LAMP Assay for SARS-CoV-2 Detection: Principles, Protocols, and Performance in 2024
This technical article provides researchers and diagnostics developers with a comprehensive analysis of Loop-Mediated Isothermal Amplification (LAMP) for SARS-CoV-2 detection.
LAMP Assay for SARS-CoV-2 Detection: Principles, Protocols, and Performance in 2024
Abstract
This technical article provides researchers and diagnostics developers with a comprehensive analysis of Loop-Mediated Isothermal Amplification (LAMP) for SARS-CoV-2 detection. We explore the foundational molecular principles of LAMP, including primer design strategies targeting conserved viral regions like the ORF1ab and N genes. A detailed methodological guide covers RNA extraction, reaction setup, and detection methods (colorimetric, fluorescent, lateral flow). The article addresses common troubleshooting scenarios, optimization strategies for sensitivity and specificity, and a critical comparative validation against gold-standard RT-PCR and emerging CRISPR-based assays. We conclude with an evaluation of LAMP's role in point-of-care diagnostics and future pandemic preparedness.
Decoding LAMP: The Molecular Principles Behind Rapid SARS-CoV-2 Isothermal Amplification
This whitepaper elucidates the core molecular principles of Strand Displacement DNA Synthesis and Loop Formation, the foundational engine of the Loop-Mediated Isothermal Amplification (LAMP) assay. Framed within ongoing research for SARS-CoV-2 detection, we detail the mechanistic biochemistry, present quantitative performance data, and provide standardized protocols to enable robust assay development for researchers and pharmaceutical professionals.
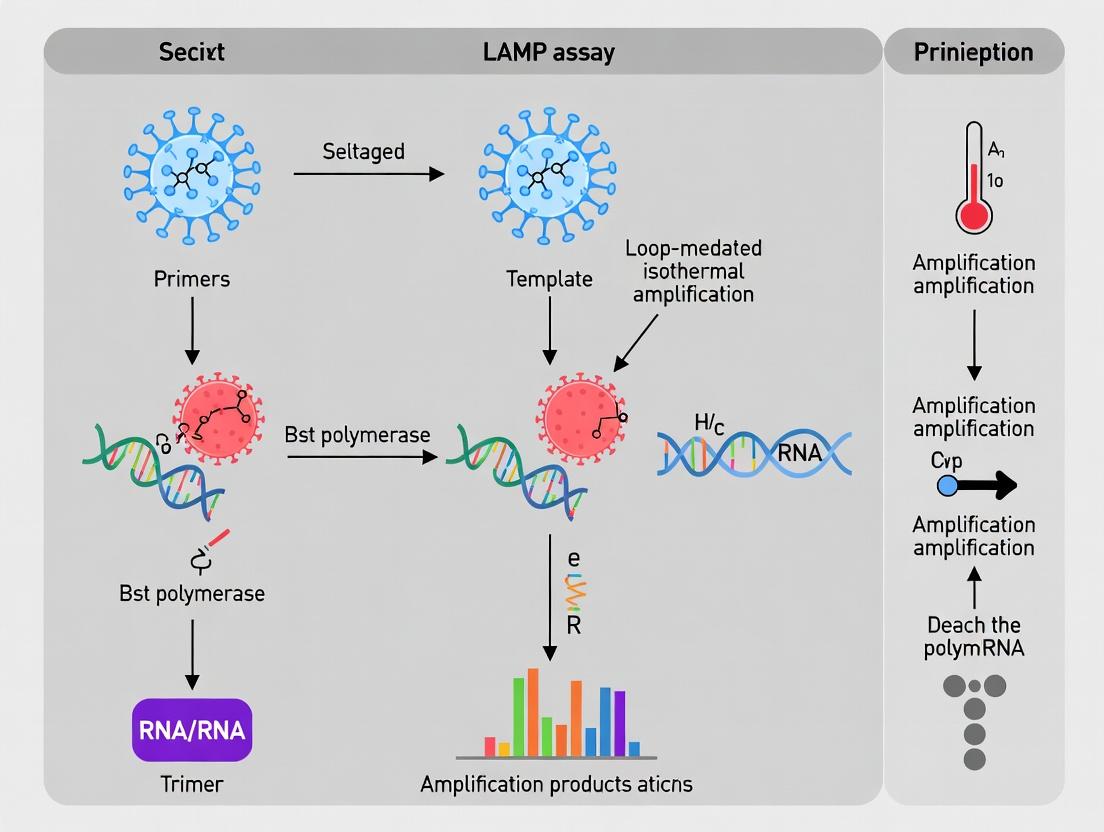
Loop-Mediated Isothermal Amplification (LAMP) is an isothermal nucleic acid amplification technique renowned for its high sensitivity, specificity, and rapid kinetics, making it ideal for point-of-care SARS-CoV-2 diagnostics. The core of LAMP’s efficiency lies in two intertwined processes: strand displacement DNA synthesis and self-priming loop formation. These processes, driven by a Bst-type DNA polymerase, enable autocycling amplification without the need for thermal denaturation. This guide deconstructs these principles in the context of optimizing LAMP assays for SARS-CoV-2 RNA targets, addressing key challenges in primer design, reaction dynamics, and signal detection.
