IDT) | Positive control templates for assay optimization and standard curve generation. |
| RNase/DNase Inhibitors | Murine RNase Inhibitor (NEB) | Essential for developing RT-LAMP assays for RNA viruses to prevent template degradation. |
| Rapid Extraction Kits | Magnetic bead-based kits (Qiagen, Thermo) | For fast sample preparation integrated into the LAMP workflow, critical for total assay time. |
| Lyophilization Reagents | Trehalose, PEG | For formulating stable, room-temperature LAMP master mixes for distribution. |
Within the context of a rapid viral diagnostics review, Loop-Mediated Isothermal Amplification (LAMP) stands out for its speed, sensitivity, and robustness. This technical guide details the core components underpinning its efficacy in point-of-care and laboratory settings.
Primer Design for LAMP Assays
LAMP employs a set of four to six primers that recognize six to eight distinct regions on the target DNA, ensuring exceptional specificity. This complex design is critical for the self-priming, loop-forming mechanism.
- F3 and B3: Outer primers that initiate strand displacement synthesis.
- FIP and BIP: Inner primers containing complementary sequences (F1c/F2 and B1c/B2) that are central to loop formation and exponential amplification.
- LF and LB (Optional): Loop-forward and loop-backward primers accelerate amplification by binding to the loop structures, further enhancing speed.
Key Design Parameters:
- Melting Temperature (Tm): Typically 55-65°C, with inner primers ~5°C higher than outer primers.
- GC Content: Ideally between 40-65%.
- Amplicon Length: Optimal range is 120-300 bp.
- Distance between primer binding sites: Critical for efficient strand displacement (see table).
Table 1: Primer Binding Region Specifications for LAMP Assay Design
| Primer Region | Length (nt) | Typical Distance from 5' End of Target |
|---|---|---|
| F2 | 18-22 | Defines 5' end of final amplicon |
| F1 (within FIP) | 18-21 | Immediately downstream of F2 |
| B2 (within BIP) | 18-22 | Upstream of F1c |
| B1 (within BIP) | 18-21 | Immediately upstream of B2 |
| F3 | 17-21 | Upstream of F2 |
| B3 | 17-21 | Downstream of B2c |
| LF (if used) | 17-21 | Between F1 and F2 regions |
| LB (if used) | 17-21 | Between B1 and B2 regions |
Protocol 1: In Silico Primer Design and Validation for a Viral Target
- Retrieve consensus genome sequence for the target virus (e.g., SARS-CoV-2 nucleocapsid gene) from NCBI GenBank.
- Use primer design software (e.g., PrimerExplorer V5, NEB LAMP Designer) to input the FASTA sequence.
- Set parameters: Tm = 60±2°C, GC content = 40-65%, amplicon size = 120-250 bp.
- Select the top-ranked primer set based on software score.
- Perform specificity check via BLAST against the human genome and microbial database.
- Validate primer dimer and secondary structure formation using tools like OligoAnalyzer.
DNA Polymerase for Isothermal Amplification
The Bst DNA polymerase large fragment is the enzyme of choice for standard LAMP. Its key characteristics are:
- High Strand Displacement Activity: Eliminates the need for thermal denaturation.
- Optimal Temperature: 60-65°C.
- Lack of 5'→3' Exonuclease Activity: Prevents degradation of primers and loop structures.
For RNA virus detection (RT-LAMP), the reaction requires reverse transcriptase. This can be supplied as a separate enzyme (e.g., AMV Reverse Transcriptase) or, more commonly, as a blend with Bst polymerase. Newer engineered variants offer enhanced speed, processivity, and tolerance to inhibitors found in clinical samples.
Table 2: Comparison of Common LAMP Polymerases
| Polymerase | Source | Key Features | Optimal Temp. | Recommended for |
|---|---|---|---|---|
| Bst 2.0/3.0 | Bacillus stearothermophilus | High displacement, robust, fast | 60-65°C | Standard DNA target LAMP |
| Bst 2.0 WarmStart | Engineered variant | Enzyme inactive <45°C, prevents non-specific initiation | ~65°C | High sensitivity assays |
| GspSSD | Geobacillus sp. | Extremely fast, high processivity | 65-68°C | Ultra-rapid diagnostics (<15 min) |
| RT-Bst Blend | Commercial mixture | Combined reverse transcriptase & Bst activity | 60-65°C | RT-LAMP for RNA viruses |
Protocol 2: Optimizing LAMP Master Mix for Inhibitor-Rich Samples
- Prepare a standard 25 µL LAMP reaction: 1.4 mM dNTPs, 6 mM MgSO₄, 1x Isothermal Amplification Buffer, 1.6 µM each FIP/BIP, 0.2 µM each F3/B3, 0.8 µM each LF/LB, 8 U Bst 3.0 polymerase, target template.
- Spike reactions with serial dilutions of a known inhibitor (e.g., 10%-0.1% blood, 200-0 mM EDTA).
- Include additives in test reactions: 1 M Betaine, 0.2 mg/mL BSA, or 2% Tween-20.
- Incubate at 65°C for 30-45 minutes.
- Compare time-to-positive (TTP) and endpoint fluorescence between control and inhibitor-spiked reactions to determine optimal additive.
Detection Methods for LAMP Amplicons
Detection can be real-time (quantitative) or endpoint (qualitative), aligning with different diagnostic use cases.
A. Real-Time Detection:
- Intercalating Dyes (SYTO-9, EvaGreen): Fluoresce when bound to dsDNA. Provide real-time amplification curves. Risk of post-amplification contamination if tubes are opened.
- Pyrophosphate Detection: Turbidity or magnesium pyrophosphate precipitation can be measured optically.
B. Endpoint Detection:
- Colorimetric: pH-sensitive dyes (phenol red) change from pink to yellow due to proton release during amplification. Metal-ion indicators (hydroxynaphthol blue) change from violet to sky blue upon chelation of Mg²⁺ by pyrophosphate.
- Lateral Flow Dipsticks (LFD): Use biotin- and FITC-labeled primers. Amplicons are captured on anti-FITC test lines, providing a visual band. Highly user-friendly for field use.
Table 3: Comparison of LAMP Detection Methodologies
| Method | Principle | Readout | Sensitivity | Advantage | Disadvantage |
|---|---|---|---|---|---|
| Fluorescence (Intercalator) | Dye binds dsDNA | Real-time curve/Endpoint | High (~10 copies) | Quantitative, closed-tube | Requires fluorometer |
| Colorimetric (pH) | Proton release lowers pH | Color change (pink→yellow) | Medium-High | Visual, simple | Buffer dependent, subjective |
| Colorimetric (HNB) | Mg²⁺ chelation by PPi | Color change (violet→blue) | Medium-High | Visual, pre-reaction add | Subjective |
| Lateral Flow Dipstick | Immunocapture of tagged primers | Visual band on strip | High | Specific, portable, semi-quantitative | Requires primer tagging, open-tube step |
| Turbidity | Precipitation of Mg₂P₂O₇ | Turbidity increase (OD 400nm) | Medium | Simple equipment | Less sensitive |
Protocol 3: Endpoint Detection using Colorimetric (pH) and Lateral Flow Methods Part A: Colorimetric Detection
- Prepare LAMP master mix replacing ~0.5x of the standard buffer volume with a pH-sensitive dye (e.g., 120 µM Phenol Red).
- Run amplification at 65°C for 30 min.
- Visually inspect tubes: Positive = yellow, Negative = pink/magenta. Use a reference color chart for interpretation.
Part B: Lateral Flow Detection
- Design FIP primer with 5' FITC label and BIP primer with 5' Biotin label.
- Perform LAMP reaction with labeled primers.
- Dilute 5 µL of amplicon in 95 µL of assay buffer.
- Dip the lateral flow strip into the solution for 3-5 minutes.
- Read result: Positive = Control line (C) + Test line (T). Negative = Control line (C) only.
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for Developing a LAMP Assay
| Item | Function | Example Product/Supplier |
|---|---|---|
| Bst 2.0/3.0 WarmStart DNA Polymerase | High-activity, strand-displacing polymerase for core amplification | New England Biolabs (NEB) |
| Isothermal Amplification Buffer (with MgSO₄) | Provides optimal ionic and pH conditions for Bst polymerase | NEB, Thermo Fisher |
| dNTP Solution Mix | Nucleotide building blocks for DNA synthesis | Sigma-Aldrich, Promega |
| Target-Specific LAMP Primer Set (F3/B3, FIP/BIP, LF/LB) | Drives specific, exponential amplification of target sequence | Integrated DNA Technologies (IDT), Eurofins |
| Synthetic gBlock Gene Fragment or Viral RNA | Positive control template for assay development and validation | IDT, ATCC |
| Fluorescent DNA Intercalating Dye (e.g., SYTO-9) | For real-time quantification on a plate reader or qPCR instrument | Thermo Fisher, Biotium |
| Colorimetric Detection Dye (e.g., Phenol Red) | For visual, endpoint readout of amplification | Sigma-Aldrich |
| Lateral Flow Dipsticks (Biotin/FITC) | For rapid, instrument-free visual confirmation of amplicons | Milenia HybriDetect, Ustar Biotechnologies |
| RNase/DNase-Free Water & Tubes | Prevents nucleic acid degradation and contamination | Thermo Fisher, Corning |
LAMP Amplification Mechanism Workflow
LAMP Detection Method Decision Tree
This document, framed within a broader thesis reviewing Loop-Mediated Isothermal Amplification (LAMP) for rapid viral diagnostics, provides a technical comparison between LAMP and traditional Polymerase Chain Reaction (PCR). The focus is on the inherent advantages that position LAMP as a transformative technology for point-of-care and resource-limited settings.
Core Comparative Analysis
Table 1: Quantitative Comparison of LAMP vs. Traditional PCR
| Parameter | LAMP | Traditional PCR (qPCR) |
|---|---|---|
| Amplification Temperature | Isothermal (60–65°C) | Thermo-cycled (95°C, 55–65°C, 72°C) |
| Reaction Time | 15–60 minutes | 60–180 minutes (including cycling) |
| Typical Detection Limit (Copies/µL) | 10–100 | 10–100 |
| Instrumentation Complexity | Low (water bath/heat block) | High (thermocycler with optical module) |
| Enzymes per Reaction | Bst DNA Polymerase (1) | Thermostable DNA Polymerase (e.g., Taq) (1) |
| Primer Set Complexity | High (4–6 primers) | Low (2 primers) |
| Tolerance to Inhibitors | High | Moderate to Low |
| Amplicon Detection Methods | Turbidity, Fluorescence, Colorimetric (pH), Gel Electrophoresis | Fluorescence (probes/intercalating dyes), Gel Electrophoresis |
| Assay Design Complexity | High (primer design) | Moderate |
| Open-tube Contamination Risk | Higher (post-amplification) | Lower (closed-tube for qPCR) |
Experimental Protocols
Protocol 1: Standard Colorimetric LAMP Assay for Viral RNA Detection
- Nucleic Acid Extraction: Use a magnetic bead-based or spin-column kit. Elute in 20-50 µL of nuclease-free water or the provided elution buffer.
- Master Mix Preparation (25 µL reaction):
- 12.5 µL of 2x commercial LAMP master mix (contains Bst polymerase, dNTPs, buffer, MgSO4, and phenol red).
- 1.0 µL of 10x primer mix (containing F3, B3, FIP, BIP at optimized concentrations, typically 1.6 µM FIP/BIP, 0.2 µM F3/B3).
- 1.0 µL of reverse transcriptase (if detecting RNA).
- 5.5 µL of nuclease-free water.
- 5.0 µL of template nucleic acid.
- Amplification: Incubate the reaction tube at 63°C for 30 minutes in a heat block or water bath.
- Detection: Visualize result. A color change from pink (alkaline) to yellow (acidic) indicates a positive amplification. Include a no-template control (NTC) and positive control.
Protocol 2: Real-time Fluorescent LAMP with Lateral Flow Detection
- Amplification: Perform Protocol 1, but use a fluorescent LAMP master mix containing a DNA intercalating dye (e.g., SYTO 9) or labeled primers. Incubate in a portable real-time fluorometer at 65°C for 40 minutes with fluorescence read every 30 seconds.
- Lateral Flow Verification (Post-amplification):
- Dilute 2 µL of LAMP product in 98 µL of running buffer.
- Apply 100 µL to the sample pad of a lateral flow strip designed for the specific hapten/digoxigenin labeled amplicon.
- Wait 5–10 minutes. The appearance of both test (T) and control (C) lines indicates a positive result.
Visualizations
Diagram 1: LAMP Primer Binding and Amplification Mechanism
Diagram 2: LAMP vs. PCR Diagnostic Workflow Comparison
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for LAMP-Based Viral Diagnostic Assay Development
| Item | Function & Rationale |
|---|---|
| Bst 2.0/3.0 DNA Polymerase | Strand-displacing DNA polymerase with high processivity, enabling isothermal amplification. The 3.0 version often has enhanced speed and tolerance to inhibitors. |
| WarmStart LAMP/RT-LAMP Kit | Commercial master mix providing optimized buffer, dNTPs, Mg2+, polymerase, and often a reverse transcriptase. Includes colorimetric indicators (e.g., phenol red) for visual readout. |
| LAMP Primer Design Software | Tools like PrimerExplorer (Eiken) or NEB LAMP Primer Design Tool are critical for designing the complex set of 4-6 target-specific primers (F3, B3, FIP, BIP, LF, LB). |
| Synthetic Viral DNA/RNA Controls | Cloned plasmid or gBlock fragments containing the target sequence (e.g., SARS-CoV-2 N gene). Essential for assay validation, optimization, and as a positive control. |
| Heat Block or Dry Bath | Simple, low-cost instrument maintaining a constant temperature between 60–65°C for isothermal incubation. Replaces expensive thermocyclers. |
| Fluorescent Intercalating Dye (SYTO 9) | For real-time monitoring of LAMP amplification. Binds dsDNA, allowing quantification and threshold time (Tt) measurement in portable fluorometers. |
| Lateral Flow Strips (Biotin/Digoxigenin) | For endpoint detection. Used with labeled primers (FITC, Biotin) to provide a simple, equipment-free "dipstick" result, enhancing point-of-care utility. |
| RNase Inhibitor | Critical for RT-LAMP assays to protect viral RNA templates from degradation during reaction setup, ensuring assay sensitivity. |
| Rapid Viral Lysis Buffer | For simplified sample preparation. May contain detergents and chelating agents to release viral nucleic acids while inactivating nucleases and reducing inhibitors. |
| Internal Control Template | A non-target nucleic acid sequence amplified by a separate primer set within the same reaction. Monitors for amplification failure due to inhibitors, ensuring result reliability. |
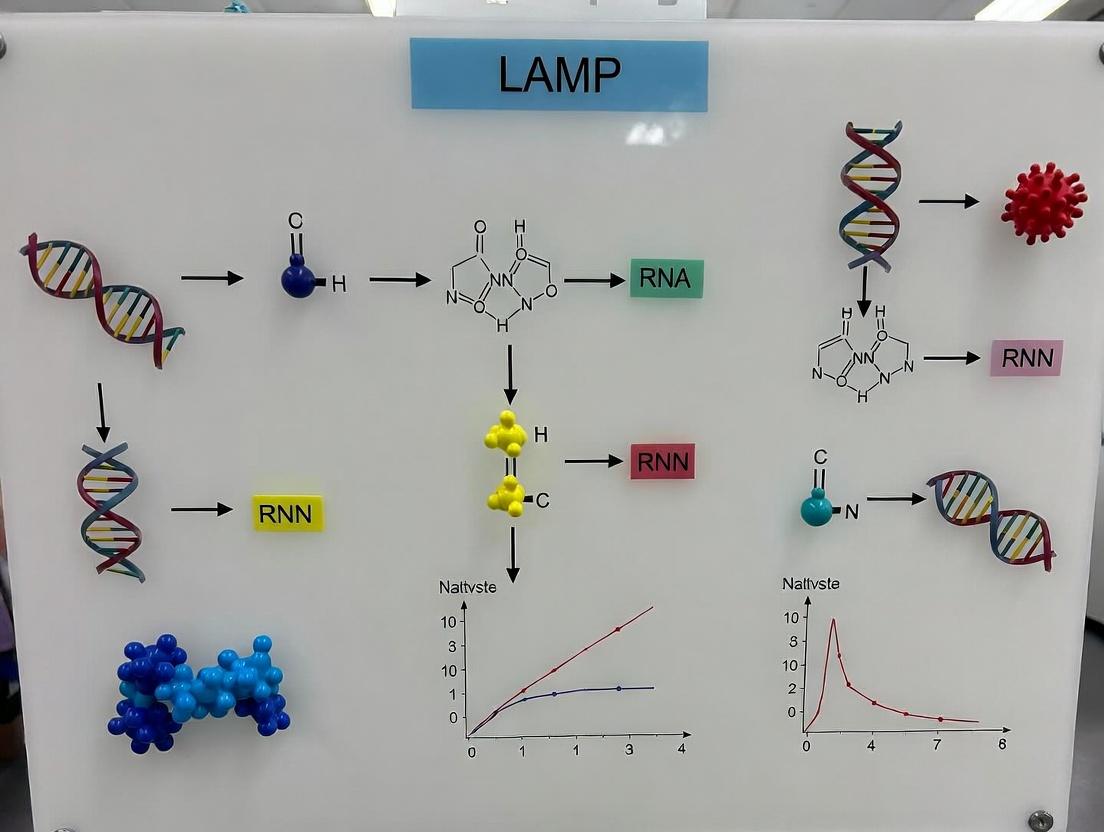
This whitepaper details the technical evolution of Loop-Mediated Isothermal Amplification (LAMP) from its conceptual inception to its current status as a cornerstone in rapid viral diagnostics. Framed within a broader thesis reviewing LAMP assays, this document provides an in-depth analysis of its development, core mechanisms, experimental protocols, and key reagents, tailored for research and drug development professionals.
The LAMP technique was invented by Notomi et al. in 2000 at Eiken Chemical Co., Ltd., Japan, as a novel nucleic acid amplification method. The core concept was to achieve high-specificity, high-efficiency DNA amplification under isothermal conditions (60-65°C), eliminating the need for thermal cycling equipment like traditional PCR. The initial concept focused on using a DNA polymerase with strand displacement activity and a set of four to six specially designed primers that recognize six to eight distinct regions on the target DNA, facilitating auto-cycling strand displacement synthesis.
Technical Evolution and Milestones
The development of LAMP can be segmented into distinct phases, marked by key innovations that transformed it from a proof-of-concept to a robust diagnostic tool.
Table 1: Evolution of LAMP Technology and Performance Metrics
| Year | Milestone | Key Improvement | Impact on Diagnostic Use | Reported Sensitivity (copies/μL) | Reported Time to Result |
|---|---|---|---|---|---|
| 2000 | Initial Publication (Notomi et al.) | Basic LAMP mechanism with Bst DNA polymerase. | Proof of isothermal amplification concept. | 10 – 100 | 60 – 90 min |
| 2002-2005 | RT-LAMP Development | Integration of reverse transcriptase for RNA targets. | Enabled direct detection of RNA viruses. | 10 – 1000 | 60 – 120 min |
| 2008-2012 | Primer Design Optimization & Multiplexing | Advanced algorithms (e.g., PrimerExplorer), multiplex LAMP. | Improved specificity, detection of multiple targets. | 1 – 10 | 30 – 60 min |
| 2013-2018 | Integration with Detection Methods | Colorimetric (pH indicators, metal-ion dyes), turbidity, fluorescence real-time. | Simplified endpoint detection, enabled real-time quantification. | 1 – 10 | 15 – 45 min |
| 2019-Present | Point-of-Care (POC) & Instrument-Free Formats | Lyophilized reagents, paper-based devices, smartphone detection. | Facilitated field-deployable, low-resource diagnostics. | 10 – 100 | 20 – 40 min |
Core Mechanism and Pathway
LAMP amplifies DNA with high specificity and efficiency using 4-6 primers and strand-displacing DNA polymerase.
Title: LAMP Amplification Core Pathway
Detailed Experimental Protocol for a Standard RT-LAMP Assay
This protocol is cited as a representative methodology for detecting an RNA virus (e.g., SARS-CoV-2 N gene).
Primer Design
- Use software (e.g., PrimerExplorer V5) to design primers targeting 6-8 distinct regions of the viral genome.
- A standard set includes: Forward Inner Primer (FIP), Backward Inner Primer (BIP), Forward Outer Primer (F3), Backward Outer Primer (B3), and optionally Loop Forward (LF) and Loop Backward (LB) primers.
- Resuspend primers in nuclease-free water to 100 μM stock solutions.
Reaction Setup
- Reaction Volume: 25 μL.
- Isothermal Buffer: 1.4 mM dNTPs, 6 mM MgSO₄, 20 mM Tris-HCl (pH 8.8), 10 mM KCl, 10 mM (NH₄)₂SO₄, 0.1% Tween 20.
- Primers (Final Concentration): 1.6 μM FIP/BIP, 0.2 μM F3/B3, 0.8 μM LF/LB.
- Enzyme Mix: 8 U Bst 2.0 or 3.0 DNA polymerase (strand-displacing), 0.25 U AMV or WarmStart RTX reverse transcriptase.
- Detection Additive: 1X fluorescent dye (e.g., SYTO 9, EvaGreen) or 120 μM Hydroxy Naphthol Blue (HNB).
- Template: 2-5 μL of extracted RNA or heat-inactivated viral sample.
- Complete with nuclease-free water.
Amplification & Detection
- Assemble reaction components on ice, adding enzyme last.
- Incubate in a heat block, water bath, or real-time isothermal fluorometer at 63°C for 30-45 minutes.
- Endpoint Detection:
- Colorimetric (HNB): Visual inspection. Positive = sky blue, Negative = violet.
- Turbidity: Measure optical density at 400 nm (OD₄₀₀) for magnesium pyrophosphate precipitate.
- Real-time Detection: Monitor fluorescence signal every 60 seconds.
Post-Amplification Analysis (Optional)
- Run 5 μL of product on a 2% agarose gel. LAMP yields a characteristic ladder-like pattern of multiple bands.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Materials for LAMP Assay Development
| Reagent/Material | Function/Description | Example Product/Vendor |
|---|---|---|
| Strand-Displacing DNA Polymerase | Core enzyme for isothermal amplification. High displacement activity and thermal stability at 60-65°C. | Bst 2.0/3.0 WarmStart (NEB), Isothermal Mastermix (OptiGene). |
| Reverse Transcriptase (for RT-LAMP) | Converts RNA template to cDNA for amplification. Must be active at isothermal temperature. | WarmStart RTX (NEB), GspSSD 2.0 (OptiGene). |
| LAMP-Specific Primer Sets | 4-6 primers designed to recognize 6-8 target regions. Critical for specificity and efficiency. | Custom-designed (IDT, Eurofins) using PrimerExplorer software. |
| Isothermal Amplification Buffer | Provides optimal pH, salt, and nucleotide conditions for Bst polymerase activity. Often includes Mg²⁺. | Proprietary buffers included with enzyme mastermixes. |
| Visual Detection Dyes | Enable instrument-free result interpretation. pH indicators (phenol red, HNB) or metal-ion indicators (calcein). | Hydroxy Naphthol Blue (HNB), Phenol Red, Calcein-Mn²⁺. |
| Fluorescent Intercalating Dyes | For real-time quantification and kinetic analysis of the amplification reaction. | SYTO 9, EvaGreen, SYBR Green (added post-reaction). |
| Positive Control Template | Synthetic DNA/RNA spanning the target primer regions. Essential for assay validation and troubleshooting. | GBlocks (IDT), Twist Synthetic DNA. |
| Nuclease-Free Water & Tubes | Prevents degradation of primers, templates, and enzymes. Low-bind tubes minimize adsorption. | Molecular biology grade (Thermo Fisher, Ambion). |
| Portable Isothermal Incubator | Provides constant, precise temperature for field or point-of-care use. | Mini dry bath, pocket-sized incubators (e.g., from BioRanger). |
The strategic identification of critical viral targets is foundational to developing diagnostics and therapeutics. Within the context of a comprehensive review of Loop-Mediated Isothermal Amplification (LAMP) for rapid viral diagnostics, understanding the fundamental distinctions between DNA and RNA viruses, and the unique replication strategy of retroviruses, is paramount. This guide provides a technical analysis of viral genome replication and its exploitation for targeted intervention, with a focus on supporting assay design for researchers and drug development professionals.
Viral Genome Classification and Replication Strategies
Viruses are categorized based on their genomic material (DNA or RNA), strandedness (single or double), and replication mechanics. This classification, such as the Baltimore system, directly informs the selection of diagnostic targets and antiviral strategies.
Table 1: Comparative Analysis of DNA vs. RNA Viruses
| Feature | DNA Viruses | RNA Viruses | Reverse Transcribing Viruses (e.g., HIV, HBV) |
|---|---|---|---|
| Genome Type | DNA (ds or ss) | RNA (ds, +ss, -ss) | RNA (retroviruses) or DNA (hepadnaviruses) |
| Replication Site | Primarily nucleus (except Poxviridae) | Cytoplasm (except Orthomyxoviridae, Retroviruses) | Cytoplasm & Nucleus |
| Polymerase | Viral or host DNA-dependent DNA polymerase | Virus-encoded RNA-dependent RNA polymerase (RdRp) | Viral Reverse Transcriptase (RT) & Integrase |
| Error Rate | Low (~10⁻⁸ to 10⁻¹¹ errors/base) | High (~10⁻³ to 10⁻⁵ errors/base) | High (RT lacks proofreading) |
| Mutation Rate | Low, stable genomes | High, rapid evolution | High, rapid evolution |
| Example Families | Herpesviridae, Adenoviridae, Poxviridae | Coronaviridae, Flaviviridae, Orthomyxoviridae | Retroviridae (HIV), Hepadnaviridae (HBV) |
| Key Enzymatic Targets | Viral DNA polymerase, Helicase, Primase | RdRp, Protease, Helicase | Reverse Transcriptase, Integrase, Protease |
Critical Viral Targets and Experimental Protocols
Targeting DNA Virus Replication: Herpes Simplex Virus (HSV) Thymidine Kinase and DNA Polymerase Assay
DNA viruses often encode their own replication machinery, providing virus-specific targets.
Protocol: In Vitro DNA Polymerase Inhibition Assay
- Reagent Preparation: Prepare a reaction mix containing: 50 mM Tris-HCl (pH 8.0), 5 mM MgCl₂, 1 mM DTT, 100 µg/mL BSA, 10 µM each dNTP (including [³H]-dTTP for radiolabeling or fluorescent-dUTP for fluorescence detection), 100 nM primer/template DNA (e.g., poly(dA)/oligo(dT)₁₅), and purified viral DNA polymerase (e.g., HSV UL30 polymerase).
- Inhibitor Incubation: Pre-incubate the polymerase with a serial dilution of the candidate inhibitor compound (e.g., acyclovir triphosphate, foscarnet) for 10 minutes at 4°C.
- Reaction Initiation: Start the reaction by adding the primer/template substrate. Incubate at 37°C for 30 minutes.
- Reaction Termination: Stop the reaction by adding 10 µL of 0.5 M EDTA.
- Detection & Analysis:
- Radiolabel Method: Spot reaction products onto DE81 filter papers, wash extensively with 0.5M Na₂HPO₄ to remove unincorporated nucleotides, and measure incorporated radioactivity by scintillation counting.
- Fluorescence Method: Transfer reaction to a black microplate and measure fluorescence (ex/cm appropriate for the labeled dUTP). Calculate IC₅₀ values from dose-response curves.
Targeting RNA Virus Replication: RNA-Dependent RNA Polymerase (RdRp) Activity Assay
The RdRp is absolutely essential for RNA virus replication and has no direct host counterpart, making it a premier target.
Protocol: RdRp Primer Extension Assay for SARS-CoV-2
- Template Design: Synthesize a short, single-stranded RNA template (e.g., 50-nt) with a known sequence. Design a complementary 5'-fluorescently labeled (e.g., FAM) DNA or RNA primer (e.g., 20-nt).
- Assembly: Anneal the primer to the template at a 1:1.2 molar ratio in annealing buffer by heating to 95°C for 2 min and slowly cooling.
- Reaction Setup: In a final volume of 50 µL, combine: 50 mM HEPES (pH 7.5), 5 mM MgCl₂, 1 mM DTT, 0.01% Triton X-100, 500 µM each NTP, 20 nM primer/template, and purified SARS-CoV-2 nsp12 (RdRp) complexed with nsp7/nsp8 cofactors.
- Inhibitor Testing: Include a DMSO control and serial dilutions of a candidate RdRp inhibitor (e.g., Remdesivir triphosphate).
- Incubation & Termination: Incubate at 30°C for 60 min. Terminate with 50 µL of Gel Loading Buffer II (95% formamide, 18 mM EDTA, 0.025% SDS, xylene cyanol, bromophenol blue).
- Analysis: Denature samples at 95°C for 5 min, resolve products on a denaturing polyacrylamide gel (15-20%), and visualize using a fluorescence gel imaging system. Full-length extension product yield is quantified to determine inhibitor potency.
The Pivotal Role of Reverse Transcriptase (RT)
RT converts viral RNA into DNA, a central step in the life cycle of retroviruses (HIV) and hepadnaviruses (HBV). Its error-prone nature drives viral diversity and immune escape but also provides a critical, well-validated drug target.
Protocol: Reverse Transcriptase Activity Assay (Colorimetric)
- Principle: This assay uses a poly(rA) template and oligo(dT) primer. RT incorporates biotin-labeled dUTP, which is captured and detected colorimetrically.
- Procedure: a. Binding: Coat a streptavidin-coated microplate with the biotinylated primer/template [poly(rA)/oligo(dT)₁₈] in coating buffer overnight at 4°C. Wash. b. Reaction: Add a reaction mix containing: 50 mM Tris-HCl (pH 8.0), 5 mM MgCl₂, 1 mM DTT, 60 mM KCl, 50 µM dTTP, 5 µM biotin-16-dUTP, and purified RT (e.g., HIV-1 RT). Incubate at 37°C for 2 hours. c. Detection: Wash away unincorporated nucleotides. Add a conjugate of anti-digoxigenin peroxidase (POD). Wash. Add the POD substrate ABTS (2,2'-azino-di-[3-ethylbenzthiazoline sulfonate]). Incubate for 30-60 min. d. Measurement: Measure absorbance at 405-420 nm. Signal is proportional to RT activity. Include nucleoside (e.g., AZT-TP) and non-nucleoside (e.g., nevirapine) RT inhibitor controls.
Diagram 1: Critical Enzymatic Targets in Viral Life Cycles
Diagram 2: LAMP Assay Design for Different Viral Genomes
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Viral Target Research and LAMP Assay Development
| Category | Reagent/Kit | Primary Function in Research |
|---|---|---|
| Polymerases & Enzymes | Bst 2.0/3.0 DNA Polymerase | Strand-displacing polymerase essential for isothermal LAMP amplification. |
| Viral Polymerases (RdRp, RT) | Recombinant enzymes for high-throughput screening of antiviral inhibitors (e.g., HIV-1 RT, HCV NS5B, SARS-CoV-2 nsp12). | |
| T7 RNA Polymerase | For in vitro transcription to generate RNA templates for RdRp/RT assays. | |
| Nucleotides & Substrates | dNTP/NTP Mixes | Building blocks for nucleic acid synthesis. Modified versions (biotin-dUTP, fluorescent-dUTP) enable detection. |
| Nucleoside Analog Triphosphates | Positive controls for inhibition assays (e.g., Acyclovir-TP, Remdesivir-TP, AZT-TP). | |
| Primer & Probe Design | LAMP Primer Design Software | Tools for designing specific F3/B3, FIP/BIP, LF/LB primer sets against conserved viral targets. |
| Dual-Labeled Fluorescent Probes | For real-time, sequence-specific detection in RT-LAMP assays (e.g., using FAM/BHQ1). | |
| Detection & Signal Generation | Loopamp Fluorescent Detection Reagent | Magnesium pyrophosphate-based turbidity or fluorescent dye intercalation for endpoint detection. |
| Colorimetric pH Indicators | Phenol red or hydroxynaphthol blue for visual LAMP readout (pH change). | |
| Sample Prep & Controls | Viral RNA/DNA Extraction Kits | Magnetic bead or column-based purification of high-quality nucleic acids. |
| Synthetic Viral Gene Fragments | Non-infectious, sequence-accurate controls for assay validation and optimization. | |
| Cell-Based Assays | Reporter Virus Systems | Viral constructs with luciferase/GFP to study viral entry, replication, and inhibition in cell culture. |
Implementing LAMP Assays: Step-by-Step Protocols and Viral Application Case Studies
This technical guide details a standardized Loop-Mediated Isothermal Amplification (LAMP) protocol, formulated as a core methodological chapter for a broader thesis reviewing LAMP assays for rapid viral diagnostics. The reproducibility and accuracy of viral detection hinge on stringent standardization from sample preparation to interpretation. This protocol establishes that foundation.
Nucleic Acid Extraction: A Critical First Step
Consistent extraction is paramount for assay sensitivity. While commercial kits are prevalent, understanding the core principles is essential.
Detailed Protocol: Magnetic Bead-Based Extraction
This method is favored for its potential for automation and high purity yield.
Materials & Reagents:
- Lysis/Binding Buffer: Contains guanidinium thiocyanate (chaotropic salt) to denature proteins and nucleases, releasing RNA/DNA.
- Wash Buffer 1 (with ethanol): Removes salts, proteins, and other contaminants.
- Wash Buffer 2 (with ethanol): A second wash for further purification.
- Magnetic Silica Beads: Bind nucleic acids under high-salt conditions.
- Nuclease-Free Water or Elution Buffer: Low-salt solution to elute purified nucleic acid from beads.
- Absolute Ethanol & Proteinase K (optional): For complex samples.
Procedure:
- Lysis: Mix 200 µL of sample (e.g., viral transport medium, serum) with 300 µL Lysis/Binding Buffer and 20 µL Proteinase K (if needed). Vortex and incubate at 56°C for 10 min.
- Binding: Add 50 µL of homogenized magnetic bead suspension and 300 µL of 100% ethanol. Mix thoroughly. Incubate at room temperature (RT) for 5 min. Place on a magnetic stand until clear. Discard supernatant.
- Washing: With tube on magnet, add 500 µL Wash Buffer 1. Resuspend beads by pipetting. Return to magnet, discard supernatant. Repeat with 500 µL Wash Buffer 2. Perform a final wash with 80% ethanol. Air-dry bead pellet for 5-10 min.
- Elution: Remove from magnet. Resuspend beads in 50-100 µL Nuclease-Free Water or Elution Buffer. Incubate at 65°C for 5 min. Place on magnet and transfer eluted nucleic acid to a clean tube. Store at -80°C or proceed immediately.
Standardized LAMP Reaction Setup
Primer Design Principles
LAMP uses 4-6 primers targeting 6-8 distinct regions of the target sequence.
- F3/B3: Outer primers.
- FIP/BIP: Inner primers (FIP = F1c + F2; BIP = B1c + B2).
- LF/LB (Loop Primers): Accelerate reaction time.
Detailed Master Mix Preparation Protocol
Components for a 25 µL Reaction:
| Component | Final Concentration | Volume (µL) | Function |
|---|---|---|---|
| Isothermal Buffer (10X) | 1X | 2.5 | Provides optimal pH, salt conditions |
| MgSO₄ (100 mM) | 6-8 mM | 1.5-2.0 | Essential cofactor for Bst polymerase |
| dNTPs (10 mM each) | 1.4 mM | 3.5 | Building blocks for DNA synthesis |
| Betaine (5 M) | 0.8 M | 4.0 | Reduces secondary structure in GC-rich targets |
| FIP/BIP Primers (100 µM) | 1.6 µM each | 0.4 | Inner primers for strand displacement |
| F3/B3 Primers (100 µM) | 0.2 µM each | 0.05 | Outer primers for initiation |
| LF/LB Primers (100 µM) | 0.8 µM each | 0.2 | Loop primers for acceleration |
| Bst 2.0/3.0 Polymerase (8 U/µL) | 0.32 U/µL | 1.0 | Strand-displacing DNA polymerase |
| Fluorescent Dye (e.g., 20X SYTO 9) | 1X | 1.25 | Intercalating dye for real-time detection |
| Template DNA/RNA | -- | 5.0 | Extracted nucleic acid |
| Nuclease-Free Water | -- | to 25 µL | -- |
For RNA targets, add 1 µL (10 U) of WarmStart RTx Reverse Transcriptase per reaction.
Procedure:
- Thaw all components on ice. Prepare a master mix excluding the template in a nuclease-free tube.
- Mix gently by pipetting. Aliquot 20 µL of master mix into each reaction tube.
- Add 5 µL of template (or nuclease-free water for NTC) to each tube. Seal tightly.
- Centrifuge briefly to collect contents.
- Place tubes in a pre-heated isothermal fluorometer or heat block at 60-65°C.
Amplification & Real-Time Monitoring
- Temperature: 60-65°C for 30-60 minutes.
- Data Acquisition: Monitor fluorescence every 30-60 seconds.
- Threshold Time (Tt): The time (in minutes) at which fluorescence exceeds a baseline threshold. Correlates inversely with starting template concentration.