Mechanistic Biochemistry: A Step-by-Step Deconstruction
Primer Design Framework
A standard LAMP assay requires four to six primers recognizing six to eight distinct regions on the target DNA.
- FIP (Forward Inner Primer): Contains the F2 sequence (complementary to F2c) at the 3’ end and the same sense as F1c at the 5’ end.
- BIP (Backward Inner Primer): Contains the B2c sequence (complementary to B2) at the 3’ end and the same sense as B1c at the 5’ end.
- F3/B3 (Forward/Backward Outer Primers): Short primers complementary to F3c and B3c.
- LF/LB (Loop Primers, optional): Accelerate amplification by binding to loop regions formed between F1/F2 and B1/B2.
The Core Cycle: Initiation, Elongation, and Looping
Phase I: Initial Synthesis and Strand Displacement
- FIP Binding & Extension: FIP binds to the F2c region on the target DNA. The Bst polymerase with high strand displacement activity initiates synthesis.
- F3 Primer Displacement: The outer primer F3 binds upstream to F2c (F3c region) and initiates synthesis. This nascent strand displaces the FIP-extended strand, releasing a single-stranded DNA intermediate.
- Formation of the 5’ End Loop: The released intermediate contains complementary F1c and F1 regions at its ends. These self-anneal, forming a 3’ end looped structure (dumbbell-shaped DNA), which serves as the primary amplification template.
Phase II: Cycling Amplification & Loop Formation
- Self-Priming and Exponential Synthesis: The 3’ end of the dumbbell (F1 region) serves as a primer for self-directed synthesis around the loop, regenerating the F1c sequence. Simultaneously, BIP binds to the complementary B2 region on the same strand, initiating synthesis that displaces the self-primed strand, leading to the formation of a double-loop (dumbbell) structure with complementary 5’ and 3’ ends.
- Strand Displacement Cycling: This final dumbbell structure is the workhorse of LAMP. Continuous self-priming from both ends and strand displacement by inner primers (FIP/BIP) and loop primers (LF/LB) yields a mixture of elongated stem-loop DNAs with various stem lengths and cauliflower-like structures due to multiple inverted repeats.
Diagram 1: Core LAMP mechanism: Strand displacement and loop formation.
Quantitative Performance Data in SARS-CoV-2 Detection
Recent studies benchmark LAMP performance against gold-standard RT-qPCR.