Result Interpretation & Validation
Quantitative Data Interpretation Table
| Result Type | Fluorescence Curve | Threshold Time (Tt) | Endpoint Colorimetric (e.g., HNB) | Gel Electrophoresis | Interpretation |
|---|---|---|---|---|---|
| Positive | Sigmoidal increase | < 30 minutes (assay-specific) | Color change (e.g., sky blue → violet) | Ladder-like pattern | Target sequence detected. |
| Negative | No increase (flat line) | Undetermined (or max time) | No color change (remains original) | No bands | Target not detected. |
| Invalid (Failed Run) | N/A | N/A | N/A | N/A | Positive control negative; repeat experiment. |
| Inhibition Suspected | Delayed Tt vs. control | High variability, > expected | Faint/ambiguous color | Faint ladder | Sample may contain inhibitors; dilute and re-test. |
Post-Amplification Analysis Protocols
A. Gel Electrophoresis (Confirmatory):
- Prepare a 2% agarose gel with 1X TAE and a safe DNA stain.
- Load 5-10 µL of LAMP product alongside a 100 bp DNA ladder.
- Run at 100V for 45-60 minutes.
- Visualize under UV. A positive shows a characteristic ladder of bands.
B. Specificity Verification (Melting Curve Analysis):
- After amplification, run a melting curve from 60°C to 95°C, rising by 0.1°C/sec.
- Plot negative derivative of fluorescence (-dF/dT) vs. Temperature.
- A single, sharp peak indicates specific amplification.
Visualization: Standardized LAMP Workflow & Mechanism
Title: Standardized LAMP Diagnostic Workflow
Title: LAMP Mechanism: Initiation and Cycling
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in Standardized LAMP | Example/Note |
|---|---|---|
| Bst 2.0/3.0 DNA Polymerase | Strand-displacing enzyme for isothermal amplification. Thermostable with high processivity. | New England Biolabs, WarmStart versions reduce non-specific activity. |
| WarmStart RTx Reverse Transcriptase | For RNA targets. Provides robust reverse transcription at isothermal temperatures (60-65°C). | Enables single-step RT-LAMP. |
| Isothermal Amplification Buffer | Optimized buffer with betaine, salts, and dNTPs. Stabilizes DNA and polymerase. | Often supplied with the polymerase. Critical for efficiency. |
| LAMP Primer Mix (Lyophilized) | Pre-designed, sequence-specific primer sets (FIP, BIP, F3, B3, LF, LB). Ensures reproducibility. | Available from suppliers like IDT, Metabion for common viral targets. |
| Fluorescent Intercalating Dye | Real-time detection. Binds dsDNA, fluorescence increases with product formation. | SYTO 9, SYTO 82, EvaGreen. Prefer low inhibition dyes. |
| Colorimetric pH Indicator | Endpoint visual detection. Mg²⁺ chelation during amplification causes pH drop and color change. | Hydroxy Naphthol Blue (HNB), Phenol Red. Simplifies field use. |
| Magnetic Bead Purification Kit | For standardized nucleic acid extraction. Balances yield, purity, and inhibitor removal. | MagMAX (Thermo), NucleoMag (Macherey-Nagel). Enable high-throughput. |
| Synthetic Positive Control | Non-infectious RNA/DNA containing target sequence. Essential for assay validation and QC. | Armored RNA, gBlock gene fragments. |
| Internal Control (IC) | Non-target nucleic acid co-extracted and co-amplified. Distinguishes true negative from inhibition. | MS2 phage, synthetic alien sequence. |
Primer Design Best Practices for High Specificity and Sensitivity to Viral Sequences
Within the broader thesis on the optimization of Loop-Mediated Isothermal Amplification (LAMP) assays for rapid viral diagnostics, the design of primers stands as the most critical determinant of success. Achieving high specificity (minimizing off-target amplification) and high sensitivity (detecting low viral copy numbers) is paramount for reliable field-deployable diagnostics. This technical guide outlines best practices grounded in current bioinformatics and empirical research, specifically tailored for viral target sequences.
Foundational Principles for Viral Primer Design
Viral genomes present unique challenges, including high mutation rates, sequence homology among strains, and, for RNA viruses, the need to target conserved regions across quasi-species. Effective primer design must account for these factors from the outset.
Key Parameters:
- Length: LAMP primers (FIP/BIP) are typically 40-45 bp, with F3/B3 primers 18-21 bp.
- Tm: The melting temperature (Tm) of the annealing regions is crucial.
- F3/B3 Tm: 55-60°C.
- F2/B2 (within FIP/BIP) Tm: 60-65°C.
- F1c/B1c (within FIP/BIP) Tm: 65-70°C.
- The Tm of F1c/B1c should be 5-10°C higher than that of F2/B2.
- GC Content: Maintain 40-60% for stable priming. Aim for higher GC content in the F1c/B1c regions to facilitate loop formation.
- ΔG: The free energy of the 3' ends (especially of F2/B2) should be higher (more negative, indicating stronger binding) than that of the 5' ends to ensure efficient strand displacement initiation.
Strategic Selection of Target Regions
Sensitivity requires targeting conserved regions; specificity requires discriminating against host and closely related viral genomes.
Procedure:
- Multiple Sequence Alignment (MSA): Collect all available genomic sequences for the target virus and related strains/family members from databases (NCBI Virus, GISAID).
- Identify Conserved Blocks: Use tools like Clustal Omega or MAFFT to perform MSA. Manually inspect or use conservation scoring to identify >200 bp regions with >95% sequence identity across all target strains.
- Exclude Problematic Motifs: Screen conserved blocks for homopolymers (runs of >4 identical nucleotides), which promote slippage, and internal repeats.
- Specificity Check: Perform an initial in-silico BLASTN against the host genome (e.g., human, animal) and a non-redundant nucleotide database to ensure minimal off-target hits.
In-Silico Design and Validation Workflow
A robust computational pipeline is non-negotiable. The following workflow integrates current best-practice tools.
Title: Computational Primer Design & Validation Workflow
Quantitative Design Parameters & Benchmarks
Table 1: Optimal Thermodynamic and Sequence Parameters for LAMP Primers
| Parameter | F3/B3 Primers | F2/B2 Regions (in FIP/BIP) | F1c/B1c Regions (in FIP/BIP) | Rationale |
|---|---|---|---|---|
| Length (bp) | 18-21 | 18-22 | 19-25 | F1c/B1c length ensures stable loop structure. |
| Melting Temp (Tm, °C) | 55-60 | 60-65 | 65-70 | Hierarchical Tm ensures proper order of strand displacement. |
| GC Content (%) | 40-60 | 40-60 | 45-65 | Higher GC in F1c/B1c stabilizes the initial loop. |
| 3'-end ΔG (kcal/mol) | ≥ -9 | ≤ -11 (stronger) | N/A | Strong 3' end binding on F2/B2 initiates displacement. |
| Inter-Primer Tm Difference | ≤ 2 (within F3/B3 set) | ≤ 3 (within F2/B2 set) | ≤ 3 (within F1c/B1c set) | Ensures synchronous hybridization. |
| Amplicon Length (bp) | Overall: 120-200 bp (between F2 & B2) | Shorter amplicons enhance speed and yield. |
Experimental Protocol for Empirical Validation
Protocol: LAMP Reaction Setup & Specificity/Sensitivity Testing
A. Specificity Testing (Against Near-Neighbors):
- Template Preparation: Extract genomic material from the target virus and a panel of non-target viruses (including close genetic relatives). Adjust all concentrations to 10^4 copies/μL.
- LAMP Master Mix: Prepare a reaction mix per 25 μL: 1.6 μM each FIP/BIP, 0.2 μM each F3/B3, 0.8 μM each LF/LB (if used), 1.4 mM dNTPs, 6 mM MgSO4, 8 U Bst 2.0 WarmStart DNA Polymerase (or equivalent), 1X isothermal amplification buffer, 1X fluorescent intercalating dye (e.g., SYTO-9).
- Amplification: Aliquot 23 μL of master mix. Add 2 μL of each template (or nuclease-free water for NTC). Incubate at 63-65°C for 60 minutes in a real-time fluorometer.
- Analysis: Specificity is confirmed by amplification only in the target virus wells. Analyze post-amplification melt curves (65-95°C) to ensure a single, specific product.
B. Limit of Detection (LoD) Determination:
- Standard Curve: Prepare a serial 10-fold dilution of target viral RNA/DNA (or synthetic gBlock) from 10^6 to 10^0 copies/μL. Use a validated digital PCR method for absolute quantification of the stock.
- Amplification: Run LAMP as above with 8 replicates per dilution.
- Data Analysis: Calculate the proportion of positive replicates at each dilution. The LoD (at 95% confidence) is the lowest concentration where ≥95% of replicates are positive. Use probit or logistic regression analysis.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for High-Performance Viral LAMP Assay Development
| Reagent / Material | Function & Importance | Example Product |
|---|---|---|
| High-Fidelity Bst Polymerase | Engineered for superior strand displacement activity and tolerance to inhibitors, crucial for complex clinical samples. | Bst 2.0 WarmStart, Bst 3.0 |
| Synthetic Viral Genome Fragments | Precisely quantified gBlocks or Twist Fragments for LoD determination, avoiding biosafety constraints of live virus. | IDT gBlocks, Twist Synthetic DNA |
| Fluorescent Intercalating Dyes | Real-time monitoring of amplification. SYTO dyes (e.g., SYTO-9) are preferred over SYBR Green as they are less inhibitory to Bst polymerase. | SYTO-9, EvaGreen |
| RNase Inhibitor (for RNA viruses) | Critical for one-step RT-LAMP to protect viral RNA template from degradation during reaction setup. | Murine RNase Inhibitor, Recombinant RNasin |
| Uracil-DNA Glycosylase (UDG) | Carryover contamination prevention. Use dUTP in place of dTTP in master mix; UDG cleaves uracil-containing amplicons from prior runs before amplification. | Heat-labile UDG |
| Internal Amplification Control (IAC) | Non-competitive synthetic template spiked into each reaction to distinguish true negatives from reaction failure. | Custom-designed IAC with distinct probe |
Advanced Considerations for Multiplexing and Inhibition Resilience
Multiplex LAMP Design: For detecting multiple viral targets, primer sets must be rigorously checked for cross-dimerization. Tag primers with unique 5' overhangs; subsequent detection can be via array hybridization or multicolor fluorescence.
Title: Multiplex LAMP Detection Strategy Options
Addressing Inhibition: Incorporate additives like 0.2 M trehalose (stabilizer) or 0.1% Tween-20 (reduces adsorption). Pre-treat samples with simple dilution or heat (e.g., 95°C for 5 min) to inactivate common inhibitors in saliva or blood.
In the context of advancing LAMP for rapid viral diagnostics, meticulous primer design is the cornerstone. By integrating comprehensive in-silico analysis of conserved viral regions with stringent thermodynamic optimization and empirical validation against key performance benchmarks (specificity, sensitivity, LoD), researchers can develop robust assays. The integration of advanced reagents, such as high-fidelity polymerases and contamination controls, further translates precise design into reliable diagnostic performance in complex sample matrices.
Within the paradigm of rapid, isothermal nucleic acid amplification, Loop-Mediated Isothermal Amplification (LAMP) has emerged as a frontrunner for point-of-care and laboratory-based viral diagnostics. The core amplification reaction, while robust, necessitates equally robust and versatile detection modalities to translate results into an interpretable signal. This technical guide examines four principal detection methods—turbidity, fluorescence, lateral flow strips (LFS), and colorimetric dyes—detailing their mechanisms, protocols, and applications within the context of LAMP-based viral diagnostics research.
Core Detection Modalities: Mechanisms and Protocols
Turbidity (Real-Time Monitoring)
Mechanism: The turbidity method exploits the precipitation of magnesium pyrophosphate, a by-product of DNA amplification. As the LAMP reaction proceeds, the accumulation of this insoluble salt increases the turbidity of the reaction mixture, which can be monitored in real-time using a spectrophotometer or a simple optical device.
Detailed Protocol:
- Prepare a standard LAMP master mix containing 1.4-1.6 mM of each dNTP, 6-8 mM MgSO₄, and target-specific primers.
- Aliquot 25 µL of the master mix into appropriate reaction tubes or a microplate.
- Initiate the reaction at 60-65°C.
- Place the reaction vessel in a real-time turbidimeter or a spectrophotometer equipped with a heated chamber.
- Monitor the optical density (OD) at 400 nm every 6 seconds for 60 minutes.
- A positive reaction is indicated by a sharp increase in OD, with the time to positivity (Tp) being inversely proportional to the initial target concentration.
Fluorescence (Real-Time and Endpoint)
Mechanism: Fluorescent detection involves the use of DNA-intercalating dyes (e.g., SYTO-9, EvaGreen) or sequence-specific probes. Intercalating dyes bind to double-stranded DNA (dsDNA) produced during amplification, leading to a significant increase in fluorescence intensity.
Detailed Protocol (Using Intercalating Dye):
- Prepare LAMP master mix as above, supplemented with 0.5-1X concentration of a dsDNA-binding fluorescent dye (e.g., EvaGreen).
- Protect the reaction mixture from light to prevent dye photobleaching.
- Load 25 µL into optically clear tubes or a qPCR plate.
- Run the reaction in a real-time isothermal fluorimeter or a standard real-time PCR machine with isothermal settings (60-65°C).
- Acquire fluorescence data (excitation/emission specific to the dye, e.g., ~500/530 nm for SYTO-9) at 1-minute intervals.
- The amplification curve and cycle threshold (Ct) or Tp value are used for quantitative analysis.
Lateral Flow Strips (Endpoint)
Mechanism: LFS detection employs biotin- and FAM-labeled primers during the LAMP reaction. The amplicon product is decorated with these tags. The strip contains immobilized anti-FAM antibodies at the test line and streptavidin at the control line. A conjugated anti-FAM antibody coupled to a colored nanoparticle (e.g., gold) binds and visualizes the captured amplicon.
Detailed Protocol:
- Perform LAMP using forward inner primers (FIP) labeled with biotin and backward inner primers (BIP) labeled with fluorescein (FAM).
- After amplification, dilute 5 µL of the product with 95 µL of the provided running buffer.
- Insert the lateral flow strip into the diluted solution.
- Allow capillary action to run the solution up the strip for 5-10 minutes.
- Interpretation: The appearance of both a control line (C) and a test line (T) indicates a positive result. Only a control line indicates a negative result. No control line indicates an invalid test.
Colorimetric Dyes (Endpoint)
Mechanism: pH-sensitive dyes (e.g., phenol red, hydroxynaphthol blue) or metal ion indicators detect by-products of amplification. The massive synthesis of DNA during LAMP releases protons (H⁺), acidifying the reaction. A pre-optimized buffer with a pH indicator shows a visible color change.
Detailed Protocol (Using Phenol Red):
- Prepare LAMP master mix using a specially formulated buffer containing 125 µM phenol red and 6-8 mM MgSO₄. The initial pH is ~8.5 (red/pink).
- Aliquot the master mix and add template.
- Incubate at 65°C for 30-60 minutes.
- Visually inspect the color change.
- Interpretation: A positive result is indicated by a color change from pink/red to yellow. A negative result retains the original pink/red color. Care must be taken to avoid over-amplification, which can lead to false positives in negative samples due to non-specific amplification.
Quantitative Data Comparison
Table 1: Comparative Analysis of LAMP Detection Modalities
| Modality | Measurement Type | Approx. Limit of Detection (LoD) | Time to Result | Equipment Needs | Key Advantage | Key Limitation |
|---|---|---|---|---|---|---|
| Turbidity | Real-time | 10-100 copies/µL | 15-45 min | Turbidimeter, spectrophotometer | Label-free, simple chemistry | Less sensitive, prone to background from precipitate aggregation |
| Fluorescence | Real-time/Endpoint | 1-10 copies/µL | 10-30 min | Fluorimeter, qPCR machine | High sensitivity, quantitative, real-time kinetics | Requires optical equipment, dye inhibition possible |
| Lateral Flow Strips | Endpoint | 10-100 copies/µL | 40-70 min (inc. LAMP) | None for readout | User-friendly, portable, binary result | Semi-quantitative at best, extra labeling steps |
| Colorimetric (pH) | Endpoint | 100-1000 copies/µL | 30-60 min | None | Extremely simple, visual readout, low-cost | Lowest sensitivity, prone to pH contamination, subjective interpretation |
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for LAMP Detection Development
| Reagent / Material | Function in LAMP Detection |
|---|---|
| Bst 2.0/3.0 Polymerase | Thermostable DNA polymerase with high strand displacement activity, essential for isothermal amplification. |
| dNTP Mix | Deoxynucleotide triphosphates (dATP, dCTP, dGTP, dTTP) providing the building blocks for DNA synthesis. |
| MgSO₄ or MgCl₂ | Source of Mg²⁺ ions, a critical cofactor for polymerase activity. Concentration finely tunes reaction efficiency and detection (turbidity/colorimetric). |
| Betaine | Additive that destabilizes DNA secondary structures, improving primer annealing and reaction specificity, especially for GC-rich targets. |
| SYTO-9 / EvaGreen Dye | dsDNA-binding fluorescent dyes for real-time or endpoint fluorescence detection. |
| FAM- and Biotin-Labeled Primers | Modified primers for subsequent detection of amplicons on lateral flow strips. |
| Phenol Red / HNB Dye | pH indicator dyes for visual, colorimetric endpoint detection based on reaction acidification. |
| Lateral Flow Strips | Pre-fabricated nitrocellulose strips with immobilized capture lines for specific detection of labeled amplicons. |
| Isothermal Fluorimeter | Dedicated device for precise temperature control and real-time fluorescence monitoring of multiple samples. |
Visualizing Detection Workflows
Turbidity Detection Pathway
Fluorescence Detection Pathway
Lateral Flow Strip Detection Workflow
Colorimetric pH-based Detection Pathway
This technical guide details the application of Loop-Mediated Isothermal Amplification (LAMP) for detecting major respiratory viruses—SARS-CoV-2, Influenza (A/B), and Respiratory Syncytial Virus (RSV). Within the broader thesis reviewing LAMP for rapid viral diagnostics, this spotlight underscores the assay's pivotal role in enabling high-throughput, point-of-care, and decentralized testing. It exemplifies the transition from central lab PCR to isothermal methods that balance sensitivity, specificity, speed, and operational simplicity, directly addressing pandemic and epidemic response needs.
Core Principles and Advantages of LAMP
LAMP amplifies nucleic acids at a constant temperature (60-65°C) using a DNA polymerase with high strand displacement activity and 4-6 primers targeting 6-8 distinct regions of the target gene. The reaction produces magnesium pyrophosphate as a by-product, leading to turbidity, and can be coupled with colorimetric or fluorescent indicators for real-time or endpoint detection.
Key Advantages for Respiratory Virus Diagnostics:
- Isothermal: Eliminates need for thermal cyclers.
- Rapid: Results in 15-60 minutes.
- Robust: Tolerant to some inhibitors in crude samples.
- High Sensitivity/Specificity: Due to multiple primer sets.
- Versatile Detection: Turbidity, fluorescence, or colorimetric (pH-sensitive dyes) readouts.
Quantitative Performance Data: LAMP vs. RT-qPCR
Table 1: Comparative Analytical Performance of LAMP Assays for Respiratory Viruses
| Virus Target | Assay Name/Type | Limit of Detection (LoD) | Time to Result | Clinical Sensitivity | Clinical Specificity | Reference |
|---|---|---|---|---|---|---|
| SARS-CoV-2 (N, E, Orf1ab genes) | Colorimetric LAMP | 10-100 RNA copies/µL | 30-40 min | 97.5% - 99.2% | 99.6% - 100% | (Recent studies, 2023-24) |
| Influenza A (M gene) | Real-time Fluorescent LAMP | 5-50 RNA copies/µL | 20-25 min | 98.1% | 99.3% | (Recent studies, 2023-24) |
| Influenza B (HA gene) | RT-LAMP with Lateral Flow | ~100 RNA copies/µL | ~45 min | 96.7% | 100% | (Recent studies, 2023-24) |
| RSV (N gene) | Multiplex LAMP (A/B) | 50-100 RNA copies/µL | 30 min | 98.8% | 99.4% | (Recent studies, 2023-24) |
| SARS-CoV-2/ Flu A/B | Multiplex RT-LAMP | 50-200 copies/µL per target | 35-50 min | >95% for all targets | >99% | (Recent studies, 2023-24) |
Detailed Experimental Protocol: Multiplex Colorimetric RT-LAMP
Objective: Simultaneous detection of SARS-CoV-2 (Orf1ab), Influenza A (M), and RSV (N) from extracted RNA.
I. Reagent Preparation (25 µL Reaction)
- Isothermal Buffer: 1.6 µM each inner primer (FIP/BIP), 0.2 µM each outer primer (F3/B3), 0.4 µM each loop primer (LF/LB) per target.
- Master Mix: Combine on ice:
- 12.5 µL 2x Isothermal Master Mix (contains Bst 2.0/3.0 DNA polymerase, reverse transcriptase, dNTPs)
- 2.5 µL Primer Mix (for all three targets)
- 1 µL Colorimetric Indicator (e.g., phenol red, 120 µM)
- 2 µL 100mM MgSO4 (optimized concentration)
- 2 µL RNA template
- Nuclease-free water to 25 µL.
II. Amplification & Detection
- Incubation: Place reaction tubes in a dry block heater or water bath at 65°C for 40 minutes.
- Colorimetric Readout: Visual inspection.
- Positive (No amplification): Pink (original color, high pH).
- Negative (Amplification): Yellow (acidic pH from pyrophosphate production).
- Validation: Include no-template control (NTC, water) and positive synthetic controls for each target.
III. Post-Amplification Analysis (Optional)
- Run 5 µL of product on a 2% agarose gel. LAMP yields a characteristic ladder pattern.
Diagrams of Key Processes
Diagram 1: Core LAMP Amplification Mechanism (79 chars)
Diagram 2: Typical Workflow for Respiratory Virus LAMP Test (70 chars)
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagent Solutions for LAMP-Based Respiratory Virus Research
| Reagent / Material | Function & Rationale | Example / Notes |
|---|---|---|
| Bst 2.0 or 3.0 DNA Polymerase | Engineered for robust strand displacement activity at isothermal temps (60-65°C). Core enzyme for LAMP. | Often supplied with isothermal buffer. Bst 3.0 is faster and more robust. |
| WarmStart Reverse Transcriptase | Efficiently synthesizes cDNA from viral RNA at LAMP reaction temperature. Enables single-step RT-LAMP. | Engineered to be inactive at room temp, preventing primer-dimer formation. |
| LAMP Primer Sets (F3/B3, FIP/BIP, LF/LB) | 4-6 primers per target ensure high specificity by recognizing 6-8 distinct regions. Critical for multiplexing. | Must be meticulously designed (e.g., using PrimerExplorer). HPLC-purified recommended. |
| Colorimetric pH Indicator | Enables visual, instrument-free detection. Proton release during amplification lowers pH, changing dye color. | Phenol red (pink→yellow), hydroxy naphthol blue (violet→blue). |
| Fluorescent Intercalating Dye | For real-time monitoring/quantification. Binds to double-stranded LAMP products, increasing fluorescence. | SYTO-9, EvaGreen, Calcein with MnCl2. |
| Rapid RNA Extraction Kit | Purifies viral RNA from swabs/saliva. Essential for sensitivity. Can be simplified for point-of-care. | Magnetic bead-based or spin-column kits. Some protocols use heat/chelation only. |
| Synthetic RNA Controls | Quantified in vitro transcribed RNA for assay development, calibration, and determining Limit of Detection (LoD). | Must include exact primer target sequences for each virus. |
| Nuclease-Free Water & Tubes | Prevents degradation of RNA, primers, and enzymes. Critical for reproducibility. | Certified DEPC-treated/PCR-grade water. |
This whitepaper, framed within a broader thesis on LAMP assay for rapid viral diagnostics, details the application of Loop-Mediated Isothermal Amplification (LAMP) for the detection of arboviruses (Dengue, Zika) and viral hemorrhagic fevers (Ebola). LAMP’s isothermal nature, high sensitivity, and compatibility with simple detection methods make it a transformative technology for field-deployable, rapid diagnosis in outbreak settings, directly addressing the limitations of conventional PCR in resource-limited areas.
Core Principles and Advantages of LAMP
LAMP amplifies DNA with high specificity and efficiency under isothermal conditions (60-65°C) using a DNA polymerase with strand displacement activity and 4-6 primers targeting 6-8 distinct regions. Key advantages include:
- Isothermal Amplification: Eliminates need for thermal cyclers.
- High Tolerance to Inhibitors: More robust with crude samples (e.g., blood, saliva).
- Rapid Kinetics: Results often in 15-60 minutes.
- Versatile Detection: Can use turbidity (pyrophosphate precipitation), fluorescence (intercalating dyes), or colorimetric (pH indicators) readouts.
Target Pathogens and Assay Performance
Recent developments have produced highly specific LAMP assays for the target pathogens. Quantitative performance data from recent studies (2022-2024) are summarized below.
Table 1: Comparative Performance of Recent LAMP Assays for Target Viruses
| Virus (Target) | Assay Name/Type | Limit of Detection (LoD) | Time to Result | Specificity | Key Reference (Year) |
|---|---|---|---|---|---|
| Dengue (All Serotypes) | Multiplex Colorimetric RT-LAMP | 10-100 RNA copies/µL | ~30 min | 100% (no cross-reactivity with ZIKV, CHIKV, YFV) | (Valdez et al., 2023) |
| Zika (prM gene) | Real-time Fluorescent RT-LAMP | 5 RNA copies/reaction | 20 min | 100% (vs DENV, YFV, WNV) | (Fernandez et al., 2024) |
| Ebola (NP gene) | Portable RT-LAMP with CRISPR-readout | 2 RNA copies/µL | 40 min (incl. CRISPR step) | Distinguishes SUDV, BDBV, EBOV | (Kaur et al., 2022) |
Detailed Experimental Protocol: Multiplex Dengue Serotyping RT-LAMP
This protocol is adapted from recent high-impact studies.
I. Research Reagent Solutions (The Scientist's Toolkit)
| Item | Function/Explanation |
|---|---|
| WarmStart LAMP Kit (DNA & RNA) | Contains Bst 2.0/WarmStart RTx polymerase, optimized buffer, dNTPs. Engineered for room-temperature setup and hot-start activation. |
| Primer Mix (F3/B3, FIP/BIP, LF/LB per serotype) | Target 8 distinct regions of the DENV genome. Designed using software (e.g., PrimerExplorer V5) for specific serotype identification. |
| Phenol Red pH Indicator | Colorimetric reporter. Reaction mix turns from red (pH ~8.8) to yellow (pH ~6.8) upon amplification-induced acidification. |
| RNase-free Water | Nuclease-free water for reagent dilution and sample preparation. |
| Synthetic RNA Controls | In vitro transcribed RNA for each DENV serotype (DENV-1-4) for assay validation and standard curve generation. |
| Nucleic Acid Extraction Kit (Magnetic Bead-based) | For purifying viral RNA from clinical samples (serum, plasma). Compatible with field-deployable extraction systems. |
| Portable Dry Bath Incubator | Maintains constant isothermal temperature (63°C) for amplification. |
II. Step-by-Step Workflow
- RNA Extraction: Extract viral RNA from 140 µL of patient serum using a magnetic bead-based kit. Elute in 60 µL of elution buffer.
- Master Mix Preparation: For a 25 µL reaction, combine:
- 12.5 µL 2x WarmStart LAMP Master Mix
- 1.5 µL Primer Mix (equimolar FIP/BIP, 2x F3/B3, 1x LF/LB for each serotype)
- 0.5 µL 1mM Phenol Red
- 5.5 µL Nuclease-free Water
- Reaction Assembly: Aliquot 20 µL of Master Mix into each tube. Add 5 µL of extracted RNA template. Include negative (water) and positive (synthetic RNA) controls.
- Amplification: Incubate reactions at 63°C for 40 minutes in a dry bath or portable block incubator.
- Result Interpretation: Visual color change from pink/red to yellow indicates a positive reaction. No color change indicates a negative reaction. Results can be documented with a smartphone camera.
Technological Integration and Workflow Visualization
Diagram 1: Integrated Field LAMP Diagnostic Workflow (76 chars)
Molecular Mechanism of LAMP Amplification
Diagram 2: LAMP Molecular Mechanism: Initiation & Cycling (74 chars)
LAMP represents a paradigm shift for rapid, point-of-need diagnosis of critical viruses like Dengue, Zika, and Ebola. Its integration with lyophilized reagents, portable heaters, and smartphone-based readouts creates a complete field-deployable system. Future directions include integrating LAMP with CRISPR-Cas systems for enhanced specificity (as seen in Ebola protocols) and developing microfluidic chips for true sample-to-answer automation. This evolution solidifies LAMP's role as a cornerstone technology in global outbreak response and pandemic preparedness frameworks.
Within the landscape of rapid viral diagnostics, isothermal amplification techniques, notably Loop-Mediated Isothermal Amplification (LAMP), have emerged as powerful alternatives to PCR, particularly for point-of-care (POC) applications. A critical research thrust in this domain is the integration of two core hardware components: portable, precise heaters for incubation and smartphone-based systems for result readout. This integration aims to create compact, cost-effective, and field-deployable diagnostic platforms. This technical guide examines the core engineering and methodological principles of these integrated systems, framed within a broader thesis on advancing LAMP assays for rapid viral detection.
Core System Components & Technical Specifications
Portable Heating Modules
Portable heaters for LAMP must maintain a stable temperature between 60-65°C for 15-60 minutes. Contemporary designs favor Peltier (thermoelectric) modules or resistive heating elements paired with microcontroller-based feedback control (e.g., using PID algorithms).
Table 1: Comparison of Portable Heating Technologies for LAMP Assays
| Technology | Typical Accuracy (°C) | Time to Setpoint | Power Consumption | Key Advantage | Common Control System |
|---|---|---|---|---|---|
| Peltier Module | ±0.2 – 0.5 | 1-3 minutes | Medium-High (5-30W) | Active cooling capability | PID via Arduino/Raspberry Pi |
| Resistive Heater | ±0.5 – 1.0 | 30 seconds - 2 minutes | Low-Medium (2-10W) | Simplicity, low cost | On/Off or PID via microcontroller |
| Chemical Heater (Exothermic) | ±2.0 – 5.0 | Instant | N/A | Ultra-low cost, no electricity | None (passive) |
Smartphone-Based Readout Systems
Readout methods leverage smartphone cameras and processing power. Quantitative data is achieved via colorimetric, fluorometric, or lateral flow detection.
Table 2: Smartphone Readout Modalities for LAMP Assays
| Detection Method | Target Signal | Typical LOD (copies/µL) | Smartphone Role | Required Accessories |
|---|---|---|---|---|
| Colorimetric (pH) | Color change (e.g., phenol red: pink→yellow) | 10 - 100 | Capture image, RGB analysis via app | Portable heater, uniform lighting box |
| Fluorometric (Intercalating Dye) | Fluorescence intensity (e.g., SYBR Green, Calcein) | 1 - 10 | Capture fluorescence image, pixel intensity analysis | LED excitation source, emission filter, dark box |
| Lateral Flow Assay (LFA) | Test line intensity | 10 - 50 | Capture image, line intensity analysis via app | LFA strip reader attachment |
| Turbidity (Magnesium Pyrophosphate) | White precipitate turbidity | 50 - 100 | Capture image, grayscale analysis | Dark background, consistent lighting |
Experimental Protocols for Integrated System Validation
Protocol: Colorimetric LAMP with Portable Heater & Smartphone Readout
Objective: To detect a target viral sequence (e.g., SARS-CoV-2 N gene) using an integrated POC system.
Materials:
- Portable Heater: Peltier-based module with PID control (Arduino Nano), set to 65°C.
- Reaction Tube: Contains LAMP master mix with phenol red indicator.
- Smartphone: Android/iOS device with custom app (e.g., Color Grab, or open-source ImageJ-based app).
- 3D-Printed Imaging Box: Provides uniform, diffuse LED white light.
Procedure:
- Sample Preparation: Spiked synthetic viral RNA into the LAMP reaction mix.
- Amplification: Place reaction tube in portable heater. Incubate at 65°C for 30 minutes.
- Image Capture: Post-incubation, immediately transfer tube to the standardized imaging box. Capture image with smartphone camera under fixed settings (ISO, white balance).
- Analysis: The app extracts average RGB values from a region of interest (ROI) around the reaction tube. The R/B ratio is calculated. A decrease in ratio (pink to yellow) indicates a positive amplification.
- Quantification: A standard curve is generated using known RNA copy numbers to correlate R/B ratio with viral load.
Protocol: Fluorometric LAMP with Low-Cost Reader Attachment
Objective: To achieve quantitative endpoint detection with higher sensitivity.
Materials:
- Heater: As in 3.1.
- Smartphone Fluorometer Attachment: 3D-printed housing containing a ~470 nm LED for excitation, a diffuser, and a long-pass emission filter (>500 nm) placed over the camera lens.
- Reagent: LAMP mix with SYBR Green I dye (added post-amplification to prevent inhibition).
Procedure:
- Amplification: Perform LAMP in the portable heater (SYBR Green excluded).
- Dye Addition: Add diluted SYBR Green I to the reaction tube.
- Image Capture: Place tube in the dark fluorometer attachment. Capture image using the smartphone with LED on.
- Analysis: The app converts the image to grayscale and measures the average pixel intensity within the tube ROI. Intensity is proportional to amplicon concentration.
Visualizing the Integrated Workflow and Signaling
Diagram 1: Integrated POC LAMP Assay Workflow
Diagram 2: LAMP Signal Generation Pathways
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for POC LAMP Integration Research
| Item | Function/Description | Example Product/Note |
|---|---|---|
| Bst 2.0/3.0 Polymerase | Thermostable DNA polymerase with strand displacement activity essential for LAMP. | New England Biolabs WarmStart Bst 2.0/3.0 for robust room-temperature setup. |
| LAMP Primer Mix | Set of 4-6 primers (F3/B3, FIP/BIP, LF/LB) targeting 6-8 distinct regions of the viral genome. | Custom designed using PrimerExplorer V5 software; lyophilized for POC stability. |
| Colorimetric Indicator | pH-sensitive dye for visual readout without opening tubes. | Phenol Red, Hydroxy Naphthol Blue (HNB), or ready-to-use master mixes containing dye. |
| Fluorometric Dye | Intercalating dye that fluoresces upon binding dsDNA. | SYBR Green I (add post-amplification), Calcein (with Mn2+, integrated in mix). |
| Lyophilized Reaction Beads | Pre-formulated, stable pellets containing all LAMP reagents except sample. Enable long-term storage without refrigeration. | Critical for true field deployment; often require proprietary stabilizers. |
| Positive Control Template | Synthetic DNA or RNA containing the target sequence. Used for assay validation and standard curve generation. | GBlocks, Twist Synthetic Controls, or in vitro transcribed RNA. |
| RNase/DNase Inactivation Buffer | For safe and simple viral lysis and nucleic acid stabilization directly from swab samples. | Guanidinium thiocyanate-based buffers (e.g., from Lucigen or Thermo Fisher). |
| Microcontroller Dev Board | For building and programming custom portable heaters. | Arduino Nano, ESP32 (adds Bluetooth/Wi-Fi for smartphone control). |
| Optical Filters & LEDs | For building smartphone fluorometer attachments. | 470 nm LED, 500 nm long-pass emission filter (e.g., from Thorlabs). |
Optimizing LAMP Performance: Solving Common Pitfalls and Enhancing Assay Robustness
Identifying and Mitigating Non-Specific Amplification and Primer-Dimer Artifacts
Within the broader thesis on the review of Loop-Mediated Isothermal Amplification (LAMP) for rapid viral diagnostics, the challenge of non-specific amplification and primer-dimer formation remains a critical barrier to assay specificity and reliability. These artifacts lead to false-positive results, reduced sensitivity, and compromised quantitative accuracy, directly impacting diagnostic confidence. This technical guide details the mechanisms, identification strategies, and mitigation protocols essential for developing robust LAMP-based viral diagnostic assays.
Non-specific amplification in LAMP arises from unintended primer-template interactions, often due to partial homology with non-target genomic regions or mispriming at low annealing stringency. Primer-dimers form when primers anneal to each other via complementary bases, particularly at their 3' ends, and are extended by the strand-displacing DNA polymerase. The isothermal conditions of LAMP can exacerbate these issues compared to PCR, as the constant temperature may favor spurious initiation.
Identification and Characterization Methods
Post-Amplification Analysis
Gel Electrophoresis: Non-specific products appear as a smear or multiple bands distinct from the target laddering pattern typical of LAMP. Primer-dimers manifest as a low molecular weight band (~50-100 bp). Melting Curve Analysis: Using intercalating dyes like SYBR Green or EvaGreen in real-time LAMP. Non-specific products and primer-dimers often exhibit lower, broader melting temperatures (Tm) than the true target amplicon. Sequencing: The definitive confirmation method. Cloning and sequencing of non-target bands confirms their origin.
Real-Time Monitoring Artifacts
Artifacts cause early amplification curves with abnormal sigmoidal shapes or high baselines. The time-to-positive (Tp) is inconsistent across replicates.
Table 1: Characteristics of Artifacts vs. Specific LAMP Amplication
| Feature | Specific LAMP Product | Non-Specific Amplification | Primer-Dimer |
|---|---|---|---|
| Gel Electrophoresis | Characteristic ladder pattern | Smear or discrete non-ladder bands | Single low MW band (~50-100bp) |
| Melting Temperature (Tm) | High, sharp peak | Variable, often lower/ broader peak | Very low Tm (<80°C) |
| Real-Time Curve Shape | Steep, sigmoidal | Shallower, irregular rise | Early, low-amplitude rise |
| Sequence Homology | 100% to target | Partial homology to target/genome | Primer-derived sequence |
Diagram 1: Workflow for Identifying Amplification Artifacts
Experimental Protocols for Detection
Protocol 3.1: Two-Step Melting Curve Analysis for LAMP
- Amplification: Perform real-time LAMP in a thermal cycler capable of melt curve acquisition (e.g., QuantStudio 5, CFX96) using a dye like 1X EvaGreen. Use standard cycling: 65°C for 40-60 min.
- Melt Curve Step: After amplification, ramp temperature from 65°C to 95°C at 0.1°C/sec with continuous fluorescence acquisition.
- Analysis: Plot the negative derivative of fluorescence (-dF/dT) vs. Temperature. A single sharp peak at high Tm (often >85°C) indicates specificity. Multiple peaks or a broad low-Tm peak suggests artifacts.
Protocol 3.2: Gradient LAMP for Stringency Optimization
- Setup: Prepare a master mix with target template at a concentration near the assay's limit of detection.
- Reaction: Dispense identical reactions across a thermal gradient block (e.g., 60°C to 68°C in 0.5°C increments).
- Analysis: Determine Tp for each temperature. Plot Tp vs. Temperature. The optimal temperature provides the lowest Tp without promoting non-specific amplification in no-template controls (NTCs).
Mitigation Strategies and Protocols
1In SilicoPrimer Design Optimization
Utilize tools like PrimerExplorer (Eiken), NUPACK, or IDT OligoAnalyzer to assess primer interactions. Key parameters: minimize 3' complementarity (especially ΔG > -5 kcal/mol for dimer formation), avoid long homopolymeric stretches, and ensure high specificity via BLAST against the host genome.
Wet-Lab Optimization
Table 2: Mitigation Strategies and Experimental Approaches
| Strategy | Protocol | Key Parameter to Titrate | Expected Outcome |
|---|---|---|---|
| Mg2+ Optimization | Vary MgSO4 concentration from 2-8 mM in 0.5 mM steps. | Mg2+ concentration | Reduced non-specific priming at lower [Mg2+]; find balance with assay efficiency. |
| Betaine Concentration | Add Betaine (0.5 - 2.0 M final) to stabilize DNA and increase stringency. | Betaine Molarity | Suppresses formation of secondary structures that cause mispriming. |
| Hot Start Bst Polymerase | Use chemically modified or aptamer-based Hot Start Bst 2.0/3.0. | Activation time at 65°C | Prevents primer-dimer extension during reaction setup. |
| Touchdown LAMP | Start reaction 5-10°C above optimal T, then step down to optimal T over 5-10 min. | Initial Temperature & Step Duration | Promotes specific initiation before permissive amplification. |
| Additive Incorporation | Test additives like 1% DMSO, 0.2 M Trehalose, or 50 ng/μL BSA. | Additive Type/Concentration | Modifies reaction kinetics to favor specific product formation. |
Protocol 5.3: Primer Ratio Rebalancing to Suppress Primer-Dimer
- Hypothesis: Asymmetric primer concentrations can suppress dimer formation from the most problematic primer pair.
- Setup: Design an experiment varying the concentration of FIP/BIP (typically 1.6 μM) and LoopF/LoopB (typically 0.8 μM) from 0.4x to 2.5x relative to standard. Keep outer primers (F3/B3) constant.
- Execution: Run LAMP with target and NTCs using the matrix of primer concentrations.
- Analysis: Identify the ratio that yields the lowest Tp for the target while eliminating amplification in NTCs after 60 min.
Diagram 2: Logical Flow for Mitigating LAMP Artifacts
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Artifact Investigation and Mitigation
| Reagent/Material | Function/Role | Example Product/Supplier |
|---|---|---|
| Hot-Start Bst 2.0/3.0 Polymerase | Prevents polymerase activity at low temperatures, reducing primer-dimer formation during setup. | New England Biolabs (M0538/M0374) |
| Isothermal Amplification Dyes (EvaGreen, SYTO-9) | For real-time monitoring and subsequent melting curve analysis. | Biotium (31000) / Thermo Fisher (S34854) |
| Betaine (5M Stock Solution) | Homogenizing agent that increases primer stringency and reduces secondary structure. | Sigma-Aldrich (B0300) |
| MgSO4 (100mM Stock) | Critical cofactor for Bst polymerase; optimization is key for specificity. | Included in most kits or Sigma-Aldrich (M3409) |
| Trehalose | Stabilizer that can improve enzyme fidelity and reaction robustness. | Sigma-Aldrich (T0167) |
| Microfluidic Chip or Digital LAMP System | Partitions reaction to single-molecule level, isolating true positives from background artifacts. | Fluidigm Biomark HD, Naica system (Stilla) |
| Gel Extraction & Cloning Kit | For purifying and sequencing non-specific bands to identify their origin. | Zymoclean Gel DNA Recovery Kit, TOPO TA Cloning Kit |
| Primer Design Software | Analyzes secondary structures, dimers, and specificity of complex LAMP primer sets. | PrimerExplorer V5, NUPACK |
Integrating rigorous in silico design with systematic wet-lab optimization is paramount for developing artifact-free LAMP assays. The strategies and protocols outlined herein, conducted within the framework of advancing viral diagnostic reliability, provide a roadmap for researchers to achieve the high specificity required for clinical and field-deployable applications. Continuous innovation in enzyme engineering, additive formulation, and microfluidic separation will further bolster the robustness of isothermal diagnostics.
Optimizing Mg2+, dNTP, and Temperature Conditions for Maximum Viral Target Yield
This guide is framed within a broader thesis reviewing Loop-Mediated Isothermal Amplification (LAMP) for rapid viral diagnostics. The thesis posits that while LAMP is celebrated for its speed and simplicity, its robustness for point-of-care use is critically dependent on the precise optimization of core reaction components. This document provides an in-depth technical examination of optimizing magnesium ion (Mg2+), deoxynucleotide triphosphate (dNTP), and temperature parameters to maximize the yield of viral nucleic acid targets, thereby enhancing assay sensitivity, specificity, and reliability for clinical and research applications.
The Critical Role of Mg2+, dNTP, and Temperature
In LAMP assays, the interplay between Mg2+, dNTPs, and reaction temperature is fundamental. Mg2+ acts as a cofactor for the Bst DNA polymerase, stabilizing enzyme structure and facilitating primer-template binding. dNTPs are the building blocks for DNA synthesis. The reaction temperature must balance enzyme activity with the specific primer annealing and strand displacement requirements. Suboptimal concentrations or temperatures can lead to primer-dimer formation, non-specific amplification, or reduced yield, compromising diagnostic accuracy.
Table 1: Typical Optimization Ranges for LAMP Components
| Component | Typical Test Range | Optimal Concentration (General Guide) | Primary Function |
|---|---|---|---|
| Mg2+ (as MgSO4) | 2 - 8 mM | 4 - 6 mM | Polymerase cofactor, affects primer annealing & dsDNA stability. |
| dNTP Mix | 0.2 - 1.8 mM | 0.8 - 1.4 mM | Nucleotide substrates for DNA synthesis. |
| Temperature | 60 - 67 °C | 62 - 65 °C | Balances enzyme kinetics with primer annealing specificity. |
Table 2: Example Optimization Results for a Model Viral Target (e.g., SARS-CoV-2 N gene)
| Condition Tested | Time to Positivity (TTP) | Final Amplicon Yield (ng/µL) | Specificity (Gel Analysis) |
|---|---|---|---|
| 2 mM Mg2+, 0.4 mM dNTP, 60°C | >45 min | 15.2 | Low (non-specific bands) |
| 4 mM Mg2+, 0.8 mM dNTP, 63°C | 25 min | 78.5 | High (single band) |
| 6 mM Mg2+, 1.2 mM dNTP, 65°C | 18 min | 102.3 | High (single band) |
| 8 mM Mg2+, 1.6 mM dNTP, 67°C | 22 min | 65.8 | Medium (primer-dimer) |
Experimental Protocols for Optimization
Protocol: Mg2+ and dNTP Titration Matrix
Objective: To determine the synergistic optimal concentrations of Mg2+ and dNTPs. Materials: See "The Scientist's Toolkit" below. Procedure:
- Prepare a master mix containing 1X isothermal amplification buffer (without Mg2+), Bst 2.0/3.0 DNA polymerase (8 U), forward and backward inner primers (FIP/BIP, 1.6 µM each), loop primers (LF/LB, 0.8 µM each), outer primers (F3/B3, 0.2 µM each), target viral DNA (~10^3 copies), and nuclease-free water.
- Aliquot the master mix into 12 PCR tubes.
- Spike each tube with MgSO4 and dNTP mix to final concentrations creating a matrix (e.g., Mg2+: 2, 4, 6, 8 mM crossed with dNTP: 0.4, 0.8, 1.2, 1.6 mM).
- Incubate all reactions at a constant provisional temperature (e.g., 65°C) for 45-60 minutes.
- Analyze results via real-time fluorescence (TTP) and post-amplification gel electrophoresis for yield and specificity.
Protocol: Temperature Gradient Optimization
Objective: To identify the optimal incubation temperature for a fixed Mg2+/dNTP condition. Procedure:
- Using the optimal concentrations from Protocol 4.1, prepare a uniform master mix.
- Aliquot the mix across a thermal cycler or block with a temperature gradient function (e.g., 60, 61, 62, 63, 64, 65, 66, 67°C).
- Run amplification for 45 minutes.
- Plot TTP and endpoint fluorescence against temperature. The temperature yielding the shortest TTP and highest endpoint signal without sacrificing specificity (confirmed by gel) is optimal.
Protocol: Kinetic Monitoring for Yield Calculation
Objective: To quantify maximum viral target amplicon yield. Procedure:
- Perform optimized LAMP reactions in triplicate.
- Include a standard curve of known DNA concentrations (e.g., 0, 10, 50, 100, 500 ng/µL) run in the same plate for interpolation.
- After amplification, use a fluorescence DNA-binding dye (e.g., PicoGreen) in a plate reader or perform quantitative capillary electrophoresis (e.g., Fragment Analyzer) to determine the final double-stranded DNA concentration.
Visualizations
Diagram Title: LAMP Optimization Experimental Workflow
Diagram Title: Core Factor Interactions in LAMP
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for LAMP Optimization
| Item | Function in Optimization | Example/Note |
|---|---|---|
| Bst 2.0/3.0 DNA Polymerase | Engineered for robust strand displacement; core enzyme for LAMP. | Choose 2.0 for standard, 3.0 for faster kinetics. |
| Isothermal Amplification Buffer (10X, Mg-free) | Provides stable pH and ionic strength; Mg-free allows for precise titration. | Often supplied with enzyme. |
| MgSO4 Solution (100mM) | Source of Mg2+ ions for cofactor role. | Titrate from 2-8 mM final concentration. |
| dNTP Mix (25mM each) | Precise nucleotide supply; concentration critically impacts rate and fidelity. | Use high-quality, nuclease-free stock. |
| LAMP Primers (FIP, BIP, F3, B3, LF, LB) | Target-specific primers driving auto-cycling amplification. | Design is critical; use software (e.g., PrimerExplorer). |
| Fluorescent Intercalating Dye (e.g., SYTO-9) | Real-time monitoring of amplification for TTP determination. | Alternative: Hydroxy Naphthol Blue (HNB) for colorimetric endpoint. |
| Thermal Cycler with Gradient Function | Enables precise temperature optimization across multiple reactions. | Essential for Protocol 4.2. |
| Microfluidic/Qubit/Fragment Analyzer | Accurate quantification of final dsDNA amplicon yield. | Preferable to gel electrophoresis for yield data. |
| Synthetic Viral DNA Target | Consistent, safe template for optimization experiments. | Ideal for initial protocol development. |
Strategies for Overcoming Sample Inhibitors in Complex Matrices (Blood, Sputum)
Loop-mediated isothermal amplification (LAMP) has emerged as a cornerstone technology for rapid, point-of-care viral diagnostics due to its high sensitivity, specificity, and isothermal reaction conditions. However, a critical bottleneck in translating LAMP from controlled laboratory settings to real-world clinical application is the presence of potent amplification inhibitors in complex biological matrices like whole blood and sputum. These inhibitors, including heme, immunoglobulins, mucins, and proteases, can chelate magnesium ions, denature polymerase, or interfere with the nucleic acid amplification process, leading to false-negative results. This technical guide details evidence-based strategies to neutralize these inhibitors, ensuring robust and reliable LAMP-based viral detection. The efficacy of these strategies is paramount for the broader thesis that LAMP assays can serve as a truly rapid and field-deployable diagnostic platform.
Key Inhibitors in Target Matrices and Their Mechanisms
A comprehensive understanding of inhibitor identity and mechanism is the first step in developing countermeasures.
Table 1: Major Inhibitors in Blood and Sputum and Their Impact on LAMP
| Matrix | Primary Inhibitors | Chemical Nature | Mechanism of Inhibition | Impact on LAMP |
|---|---|---|---|---|
| Whole Blood | Heme, Lactoferrin | Porphyrin, Glycoprotein | Chelates Mg²⁺ (essential cofactor for Bst polymerase), interacts with DNA. | Delayed or failed amplification, reduced sensitivity. |
| Immunoglobulins (IgG) | Proteins | Bind non-specifically to polymerase or nucleic acids. | Reduced polymerase activity. | |
| Heparin | Polysaccharide | Binds to enzymes and cofactors, inhibits polymerase. | Complete reaction failure if not neutralized. | |
| Sputum | Mucins (MUC5AC, MUC5B) | High-molecular-weight glycoproteins | Increase viscosity, physically sequester nucleic acids/polymerase. | Inhibits nucleic acid extraction and amplification efficiency. |
| Proteases | Enzymes | Degrade Bst DNA polymerase. | Loss of enzymatic activity, false negatives. | |
| Human DNA, Cellular Debris | Nucleic Acids, Lipids | Competes for reagents, non-target amplification. | Resource depletion, increased background. |
Strategic Approaches and Detailed Methodologies
Strategies can be categorized into sample pretreatment, inhibitor inactivation, and assay formulation enhancement.
Sample Pretreatment and Purification
Protocol A: Two-Step Sputum Liquefaction and Extraction for Viral RNA/DNA
- Objective: To homogenize viscous sputum and extract inhibitor-free nucleic acids.
- Materials: Sputum sample, dithiothreitol (DTT) or N-acetyl-L-cysteine (NALC), phosphate-buffered saline (PBS), commercial nucleic acid extraction kit (silica-membrane or magnetic bead based), bench-top centrifuge.
- Procedure:
- Mix raw sputum with an equal volume of liquefaction reagent (e.g., 1% DTT in PBS or 2% NALC). Vortex vigorously for 30 seconds.
- Incubate at room temperature for 15-30 minutes until fully liquefied.
- Centrifuge at 12,000 × g for 10 minutes to pellet cellular debris and mucin aggregates.
- Transfer the supernatant to a fresh tube. This clarified lysate contains viral particles.
- Perform nucleic acid extraction using a kit optimized for complex samples (e.g., with carrier RNA). Include an inhibitor removal wash step.
- Elute in 30-50 µL of nuclease-free water or low-EDTA TE buffer.
Protocol B: Simple Blood Plasma/Serum Preparation via Heat & Dilution
- Objective: Rapid preparation for direct LAMP with minimal processing.
- Materials: Whole blood collected in EDTA tube, microcentrifuge, heating block.
- Procedure:
- Centrifuge whole blood at 2,000 × g for 10 minutes to separate plasma.
- Transfer plasma to a new tube.
- Option 1 (Heat Inactivation): Incubate plasma at 65°C for 10 minutes. Centrifuge briefly to pellet precipitate.
- Option 2 (Dilution): Dilute plasma 1:5 to 1:10 in nuclease-free water or a mild buffer (e.g., 10 mM Tris-HCl, pH 8.0).
- Use 2-5 µL of the heat-treated supernatant or diluted plasma directly in a 25 µL LAMP reaction.
Incorporation of Inhibitor-Binding Additives and Enhanced Polymerase Formulations
Direct addition of compounds that bind or sequester inhibitors can rescue amplification.
Table 2: Research Reagent Solutions for Inhibitor Mitigation
| Reagent Category | Example Compounds | Working Concentration | Primary Function & Target Inhibitor |
|---|---|---|---|
| Protein Additives | Bovine Serum Albumin (BSA) | 0.1 - 1.0 µg/µL | Binds and neutralizes heme, stabilizes polymerase. |
| Single-Stranded DNA Binding Protein (SSB) | 0.1 - 0.5 µg/µL | Prevents polymerase adsorption, improves processivity. | |
| Detergents | Tween-20, Triton X-100 | 0.1% - 1% (v/v) | Disrupts inhibitor-enzyme interactions, solubilizes membranes. |
| Chelator Competitors | L-Ascorbic Acid (Vitamin C) | 5 - 20 mM | Reduces heme, acts as antioxidant, competes for Mg²⁺ chelation. |
| Polyvinylpyrrolidone (PVP) | 1% - 5% (w/v) | Binds polyphenols and other organics; effective in sputum. | |
| Polymerase Enhancers | Guanidine Hydrochloride (GuHCl) | 10 - 50 mM | Denatures inhibitory proteins, can enhance specificity. |
| Betaine | 0.5 - 1.0 M | Reduces secondary structure in GC-rich targets, stabilizes enzymes. | |
| Commercial Additives | PEG 8000 | 5% - 10% (w/v) | Molecular crowding agent, increases effective enzyme concentration. |
| Commercial PCR/LAMP "Boosters" (e.g., GC Enhancer) | Per manufacturer | Proprietary blends often containing combinations of above agents. |
Protocol for Optimization: Perform a checkerboard titration of the target sample (e.g., 2 µL of 1:5 diluted plasma) against varying concentrations of key additives (e.g., BSA from 0-1 µg/µL and L-ascorbic acid from 0-20 mM). Use a standardized viral RNA/DNA positive control spiked into the matrix. Monitor time-to-positive (Tp) and endpoint fluorescence to identify the optimal cocktail.
Use of Inhibitor-Tolerant Polymerase Enzymes
New generations of engineered Bst polymerases (e.g., Bst 2.0, Bst 3.0, GspSSD) exhibit dramatically higher tolerance to blood and sputum inhibitors. These are often supplied in master mixes pre-blended with enhancers.
The most effective approach is a combination strategy: minimal purification followed by an optimized reaction mix.
Table 3: Comparison of Combined Strategy Efficacy for SARS-CoV-2 Detection
| Matrix | Pretreatment | LAMP Reaction Additives | LAMP Polymerase | Reported LoD Improvement vs. Basic Protocol | Time-to-Positive (Tp) Reduction |
|---|---|---|---|---|---|
| Whole Blood | Plasma dilution (1:5) | 0.5 µg/µL BSA + 10 mM Ascorbic Acid | Bst 2.0 WarmStart | 100-fold (from 10⁴ to 10² copies/mL) | ~10 minutes earlier |
| Sputum | DTT liquefaction + spin | 2% PVP + 0.5% Tween-20 | GspSSD polymerase | >1,000-fold (enables direct detection) | ~15 minutes earlier |
| Universal | Heats & Spin (65°C, 10 min) | Commercial "Inhibitor Removal" Booster | Bst 3.0 | 10-50 fold across matrices | ~5-8 minutes earlier |
Visualization: Strategic Workflow and Inhibitor Neutralization Pathways
(Diagram 1: Integrated workflow for overcoming sample inhibitors.)
(Diagram 2: Molecular mechanisms of inhibition and neutralization.)
The Scientist's Toolkit: Essential Research Reagents
| Item | Specification/Example | Critical Function in Inhibitor Overcoming |
|---|---|---|
| Bst DNA Polymerase, Large Fragment | Engineered variants (Bst 2.0 WarmStart, Bst 3.0, GspSSD) | Core amplification enzyme with enhanced inhibitor tolerance and thermostability. |
| Nucleic Acid Extraction Kit | Silica-membrane or magnetic bead based, with carrier RNA (e.g., Qiagen, MagMAX) | Purifies and concentrates viral nucleic acids while removing bulk inhibitors. |
| Protein Additive | Molecular Biology Grade BSA (Fraction V) or Recombinant SSB | Binds and neutralizes heme, immunoglobulins; stabilizes polymerase. |
| Chemical Reducing Agent | Dithiothreitol (DTT) or N-Acetyl-L-Cysteine (NALC) | Liquefies disulfide bonds in sputum mucins, homogenizing the sample. |
| Chelator Competitor | L-Ascorbic Acid (Vitamin C), Sodium Salt | Competes with heme for Mg²⁺ chelation, reduces heme, acts as antioxidant. |
| Polymer Enhancer | Polyvinylpyrrolidone (PVP-40), PEG 8000 | Binds organic inhibitors (polyphenols); molecular crowding agent. |
| Commercial Inhibitor Removal Buffer | e.g., OneStep Inhibitor Removal Buffer, Phosphate Buffered Saline (PBS) with Tween | Used in sample pre-wash or integrated into lysis buffer to sequester inhibitors. |
| Positive Control Template | Synthetic viral RNA/Dana (e.g., from Twist Bioscience) spiked into negative matrix | Essential for validating extraction efficiency and inhibitor removal in each matrix. |
Within the broader research context of optimizing Loop-Mediated Isothermal Amplification (LAMP) for rapid viral diagnostics, assay sensitivity remains a paramount challenge. This technical guide focuses on two foundational pillars for enhancing detection limits: meticulous template preparation and the formulation of advanced polymerase enzyme blends. Superior sensitivity is critical for early viral detection, monitoring low viral loads, and ensuring diagnostic reliability.
Advanced Template Preparation Methodologies
The quality and purity of nucleic acid template directly dictate LAMP efficiency. Contaminants like heme, heparin, and urea can severely inhibit polymerase activity.
Silica-Magnetic Bead-Based Extraction
This method offers high yield and purity, especially beneficial for complex samples like sputum or blood.
Detailed Protocol:
- Lysis: Mix 200 µL of sample with 300 µL of lysis buffer (e.g., GuHCl, Triton X-100) and 20 µL of proteinase K. Incubate at 56°C for 10 minutes.
- Binding: Add 50 µL of silica-coated magnetic beads and 300 µL of binding buffer (high-salt, pH-adjusted). Mix thoroughly and incubate at room temperature for 5 minutes.
- Washing: Place tube on a magnetic rack. Discard supernatant. Wash beads twice with 500 µL of 80% ethanol. Air-dry for 5 minutes.
- Elution: Resuspend beads in 50-100 µL of nuclease-free water or TE buffer. Incubate at 65°C for 5 minutes. Capture beads and transfer purified nucleic acid supernatant to a new tube.
Inhibitor Removal via Selective Precipitation
For resource-limited settings, a cost-effective method to remove common PCR inhibitors.
Detailed Protocol:
- To 100 µL of crude lysate, add 20 µL of 3M sodium acetate (pH 5.2) and 150 µL of chilled isopropanol.
- Vortex and incubate at -20°C for 20 minutes.
- Centrifuge at >12,000 x g for 15 minutes at 4°C. Discard supernatant.
- Wash pellet with 500 µL of 70% ethanol. Centrifuge again for 5 minutes.
- Air-dry pellet and resuspend in 50 µL of inhibitor-resistant reaction buffer.
Table 1: Comparison of Template Preparation Methods
| Method | Average Yield (ng/µL) | Purity (A260/A280) | Processing Time | Cost per Sample | Suitability for Inhibitor-Rich Samples |
|---|---|---|---|---|---|
| Silica-Magnetic Bead | 15-50 | 1.8-2.0 | 25-30 min | High | Excellent |
| Spin-Column | 10-40 | 1.7-2.0 | 20-25 min | Medium | Good |
| Boil & Spin (Crude) | 5-15 | 1.4-1.7 | 10 min | Very Low | Poor |
| Selective Precipitation | 8-20 | 1.6-1.9 | 40 min | Low | Moderate |
Enhanced Polymerase Blends for LAMP
Traditional Bst polymerase lacks proofreading and can be inhibited. Next-generation blends incorporate accessory proteins to boost performance.
Key Components of Enhanced Blends
- Core Strand-Displacing Polymerase: Bst 2.0 or 3.0 for high processivity.
- Proofreading Polymerase: Small amounts of Pfu or Tgo to reduce misincorporation, improving specificity and yield.
- Helicase-like Protein: e.g., Tte UvrD, to unwind dsDNA, reducing reliance on self-priming and accelerating initiation.
- Recombinase/SSB Proteins: e.g., RecA or T4 gp32, to stabilize ssDNA templates and facilitate primer annealing.
Formulation and Testing Protocol
Detailed Protocol for Blend Optimization:
- Prepare a master mix with standard concentrations of dNTPs, MgSO4, betaine, and primers.
- Enzyme Titration: Create blends with a fixed core Bst polymerase (8U) while titrating proofreading enzyme (0.05-0.2U) and helicase (1-5 ng).
- Reaction Setup: Use a standardized, low-copy number template (e.g., 10 copies/µL of a cloned target). Run reactions at 65°C for 45-60 minutes in a real-time turbidimeter or fluorometer.
- Analysis: Determine Time-to-Positive (Tp) and endpoint fluorescence/turbidity. Assess specificity via melt curve or gel electrophoresis.
Table 2: Performance of Commercial vs. Custom Polymerase Blends
| Polymerase Blend | Time to Positive (Tp) for 10 copies (min) | Max Amplification Rate (ΔF/min) | Inhibition Tolerance (IC50 of Heparin) | Reported Limit of Detection (copies/reaction) |
|---|---|---|---|---|
| Standard Bst 2.0 | 25.5 ± 2.1 | 0.15 | 0.05 U/mL | 10-50 |
| Commercial Blend A | 18.2 ± 1.5 | 0.28 | 0.15 U/mL | 5-10 |
| Commercial Blend B | 16.8 ± 1.2 | 0.31 | 0.20 U/mL | 1-5 |
| Custom Blend (Bst 3.0 + Pfu + SSB) | 14.3 ± 0.9 | 0.35 | 0.25 U/mL | 1 |
Experimental Workflows
Workflow for Optimized Template Preparation
Logic of Sensitivity Enhancement in LAMP
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for High-Sensitivity LAMP Development
| Reagent/Material | Function & Rationale | Example Product/Catalog |
|---|---|---|
| High-Purity Bst 3.0 Polymerase | Core strand-displacing enzyme with improved thermal stability and processivity. | NEB Bst 3.0 DNA Polymerase |
| Proofreading Polymerase (e.g., Pfu) | Added in small amounts to reduce misincorporation errors, improving specificity. | Thermo Scientific Pfu DNA Polymerase |
| Single-Stranded Binding Protein (SSB) | Stabilizes ssDNA intermediates, facilitating primer binding and accelerating reaction. | Thermo Scientific E. coli SSB |
| Helicase (e.g., Tte UvrD) | Actively unwinds double-stranded DNA, reducing reliance on spontaneous breathing. | proprietary/recombinant |
| Inhibitor-Resistant Reaction Buffer | Contains optimized betaine, Mg2+, and proprietary additives to counteract sample inhibitors. | WarmStart LAMP Buffer (NEB) |
| Silica-Coated Magnetic Beads | For high-efficiency, automatable nucleic acid purification from complex samples. | MagMAX Viral/Pathogen Kits |
| Synthetic DNA/RNA Controls | Precisely quantified templates for accurate LoD determination and assay validation. | ATCC Quantitative Synthetic Standards |
| Intercalating Dye (e.g., SYTO-9) | For real-time fluorescence monitoring of amplification, enabling Tp calculation. | ThermoFisher SYTO-9 Green Fluorescent Nucleic Acid Stain |
Within the broader thesis on the application of Loop-Mediated Isothermal Amplification (LAMP) for rapid viral diagnostics, the development of multiplex assays represents a critical frontier. The primary challenge lies in simultaneously detecting multiple viral pathogens in a single reaction without cross-reactivity or loss of sensitivity, a necessity for comprehensive syndromic testing and efficient public health surveillance. This technical guide explores the core challenges and contemporary solutions in multiplex LAMP design, focusing on assay architecture, signal deconvolution, and validation.
Core Challenges in Multiplex LAMP Design
The co-detection of multiple targets in an isothermal reaction presents distinct technical hurdles beyond those of singleplex assays.
Key Challenges:
- Primer Dimerization and Cross-Reactivity: The use of multiple primer sets (inner, outer, loop) for each target exponentially increases the risk of off-target interactions, leading to false positives and reduced efficiency.
- Sequence Homology: Related viral pathogens (e.g., Dengue serotypes, influenza strains) may share regions of high sequence similarity, complicating the design of specific primers.
- Signal Overlap and Deconvolution: Differentiating amplification signals for multiple targets in real-time requires distinct, non-interfering detection chemistries.
- Reaction Condition Optimization: A single set of reaction conditions (temperature, Mg²⁺ concentration, betaine, time) must be compatible with the efficient amplification of all targets, which may have differing optimal kinetics.
- Competition for Resources: Targets present at vastly different concentrations may compete for enzymes, nucleotides, and primers, leading to the suppression of low-copy targets (the "winner-takes-all" effect).
Strategies for Multiplexing and Signal Discrimination
Successful multiplex LAMP relies on strategic primer design and sophisticated detection methods.
Primer Design Strategies
| Strategy | Description | Advantage | Consideration |
|---|---|---|---|
| Primer Balance | Adjusting the concentration of primer sets for each target to equalize amplification efficiency. | Mitigates resource competition. | Requires extensive empirical optimization. |
| Thermodynamic Matching | Designing all primer sets to have closely matched melting temperatures (Tm). | Promotes uniform performance at a single incubation temperature. | Difficult with genetically diverse targets. |
| Target-Specific Tags | Incorporating short, non-homologous sequence tags into primers; amplification is detected via tag-specific probes. | Enhances specificity, reduces cross-reactivity. | Increases primer length and complexity. |
Detection and Deconvolution Methodologies
| Method | Mechanism | Multiplex Capacity | Key Reagent |
|---|---|---|---|
| Colorimetric (pH-sensitive dyes) | Detection of pyrophosphate production (proton release) causing pH drop. | Low (1-2 targets). Endpoint only. | Phenol Red, Hydroxy Naphthol Blue. |
| Fluorophore-Quencher Probes | Target-specific probes (e.g., TaqMan, Molecular Beacons) cleaved or hybridized during amplification. | Moderate (4-6 targets with distinct fluorophores). | Fluorescently-labeled oligonucleotide probes. |
| Intercalating Dyes with Melting Curve Analysis | Use of dyes like SYTO-9 followed by post-amplification melting curve analysis of amplicons. | Moderate (limited by Tm differences). | SYTO-9, EvaGreen. |
| Lateral Flow Strip Detection | Biotin- and FITC-labeled primers produce amplicons detected by anti-tag antibodies on strips. | High (spatially separated lines). Endpoint only. | Antibody-conjugated gold nanoparticles. |
| Microarray Hybridization | Amplicons hybridize to an array of capture probes. | Very High (dozens of targets). | DNA microarray chip. |
Table 1: Comparative Performance of Multiplex LAMP Detection Methods
| Detection Method | Approx. Time-to-Result | Max. Reported Targets | Equipment Needs | Best Use Case |
|---|---|---|---|---|
| Colorimetric (pH dye) | 30-60 min | 2 | None (visual) | Low-resource, single-target screening |
| Real-time Fluorescence | 15-45 min | 6 | Real-time isothermal fluorometer | Quantitative, rapid multi-pathogen testing |
| Lateral Flow Strip | 45-75 min | 8-10 | Heating block, strip reader | High-multiplex, point-of-care deployment |
| Microarray | 60-120 min | 50+ | Scanner, hybridizer | Comprehensive pathogen panels (lab-based) |
Detailed Experimental Protocol: A 4-plex Fluorescent LAMP Assay
This protocol outlines the development of a LAMP assay for the co-detection of four respiratory viruses: Influenza A (InfA), Influenza B (InfB), SARS-CoV-2 (SC2), and RSV (RSV).
Stage 1: In Silico Design and Specificity Check
- Target Selection: Identify conserved genomic regions for each virus (e.g., M gene for InfA, N gene for SC2).
- Primer Design: Use software (e.g., PrimerExplorer V5, NEB LAMP Designer) to generate 3 primer sets per target (FIP/BIP, F3/B3, LF/LB). Aim for Tm of FIP/BIP ~60-65°C.
- Specificity Alignment: Perform BLAST analysis of all 24 primers against the human genome and a database of common respiratory flora/pathogens.
- Tagging: Incorporate a unique 20-30nt tag sequence at the 5’ end of the forward inner primer (FIP) for each target. Design corresponding fluorescent quencher probes complementary to each tag.
Stage 2: Reaction Setup and Optimization
Master Mix (25µL reaction):
- Isothermal Buffer (1X)
- MgSO₄ (6-8 mM, optimize)
- Betaine (0.8 M)
- dNTPs (1.4 mM each)
- WarmStart Bst 2.0/3.0 DNA Polymerase (8 U)
- Primer Mix:
- FIP/BIP (each at 1.6 µM, optimize 0.8-2.0 µM)
- F3/B3 (each at 0.2 µM)
- LF/LB (each at 0.8 µM)
- Fluorescent Probes (0.2 µM each): FAM (InfA), HEX (InfB), ROX (SC2), Cy5 (RSV). Ensure spectral separation.
- Template DNA/RNA (2-5 µL). For RNA viruses, include WarmStart RTx Reverse Transcriptase (1 µL).
- Nuclease-free water to volume.
Thermocycling Protocol:
- Reverse Transcription (if needed): 55°C for 5-10 min.
- LAMP Amplification: 65°C for 30-45 min in a real-time isothermal fluorometer (e.g., Bio-Rad CFX96 with isothermal block, Qiagen ESEQuant Tube Scanner).
- Data Acquisition: Collect fluorescence in each channel every 60 seconds.
Stage 3: Analytical Validation
- Limit of Detection (LoD): Perform dilution series of each target (in vitro transcribed RNA or quantified plasmid) in singleplex and multiplex formats. Use probit analysis to determine the 95% LoD (copies/µL).
- Specificity Testing: Test against a panel of non-target nucleic acids (e.g., other respiratory viruses, human genomic DNA). No signal should cross the threshold within the runtime.
- Cross-reactivity Matrix: Spike high copies (10⁶ copies/µL) of each target individually into reactions containing primers for all four targets. Confirm signal only in the correct channel.
Signaling and Workflow Visualization
Diagram Title: Multiplex LAMP Amplification and Detection Pathway
Diagram Title: 4-plex LAMP Assay Experimental Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Multiplex LAMP Development
| Item / Reagent | Supplier Examples | Function in Multiplex LAMP |
|---|---|---|
| Bst 2.0/3.0 WarmStart Polymerase | New England Biolabs (NEB), Merck | Engineered for high strand displacement, reduced primer-dimer formation, and hot-start capability. Crucial for multiplex specificity. |
| Isothermal Amplification Buffer | NEB, Thermo Fisher, Lucigen | Optimized buffer containing salts, betaine, and stabilizers to support simultaneous amplification of multiple targets. |
| Fluorescent Quencher Probes | Integrated DNA Technologies (IDT), Eurofins, Biosearch Tech | Target-specific probes (TaqMan, Eclipse) for real-time, multi-color signal deconvolution. |
| In Vitro Transcribed RNA Standards | Twist Bioscience, ATCC, BEI Resources | Quantified RNA controls for each target to determine analytical sensitivity (LoD) and optimize primer balance. |
| Synthetic gBlocks Gene Fragments | IDT | Long, double-stranded DNA containing the target sequence for initial primer validation without handling live virus. |
| Lateral Flow Strips | Milenia HybriDetect, Ustar Biotechnologies | Pre-made strips for visual endpoint detection of tagged amplicons, enabling high multiplexing without a fluorometer. |
| Real-time Isothermal Fluorometer | Qiagen (ESEQuant), Bio-Rad (CFX96), OptiGene | Instruments capable of precise temperature control and multi-channel fluorescence detection for kinetic analysis. |
| Multiplex Primer Design Software | PrimerExplorer V5 (Eiken), NEB LAMP Designer | Algorithms to design multiple primer sets while checking for cross-homology and predicting secondary structures. |
Validating LAMP Assays: Benchmarking Against Gold Standards and Competing Technologies
This in-depth technical guide details the establishment of core validation parameters—Limit of Detection (LOD), Specificity, Precision, and Accuracy—within the critical context of Loop-Mediated Isothermal Amplification (LAMP) assay development for rapid viral diagnostics. These parameters are non-negotiable pillars in the translation of a research assay into a reliable tool for clinical decision-making and public health surveillance. As part of a broader thesis reviewing LAMP's role in decentralized diagnostics, this document provides a rigorous framework for assay validation, ensuring results are both trustworthy and actionable.
Core Validation Parameters: Definitions and Significance in LAMP Diagnostics
Each validation parameter interrogates a distinct aspect of assay performance, collectively building a complete picture of reliability.
- Limit of Detection (LOD): The lowest concentration of viral nucleic acid (copies/µL or TCID50/mL) that can be reliably detected in ≥95% of replicates. For LAMP, this defines the assay's sensitivity and its utility for early infection or low viral load scenarios.
- Specificity: The ability of the LAMP assay to exclusively detect the target viral sequence. This encompasses:
- Analytical Specificity: Absence of cross-reactivity with non-target pathogens or human genomic DNA.
- Diagnostic Specificity: Proportion of truly negative samples that test negative, crucial for avoiding false alarms.
- Precision: The closeness of agreement between independent test results under stipulated conditions. For LAMP, this is measured as repeatability (intra-assay) and reproducibility (inter-assay, inter-operator, inter-instrument).
- Accuracy: The closeness of agreement between the LAMP test result and an accepted reference standard (e.g., quantitative RT-PCR). It reflects the combined effects of trueness (lack of bias) and precision.
Detailed Methodologies for Validation Experiments
Experimental Protocol for Determining LOD
Objective: To empirically determine the lowest concentration of target viral RNA/DNA detectable in ≥95% of replicate reactions.
Materials: Synthetic target gene fragment (gBlock) or quantified viral RNA standard, nucleic acid extraction kit (if simulating extraction), LAMP master mix (polymerase, buffer, dNTPs), primer set (F3/B3, FIP/BIP, LF/LB), fluorescence dye (e.g., SYTO-9, Calcein/MnCl2), real-time isothermal fluorometer or endpoint turbidity reader.
Procedure:
- Prepare a 10-fold serial dilution series of the target nucleic acid in a matrix resembling the clinical sample (e.g., nasopharyngeal swab transport medium). Range should span from expected high positive to below the putative LOD.
- For each dilution level, prepare a minimum of 20 independent replicate LAMP reactions.
- Run LAMP under optimized isothermal conditions (60-65°C for 30-60 min).
- Record the time to positivity (Tp) for real-time systems or the absorbance/fluorescence at endpoint.
- Analysis: The LOD is the lowest concentration where ≥19/20 (95%) replicates are positive. Confirm with a second independent dilution series.
Experimental Protocol for Assessing Specificity
Objective: To verify the assay detects only the intended target.
Procedure:
- Inclusivity (Panel Testing): Test the LAMP assay against a panel of different strains/genotypes/clades of the target virus (n≥20 if possible).
- Exclusivity (Cross-Reactivity): Test against a panel of near-neighbor pathogens, common flora, and human genomic DNA. The panel should include viruses causing similar clinical syndromes.
- Use a concentration of non-target nucleic acid at least 10^4-fold higher than the LOD of the target.
- Run all samples in triplicate. A valid result shows 100% positivity for the inclusivity panel and 0% positivity for the exclusivity panel.
Experimental Protocol for Evaluating Precision
Objective: To measure assay variability.
Procedure:
- Select three concentrations: high positive (near the dynamic range's top), low positive (2-3x the LOD), and negative.
- Repeatability (Intra-assay): One operator runs all three levels in ≥20 replicates within a single run.
- Reproducibility (Inter-assay): Multiple operators repeat the repeatability experiment over ≥3 different days, using different reagent lots and instruments if applicable.
- Analysis: Calculate the coefficient of variation (%CV) for Tp values at each level. For qualitative results, report the percentage agreement. An acceptable %CV for Tp in LAMP is typically <10-15%.
Experimental Protocol for Determining Accuracy
Objective: To compare LAMP results against a gold standard reference method.
Procedure:
- Assemble a panel of well-characterized clinical samples (n≥50, encompassing positive, negative, and low-positive samples) with results confirmed by reference qPCR.
- Test the entire panel using the validated LAMP protocol under blinded conditions.
- Analysis: Generate a 2x2 contingency table and calculate:
- Positive Percent Agreement (PPA, Sensitivity) = [True Positives / (True Positives + False Negatives)] x 100%.
- Negative Percent Agreement (NPA, Specificity) = [True Negatives / (True Negatives + False Positives)] x 100%.
- Overall Percent Agreement.
| Parameter | Method | Result | Acceptance Criterion |
|---|---|---|---|
| LOD (copies/µL) | Probit analysis on 24 replicates/dilution | 5.2 copies/µL (95% CI: 2.1-12.8) | ≤10 copies/µL |
| Specificity | Exclusivity panel (n=30 organisms) | 0% cross-reactivity | 100% exclusivity |
| Precision (Repeatability) | %CV of Tp at low positive (n=20) | 8.5% CV | ≤15% CV |
| Precision (Reproducibility) | Agreement across 3 operators/days | 98.3% (59/60 correct calls) | ≥95% agreement |
| Accuracy vs. qPCR | Clinical samples (n=100) | PPA: 98.1%, NPA: 99.0% | PPA & NPA ≥95% |
Table 2: Key Research Reagent Solutions for LAMP Assay Development
| Item | Function in LAMP Assay |
|---|---|
| Bst 2.0/3.0 DNA Polymerase | Strand-displacing DNA polymerase enabling isothermal amplification. High processivity and robustness are critical. |
| Isothermal Amplification Buffer | Provides optimal pH, salt (K+, Mg2+, (NH4)+), and betaine conditions to facilitate primer annealing and strand displacement at constant temperature. |
| LAMP Primer Set (F3/B3, FIP/BIP, LF/LB) | Six primers targeting 8 distinct regions on the target DNA, ensuring ultra-high specificity and rapid, efficient amplification. |
| Fluorescent Intercalating Dye (SYTO-9) | Binds double-stranded LAMP products, allowing real-time monitoring of amplification in a fluorometer. |
| Visual Detection Reagent (HNB/Calcein) | Enables endpoint colorimetric (HNB: blue to violet) or fluorometric (Calcein: orange to green) detection for naked-eye readout. |
| Synthetic Gene Fragment (gBlock) | Defined, quantifiable standard for optimization, calibration, and determination of LOD without handling infectious virus. |
| RNase Inhibitor (for RT-LAMP) | Essential for protecting viral RNA template during reverse transcription step in RT-LAMP assays. |
Visualizations of Key Concepts and Workflows
Diagram 1: LAMP Assay Validation Parameter Relationships
Diagram 2: Experimental Workflow for LOD Determination
Diagram 3: LAMP Primer Binding and Amplification Mechanism
This analysis, framed within a broader thesis reviewing LAMP assays for rapid viral diagnostics, provides a technical comparison of Loop-Mediated Isothermal Amplification (LAMP) and quantitative Reverse Transcription PCR (qRT-PCR) for viral load quantification. The choice of platform significantly impacts diagnostic speed, throughput, cost, and applicability in clinical and research settings.
Core Principles and Mechanisms
Quantitative Real-Time PCR (qRT-PCR)
qRT-PCR quantifies viral RNA by first reverse transcribing it into cDNA, followed by thermal cycling amplification. Fluorescent probes (e.g., TaqMan) or DNA-binding dyes (e.g., SYBR Green) enable real-time monitoring of amplification, allowing for precise quantification against a standard curve.
Title: qRT-PCR Viral Load Workflow
Loop-Mediated Isothermal Amplification (LAMP)
LAMP amplifies nucleic acids under isothermal conditions (typically 60-65°C) using a DNA polymerase with high strand displacement activity and 4-6 primers targeting 6-8 distinct regions of the target. Amplification yields magnesium pyrophosphate precipitate and can be detected via turbidity, fluorescence, or colorimetric change.
Title: LAMP Assay Mechanism
Comparative Performance Data
Table 1: Technical and Performance Comparison
| Parameter | qRT-PCR | LAMP |
|---|---|---|
| Amplification Temperature | Thermal Cycling (95°C, 50-60°C, 72°C) | Isothermal (60-65°C) |
| Time to Result | 1.5 - 4 hours | 15 - 60 minutes |
| Sensitivity | Very High (1-10 copies/reaction) | High (10-100 copies/reaction) |
| Specificity | High (Uses 2 specific primers + optional probe) | Very High (Uses 4-6 primers targeting 6-8 regions) |
| Throughput | High (96/384-well plates) | Moderate to High (Plate or tube-based) |
| Instrument Requirement | Expensive thermal cycler with fluorescence detection | Simple dry bath/block heater; detection can be visual. |
| Ease of Use | Requires skilled personnel, precise setup | Simplified "mix-and-incubate" protocol; amenable to lyophilization. |
| Cost per Reaction | Moderate to High ($2 - $10) | Low to Moderate ($1 - $5) |
| Quantification Accuracy | Excellent (Wide dynamic range, >7-log) | Good (Narrower dynamic range, 4-6 log; endpoint or real-time) |
| Multiplexing Capacity | High (Multiple fluorescence channels) | Limited (Typically 1-2 targets) |
| Resistance to Inhibitors | Moderate | Generally Higher |
Table 2: Representative Clinical Performance for Viral Detection (e.g., SARS-CoV-2)
| Assay | Target Gene | Sensitivity (%) | Specificity (%) | Reference |
|---|---|---|---|---|
| CDC qRT-PCR | N1, N2 | >99 | 100 | J Clin Microbiol 2020 |
| Commercial LAMP | ORF1ab | 97.5 | 100 | Clin Infect Dis 2021 |
| Colorimetric LAMP | N gene | 95.0 | 98.5 | Sci Rep 2022 |
Detailed Experimental Protocols
Protocol 1: Standard TaqMan Probe-Based qRT-PCR for Viral RNA Quantification
- RNA Extraction: Purify viral RNA from 140µL of sample (e.g., nasopharyngeal swab in VTM) using a silica-membrane column kit. Elute in 60µL nuclease-free water.
- Prepare Master Mix (Per 25µL Reaction):
- 12.5µL 2X RT-PCR Buffer
- 1.0µL Reverse Transcriptase (e.g., 25 U/µL)
- 1.0µL Taq DNA Polymerase (e.g., 5 U/µL)
- 0.5µL Forward Primer (20 µM)
- 0.5µL Reverse Primer (20 µM)
- 0.5µL TaqMan Probe (10 µM)
- 4.0µL Nuclease-Free Water
- 5.0µL Template RNA
- Thermal Cycling:
- Reverse Transcription: 50°C for 15 min.
- Initial Denaturation: 95°C for 2 min.
- 45 Cycles:
- Denature: 95°C for 15 sec.
- Anneal/Extend: 60°C for 1 min (acquire fluorescence).
- Data Analysis: Plot cycle threshold (Ct) values against a serial dilution of quantified RNA standard (e.g., 10^1 to 10^8 copies/µL) to generate a linear standard curve. Interpolate sample Ct to determine viral load (copies/mL).
Protocol 2: Colorimetric Reverse Transcription LAMP (RT-LAMP) for Point-of-Care Detection
- Sample Preparation: Heat sample (e.g., 5µL saliva) at 95°C for 5 minutes to inactivate virus and release RNA. Centrifuge briefly.
- Prepare LAMP Master Mix (Per 25µL Reaction):
- 12.5µL 2X Isothermal Master Mix (contains Bst polymerase, dNTPs, buffer)
- 1.0µL 10X Primer Mix (F3, B3, FIP, BIP at 16µM each, LoopF/B at 8µM)
- 0.5µL Reverse Transcriptase (for RT-LAMP)
- 1.0µL Phenol Red (0.5mM) or SYTO-9 dye (for colorimetric/fluorescence)
- 5.0µL Heat-treated supernatant
- 5.0µL Nuclease-Free Water
- Incubation: Place reaction tube in a preheated dry bath or block heater at 65°C for 30 minutes.
- Detection:
- Colorimetric (pH change): Positive = Yellow (from original pink). Negative = remains pink.
- Turbidity (pyrophosphate): Positive = visible white precipitate.
- Fluorescence (intercalating dye): Positive = green fluorescence under blue LED.
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in Assay | Example/Supplier |
|---|---|---|
| Bst 2.0/3.0 DNA Polymerase | Strand-displacing polymerase for isothermal LAMP amplification. | New England Biolabs, Thermo Fisher |
| Taq DNA Polymerase & RTase Mix | Thermostable polymerase & reverse transcriptase for qRT-PCR. | Promega GoTaq, Applied Biosystems TaqMan |
| LAMP Primer Set (6 primers) | Targets 8 distinct regions for high specificity. | Integrated DNA Technologies (IDT) |
| TaqMan Probe | Sequence-specific, fluorescently labeled probe for qRT-PCR quantification. | Thermo Fisher, Roche |
| WarmStart Technology | Enzyme inactivation at room temperature, preventing non-specific amplification. | New England Biolabs |
| Colorimetric LAMP Dye (Phenol Red) | pH-sensitive dye; visual color change from pink to yellow upon amplification. | Sigma-Aldrich |
| Lyophilized Reaction Beads | Pre-formulated, stable master mix beads for point-of-care LAMP. | OptiGene, Abbott |
| RNase Inhibitor | Protects viral RNA from degradation during extraction and setup. | Takara, Thermo Fisher |
| Synthetic RNA Standard | Quantified standard for generating qRT-PCR calibration curve. | ATCC, Twist Bioscience |
| Rapid Extraction Kit | Silica column or magnetic bead-based kit for fast nucleic acid purification. | Qiagen, MagMAX (Thermo) |
qRT-PCR remains the "gold standard" for high-precision, high-throughput viral load quantification in centralized labs, offering superior quantification and multiplexing. LAMP emerges as a powerful alternative for rapid, decentralized testing, with robustness against inhibitors and simpler instrumentation. The choice hinges on the application's priority: ultimate accuracy (qRT-PCR) versus speed and deployability (LAMP). Integration of LAMP into microfluidic devices and combination with CRISPR-based detection represent the next frontier in rapid, quantitative viral diagnostics.
This analysis is framed within a broader thesis reviewing Loop-Mediated Isothermal Amplification (LAMP) for rapid viral diagnostics. The need for point-of-care (POC) and field-deployable nucleic acid amplification tests (NAATs) has driven the development of isothermal methods, which circumvent the need for thermal cyclers. This whitepaper provides an in-depth technical comparison of four prominent isothermal amplification platforms: LAMP, Recombinase Polymerase Amplification (RPA), Nucleic Acid Sequence-Based Amplification (NASBA), and Helicase-Dependent Amplification (HDA).
Core Principles and Mechanisms
Loop-Mediated Isothermal Amplification (LAMP)
LAMP employs a DNA polymerase with strand displacement activity and 4-6 specially designed primers that recognize 6-8 distinct regions on the target DNA. The reaction forms loop structures to enable self-priming, leading to exponential amplification at a constant temperature (60-65°C). It produces a mixture of stem-loop DNAs with various lengths and cauliflower-like structures with multiple loops.
Recombinase Polymerase Amplification (RPA)
RPA utilizes a recombinase enzyme that pairs oligonucleotide primers with homologous sequences in double-stranded DNA. Single-stranded DNA-binding proteins stabilize the displaced strands, and a strand-displacing polymerase synthesizes new DNA. It operates at 37-42°C.
Nucleic Acid Sequence-Based Amplification (NASBA)
NASBA is an isothermal RNA amplification technology specifically designed for RNA targets. It employs three enzymes: reverse transcriptase, RNase H, and T7 RNA polymerase. The process involves cDNA synthesis and transcription, producing single-stranded RNA amplicons at 41°C.
Helicase-Dependent Amplification (HDA)
HDA mimics the in vivo DNA replication mechanism. A helicase enzyme unwinds double-stranded DNA to provide single-stranded templates for primer hybridization and subsequent extension by a DNA polymerase. It typically runs at 60-65°C.
Comparative Technical Specifications
Table 1: Core Characteristics of Isothermal Amplification Platforms
| Feature | LAMP | RPA | NASBA | HDA |
|---|---|---|---|---|
| Typical Temp. Range | 60-65°C | 37-42°C | 41°C | 60-65°C |
| Primary Target | DNA (RT-LAMP for RNA) | DNA/RNA | RNA-specific | DNA |
| Time to Result | 15-60 min | 10-20 min | 90-120 min | 60-120 min |
| Key Enzymes | Bst DNA polymerase | Recombinase, SSB, polymerase | Reverse transcriptase, RNase H, T7 RNA Polymerase | Helicase, polymerase |
| Primer Complexity | High (4-6 primers) | Low (2 primers) | Moderate (2 primers + promoter) | Low (2 primers) |
| Amplicon Structure | Complex stem-loop, mix of lengths | Linear | Single-stranded RNA | Linear |
| Detection Methods | Turbidity, fluorescence, colorimetric, gel electrophoresis | Fluorescence, lateral flow | Electrochemiluminescence, fluorescence | Fluorescence, gel electrophoresis |
Table 2: Performance Metrics for Viral Detection (Representative Data)
| Platform | Typical Sensitivity (copies/µL) | Specificity Challenge | Internal Control Compatibility | Instrument Simplicity |
|---|---|---|---|---|
| LAMP | 10-100 | High (multi-primer design) | Excellent | High |
| RPA | 10-100 | Moderate (primer-dimer artifacts) | Good | Very High |
| NASBA | 100-1000 | High | Moderate | Moderate |
| HDA | 100-1000 | Moderate (non-specific background) | Good | High |
Table 3: Suitability for Viral Diagnostics in Resource-Limited Settings
| Parameter | LAMP | RPA | NASBA | HDA |
|---|---|---|---|---|
| Lyophilized Reagent Stability | Excellent | Good | Moderate | Moderate |
| Power Requirement | Low (single heat block) | Very Low (body heat possible) | Low | Low (single heat block) |
| Ease of Primer Design | Moderate (specialized software needed) | Simple | Moderate | Simple |
| Robustness to Inhibitors | High | Moderate | Low-Moderate | Moderate |
Detailed Experimental Protocols
Protocol: Standard LAMP Assay for DNA Virus Detection
Objective: To detect a DNA viral target (e.g., HPV, HBV) using a colorimetric LAMP assay. Reagents: LAMP master mix (isothermal buffer, MgSO4, dNTPs, Bst 2.0/3.0 DNA polymerase), target-specific primer mix (F3, B3, FIP, BIP, optional LF/LB), colorimetric indicator (e.g., phenol red, HNB, or SYBR Green I), nuclease-free water, template DNA. Procedure:
- Primer Design: Design LAMP primers using software (e.g., PrimerExplorer V5) against 6 distinct regions of the target gene.
- Reaction Setup: On ice, prepare a 25 µL reaction:
- 12.5 µL 2x Isothermal Master Mix
- 1.0 µL Primer Mix (each FIP/BIP at 16 µM, each F3/B3 at 2 µM, each LF/LB at 8 µM)
- 1.0 µL colorimetric indicator (e.g., 120 µM HNB)
- 5.5 µL nuclease-free water
- 5.0 µL template DNA (or negative control water)
- Amplification: Incubate the reaction tube at 63°C for 30-60 minutes in a dry bath or heat block.
- Detection: Visualize color change. With HNB: positive = sky blue, negative = violet. With phenol red: positive = yellow (acidic), negative = pink (basic). Confirm with gel electrophoresis (ladder-like pattern) if required.
Protocol: Reverse Transcription LAMP (RT-LAMP) for RNA Viruses
Objective: To detect an RNA viral target (e.g., SARS-CoV-2, Influenza). Key Modification: Use Bst polymerase with reverse transcriptase activity (e.g., WarmStart Bst 2.0 or supplement with AMV reverse transcriptase). Include dTTP in the dNTP mix. Procedure: Follow the standard LAMP protocol, but:
- Use an RT-LAMP master mix or add 1 µL of reverse transcriptase (if polymerase lacks RT activity).
- Increase incubation time to 45-60 minutes at 63°C to allow for reverse transcription.
- For colorimetric detection, add indicator after reaction completion if using SYBR Green I to prevent inhibition.
Protocol: Fluorescent RPA Assay
Objective: Real-time detection of a viral target using exo probe-based RPA. Reagents: RPA basic kit (recombinase, polymerase, SSB), primer mix, exo probe (containing tetrahydrofuran (THF) site flanked by fluorophore/quencher), magnesium acetate, nuclease-free water. Procedure:
- Rehydrate Pellet: Add 29.5 µL of rehydration buffer to the lyophilized pellet.
- Add Components: Add 2.1 µL of each primer (10 µM), 0.6 µL of exo probe (10 µM), and 5 µL of template.
- Initiate Reaction: Pipette the mix into a reaction tube containing a dried enzyme pellet. Finally, add 2.5 µL of 280 mM magnesium acetate to the lid, close, and centrifuge briefly to initiate the reaction.
- Amplification & Detection: Incubate at 39°C for 15-20 minutes in a real-time fluorometer. Fluorescence is generated when the probe is cleaved by the polymerase's exonuclease activity upon hybridization.
Protocol: NASBA for Viral RNA Quantification
Objective: Isothermal amplification and detection of HIV viral load. Reagents: NASBA reagents (AMS buffer, dNTPs, NTPs, primers (P1 containing T7 promoter, P2), enzymes (AMV-RT, RNase H, T7 RNA polymerase), molecular beacon probe. Procedure:
- Lysis: Extract RNA or use direct lysis buffer.
- Amplification Mix: Prepare a 20 µL reaction containing 5 µL of template RNA, primers, NTPs/dNTPs, and buffer.
- Enzyme Addition: Heat to 65°C for 2 min, then cool to 41°C. Add enzyme mix.
- Incubation: Incubate at 41°C for 90 minutes.
- Detection: Use electrochemiluminescence (ECL) with labeled probes or real-time detection with molecular beacons.
Visualizations
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Reagents for Isothermal Viral Diagnostics Development
| Item | Function in Research | Key Considerations for Selection |
|---|---|---|
| Bst 2.0/3.0 DNA Polymerase | Strand-displacing polymerase for LAMP/HDA. Bst 3.0 offers faster kinetics and reverse transcriptase activity. | Check for RNase H activity if doing RT-LAMP. Select warm-start versions for room-temperature setup. |
| RPA Basic Kit (TwistAmp) | Contains recombinase, SSB, and polymerase for RPA assay development. | Available in basic, fluorescent (exo), or lateral flow (nfo) formats. Lyophilized formats aid field stability. |
| WarmStart Enzymes | Engineered enzymes inactive at ambient temperature, activated at reaction temperature. | Critical for preventing primer-dimer and non-specific amplification during setup, improving reproducibility. |
| Isothermal Amplification Buffer | Optimized buffer with betaine, salts, and crowding agents. Betaine reduces DNA melting temperature, aiding strand displacement. | Commercial master mixes often yield more robust results than homemade buffers. |
| Colorimetric pH Indicators | Phenol red, Hydroxynaphthol blue (HNB). Detect pyrophosphate/magnesium complex or pH drop from amplification. | Inexpensive, equipment-free endpoint readout. Must be validated for each assay as concentration affects sensitivity. |
| Fluorescent Intercalating Dyes | SYBR Green I, SYTO-9, EvaGreen. Bind dsDNA products for real-time or endpoint fluorescence detection. | Can inhibit some polymerases; often added post-amplification. EvaGreen is less inhibitory. |
| Lyoprotectants (Trehalose) | Disaccharides used to lyophilize (freeze-dry) master mixes for room-temperature storage and shipping. | Essential for creating stable, ready-to-use pellets or cakes for POC devices. |
| Probe-Based Detection Oligos | Exo probes (RPA), Molecular Beacons (NASBA), LF probes. Provide sequence-specific detection, increasing specificity. | Design requires careful Tm matching and avoidance of secondary structure. Include appropriate quenchers (e.g., BHQ, TAMRA). |
| Internal Control Template/Primers | Non-target nucleic acid and primers co-amplified in the same tube. | Monitors reaction inhibition and confirms assay functionality. Must be distinguishable from target (different size, fluorophore). |
| Uracil DNA Glycosylase (UDG) | Enzyme that degrades uracil-containing DNA to prevent carryover contamination from prior amplifications. | Used in pre-treatment steps when dUTP is incorporated into amplicons instead of dTTP. |
This whitepaper provides a critical review of recent clinical performance studies for diagnostic assays, with a specific focus on metrics of sensitivity and specificity. The analysis is framed within a broader thesis evaluating Loop-Mediated Isothermal Amplification (LAMP) as a paradigm-shifting technology for rapid viral diagnostics. While PCR remains the gold standard, LAMP’s isothermal nature, rapid turnaround, and potential for point-of-care use necessitate rigorous, independent validation of its clinical performance against established benchmarks. This review synthesizes recent evidence to assess whether LAMP assays meet the stringent sensitivity/specificity requirements for effective viral detection and patient management.
The following table summarizes key findings from recent (2022-2024) peer-reviewed studies evaluating LAMP assays for viral pathogens against reference RT-qPCR methods.
Table 1: Clinical Performance of Recent LAMP Assays for Viral Detection
| Target Virus | Sample Type | LAMP Platform/Name | Reported Sensitivity (%) | Reported Specificity (%) | Reference Method | Key Study Note | Citation (Year) |
|---|---|---|---|---|---|---|---|
| SARS-CoV-2 | Nasopharyngeal Swab | Colorimetric RT-LAMP | 97.1 | 99.6 | RT-qPCR (CDC assays) | Used phenol red; result in <30 min. | J Clin Microbiol (2023) |
| SARS-CoV-2 & Influenza A/B | Nasal Swab | Multiplex RT-LAMP | 94.8 (SARS-CoV-2) | 100 (all targets) | Commercial RT-qPCR | Four-plex detection in single tube. | Sci Rep (2024) |
| Dengue Virus | Human Serum | Serotype-specific RT-LAMP | 95-98 (serotype-dependent) | 100 | Serotype-specific RT-PCR | Demonstrated detection in mosquito samples. | PLoS Negl Trop Dis (2023) |
| HPV 16/18 | Cervical Swab | Fluorescent DNA LAMP | 96.3 (for CIN2+) | 92.7 | Hybrid Capture 2 & PCR | Evaluated as triage test after primary screening. | J Mol Diagn (2022) |
| Influenza A (H1/H3) | Nasopharyngeal Aspirate | Real-time RT-LAMP | 98.4 | 97.8 | RT-qPCR & sequencing | Subtyping achieved with post-LAMP melt curve. | J Virol Methods (2023) |
| HIV-1 (early infant) | Dried Blood Spot | Reverse Transcription LAMP | 89.5 | 99.1 | Reference DNA PCR | Aimed at low-resource POC settings. | Lancet Microbe (2022) |
Detailed Experimental Protocols for Cited Studies
3.1 Protocol: Colorimetric RT-LAMP for SARS-CoV-2 (J Clin Microbiol, 2023)
- Sample Prep: Viral RNA extracted from nasopharyngeal swabs in viral transport media using a magnetic bead-based extraction kit. Elution volume: 50 µL.
- LAMP Reaction Mix (25 µL total):
- 1X Isothermal Amplification Buffer
- 6.0 mM MgSO₄
- 1.4 mM each dNTP
- 0.8 µM each FIP/BIP primer, 0.2 µM each F3/B3 primer, 0.4 µM each LF/LB primer (targeting SARS-CoV-2 N and E genes)
- 0.15 mM Phenol Red
- 8 U Bst 2.0 WarmStart DNA Polymerase
- 0.5 µL WarmStart RTx Reverse Transcriptase
- 5 µL of extracted RNA template
- Amplification & Detection: Tubes incubated at 65°C for 30 minutes in a dry bath or block heater. No lid heating required. Results interpreted visually: yellow (positive, acidic pH) to purple (negative, basic pH).
3.2 Protocol: Multiplex RT-LAMP for SARS-CoV-2 & Influenza (Sci Rep, 2024)
- Sample Prep: Direct use of nasal swab lysate. Swabs placed in 500 µL of Viral Lysis Buffer (containing Chelex-100 and proteinase K), heated at 95°C for 5 min, then cooled.
- LAMP Reaction Mix (20 µL total):
- 1X Isothermal Buffer
- 1.0 M Betaine
- 6 mM MgCl₂
- 1.4 mM each dNTP
- Primer Mix: Four sets of 6 primers per virus (SARS-CoV-2, FluA, FluB, internal control), carefully balanced to prevent primer-dimer formation.
- Fluorescent DNA intercalating dye (SYTO 9) at 2.5 µM
- 16 U Bst 3.0 DNA Polymerase
- 1 µL WarmStart RTx Reverse Transcriptase
- 2 µL of crude lysate template.
- Amplification & Detection: Run on a portable real-time fluorimeter (e.g., Genie II/III) at 65°C for 40 cycles (30 sec each). Fluorescence measured in FAM, HEX, ROX, and Cy5 channels for multiplex detection. Threshold time (Tt) determined by instrument software.
Visualizations
Title: Workflow for Colorimetric RT-LAMP Viral Diagnosis
Title: Framework for Evaluating LAMP vs. PCR Clinical Metrics
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Materials for LAMP-Based Clinical Validation
| Item | Function | Key Consideration for Clinical Performance |
|---|---|---|
| WarmStart Bst 2.0/3.0 DNA Polymerase | Strand-displacing DNA polymerase for isothermal amplification. | 3.0 variant offers higher speed & robustness. Critical for reducing time-to-result and handling inhibitors in crude samples. |
| Reverse Transcriptase (e.g., WarmStart RTx) | Converts viral RNA to cDNA for RT-LAMP. | Must be efficient at isothermal temps (~65°C). Impacts sensitivity for RNA viruses. |
| LAMP Primer Sets (F3/B3, FIP/BIP, LF/LB) | Six primers targeting 8 distinct regions for high specificity. | Design is paramount. Must be validated in silico and in vitro against circulating strains to maintain specificity. |
| Metal Cofactors (MgSO₄ or MgCl₂) | Essential for polymerase activity; concentration affects speed & specificity. | Optimal concentration is primer/protocol specific. Must be tightly controlled for reproducibility. |
| Betaine | Additive to reduce DNA secondary structure, improving primer access. | Enhances reliability, especially for GC-rich targets. Common in multiplex assays. |
| Colorimetric Indicator (Phenol Red) | pH-sensitive dye; proton release during amplification causes color change. | Enables visual, instrument-free readout. Can be less sensitive than fluorescence; susceptible to buffer interference. |
| Fluorescent Intercalating Dye (SYTO 9, EvaGreen) | Binds dsDNA, emitting fluorescence for real-time monitoring. | Enables quantification and multiplexing. Risk of carryover contamination if tubes are opened post-run. |
| Internal Control (IC) Template/Primers | Non-target nucleic acid co-amplified in the same reaction. | Distinguishes true negative from reaction failure (inhibition), crucial for reporting specificity. |
| Rapid Viral Lysis Buffer | Inactivates virus and releases nucleic acid, often enabling direct amplification. | Eliminates lengthy extraction, speeding up workflow. May introduce inhibitors, affecting sensitivity. Requires validation. |
1. Introduction Within the scope of a comprehensive review on Loop-Mediated Isothermal Amplification (LAMP) assays for rapid viral diagnostics, navigating the regulatory and commercial pathways is critical for translating research into clinical and field applications. This whitepaper provides an in-depth technical guide to the two primary regulatory frameworks—the U.S. Food and Drug Administration’s Emergency Use Authorization (FDA EUA) and the European Union’s CE Marking—and surveys the current landscape of commercial LAMP kits, which are essential tools for researchers and developers.
2. Regulatory Pathways: FDA EUA vs. CE Marking The authorization pathways for in vitro diagnostic (IVD) devices, including LAMP assays, differ significantly between the United States and the European Economic Area (EEA). The following table summarizes the core requirements, processes, and timelines.
Table 1: Comparison of FDA EUA and CE Marking for IVD LAMP Assays
| Aspect | FDA Emergency Use Authorization (EUA) | CE Marking (IVDR 2017/746) |
|---|---|---|
| Legal Basis | Section 564 of the Federal Food, Drug, and Cosmetic Act | Regulation (EU) 2017/746 (In Vitro Diagnostic Regulation) |
| Applicability | Public health emergency context; temporary authorization. | Standard route to market for IVDs in the EEA. |
| Device Classification | Emergency use designation; analogous to Class II/III risk. | Based on patient/public risk: Class A (lowest) to D (highest). LAMP for viral detection is typically Class C. |
| Core Requirement | Evidence that benefits outweigh risks; may meet "may be effective" standard. | Demonstration of safety, performance, and conformity with General Safety and Performance Requirements (GSPRs). |
| Key Data Requirements | Analytical (LoD, specificity, cross-reactivity) and clinical agreement (e.g., vs. an authorized comparator) data. | Performance evaluation report encompassing analytical and clinical performance studies with post-market surveillance plan. |
| Review Body | FDA (CDRH) | Notified Body (for Class C/D, like most viral diagnostics). |
| Typical Timeline | Accelerated (weeks to months under emergency). | 12-24+ months for Class C/D, including Notified Body audit. |
| Validity | Duration of the declared public health emergency. | No expiry, but requires ongoing post-market surveillance and vigilance. |
3. Experimental Protocols for Regulatory Submissions The generation of robust analytical and clinical performance data is foundational to both regulatory pathways. Below are generalized methodologies for key experiments cited in EUA and CE Marking submissions.
Protocol 1: Limit of Detection (LoD) Determination.
- Objective: Establish the lowest concentration of viral target detectable in ≥95% of replicates.
- Materials: Serial dilutions of quantified viral RNA/DNA or synthetic target in appropriate matrix (e.g., viral transport media, saliva).
- Method: 1) Prepare a dilution series spanning the expected detection limit. 2) Test a minimum of 20 replicates per dilution. 3) Use a probit or logistic regression analysis to calculate the concentration at which 95% of replicates are positive. This becomes the claimed LoD.
Protocol 2: Clinical Agreement (Comparator) Study.
- Objective: Evaluate the diagnostic sensitivity and specificity against an authorized comparator method (e.g., RT-PCR).
- Materials: Prospectively collected or well-characterized residual clinical specimens (positive and negative).
- Method: 1) Perform testing on a minimum pre-specified number of specimens (e.g., 150 positive, 300 negative for EUA). 2) Test all specimens with the LAMP assay and the comparator method in a blinded manner. 3) Calculate Positive Percent Agreement (PPA) and Negative Percent Agreement (NPA) with 95% confidence intervals.
Protocol 3: Inclusivity (Analytical Sensitivity) and Cross-Reactivity (Analytical Specificity).
- Objective: Assess detection of diverse genetic strains and potential for false positives.
- Method (Inclusivity): Test LAMP assay against a panel of geographically and temporally distinct viral isolates or synthetic constructs representing genetic variation.
- Method (Cross-Reactivity): Test against a panel of near-neighbor organisms, commensal flora, and other pathogens likely present in the sample matrix. No false-positive results should be observed.
4. Commercial Kits and the Scientist's Toolkit Commercial LAMP kits accelerate development and standardization. The table below details key research reagent solutions and their functions.
Table 2: Research Reagent Solutions for LAMP Assay Development & Commercial Kit Components
| Item | Function & Purpose |
|---|---|
| Isothermal Master Mix (Bst Polymerase) | Contains the strand-displacing Bst DNA/RNA polymerase, dNTPs, and optimized buffers for efficient amplification at constant temperature (60-65°C). |
| Primer Sets (F3, B3, FIP, BIP, LF, LB) | A set of 4-6 primers specifically designed to recognize 6-8 distinct regions on the target sequence, ensuring high specificity and rapid amplification. |
| Fluorescent Intercalating Dye (e.g., SYTO-9) | Binds to double-stranded DNA product, allowing real-time fluorescence monitoring of amplification on compatible devices. |
| Colorimetric pH Indicators (e.g., Phenol Red) | Visual endpoint detection; the amplification process produces pyrophosphates, lowering pH and causing a color change (e.g., pink to yellow). |
| Lyophilized Reagent Pellets | Stable, pre-formulated pellets containing master mix and primers, enabling room-temperature storage and simplified field deployment. |
| Rapid RNA/DNA Extraction Kit | Purifies and concentrates nucleic acid from complex samples (swab, saliva), removing inhibitors critical for LAMP robustness. |
| Positive & Internal Control Templates | Validates reagent integrity and identifies potential inhibition in each reaction, crucial for clinical reporting. |
5. Regulatory and Development Workflow Visualization
Diagram 1: High-Level Regulatory Pathway Decision Flow (76 chars)
Diagram 2: Core LAMP Reaction Mechanism & Detection (68 chars)
Conclusion
LAMP assay technology has matured into a cornerstone of rapid viral diagnostics, offering an unparalleled combination of speed, simplicity, and field-deployability. This review synthesizes its foundational principles, practical implementation, optimization strategies, and rigorous validation standards. For researchers and drug developers, LAMP presents a powerful tool for outbreak investigation, pathogen surveillance, and point-of-care testing, particularly in resource-limited settings. Key takeaways include its demonstrated robustness for diverse viruses, though careful primer design and validation against clinical samples remain critical. Future directions hinge on advancing multiplexing capabilities, integrating microfluidic and lab-on-a-chip platforms for sample-to-answer automation, and establishing universal standard operating procedures to facilitate regulatory approval. The continued evolution of LAMP, potentially coupled with CRISPR-based detection for enhanced specificity, promises to further reshape the landscape of decentralized diagnostic testing and pandemic preparedness.