Table 1: Performance Metrics of SARS-CoV-2 LAMP Assays (Selected Studies, 2023-2024)
| Target Gene | Limit of Detection (LoD) | Time to Positive | Sensitivity (%) | Specificity (%) | Reference Assay |
|---|---|---|---|---|---|
| ORF1ab & N | 10 copies/µL | 15-20 min | 98.5 | 99.8 | CDC RT-qPCR |
| E & N | 5.2 copies/µL | < 30 min | 97.1 | 100 | WHO-validated PCR |
| N gene only | 100 copies/µL | 40 min | 95.0 | 98.2 | Commercial RT-qPCR |
| S gene | 12.5 copies/µL | 25 min | 99.0 | 99.5 | Multiplex RT-qPCR |
Table 2: Impact of Key Reaction Parameters on LAMP Efficiency
| Parameter | Optimal Range | Effect of Deviation | Recommended Optimization Step |
|---|---|---|---|
| Temperature | 60-65°C | <60°C: Slower kinetics, reduced specificity. >67°C: Enzyme inactivation. | Gradient testing (58-68°C). |
| Mg²⁺ Concentration | 4-8 mM | Critical for polymerase activity. Too low: No amplification. Too high: Non-specific amplification. | Titration (2-10 mM). |
| dNTPs | 1.0-1.4 mM | Lower reduces yield; higher can inhibit reaction. | Fixed at 1.2 mM for initial screens. |
| Betaine | 0.6-1.2 M | Reduces secondary structure, improves strand displacement. Essential for GC-rich targets. | Include at 0.8 M standard. |
| Polymerase (Bst 3.0) | 8-16 U/reaction | Higher units accelerate time-to-positive; cost trade-off. | Start with 8 U, increase if LoD is insufficient. |
Detailed Experimental Protocol: SARS-CoV-2 LAMP Assay
Primer Design Protocol
- Sequence Retrieval: Obtain SARS-CoV-2 target sequence (e.g., N gene, NC_045512.2) from NCBI GenBank.
- Region Selection: Using software (e.g., PrimerExplorer V5, Eiken Chemical), define six distinct regions: F3, F2, F1, B1, B2, B3, in that order, from the 5' to 3' end of the sense strand.
- Primer Specification:
- FIP: 5’-(F1c)-TTTT-(F2)-3’ (≈40-45 nt).
- BIP: 5’-(B1c)-TTTT-(B2c)-3’ (≈40-45 nt).
- F3/B3: 18-22 nt, Tm ≈ 55-60°C.
- LF/LB: 18-22 nt, targeting sequences between F1/F2 and B1/B2.
- Specificity Check: BLAST all primers against the human genome and respiratory pathogen databases.
One-Step Reverse Transcription LAMP (RT-LAMP) Reaction Setup
- Total Volume: 25 µL.
- Reagent Master Mix:
- 1.5 µL Primer Mix (FIP/BIP: 40 µM each; LF/LB: 20 µM each; F3/B3: 5 µM each)
- 12.5 µL 2x LAMP Buffer (40 mM Tris-HCl pH 8.8, 20 mM KCl, 16 mM MgSO₄, 20 mM (NH₄)₂SO₄, 0.2% Tween-20, 1.6 M Betaine)
- 2.5 µL dNTP Mix (10 mM each)
- 1.0 µL Bst 3.0 DNA Polymerase (8-16 U/µL)
- 1.0 µL WarmStart RTx Reverse Transcriptase (or equivalent)
- 2.0 µL Template RNA (or nuclease-free water for NTC)
- Up to 25 µL with nuclease-free water.
- Cycling Conditions: Incubate at 63°C for 30-60 minutes; enzyme inactivation at 80°C for 5 min.
- Detection: Real-time turbidity (650 nm), fluorescence (intercalating dye like SYTO-9), or endpoint colorimetric change (pH indicator like phenol red).
Diagram 2: RT-LAMP experimental workflow for SARS-CoV-2.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Strand Displacement & LAMP Research
| Reagent/Material | Function & Role in Core Principle | Example Product/Supplier |
|---|---|---|
| Bst 3.0 DNA Polymerase | Engine of the reaction. High strand displacement activity at isothermal temperatures (60-65°C). Lacks 5’→3’ exonuclease activity. | New England Biolabs, WarmStart Bst 3.0 |
| WarmStart Reverse Transcriptase | For RT-LAMP. Converts target SARS-CoV-2 RNA into cDNA for amplification. Engineered for activity at LAMP temperature. | WarmStart RTx (NEB), Maxima H Minus |
| LAMP Primer Mix (Custom) | Specificity drivers. Inner primers (FIP/BIP) initiate strand displacement and loop formation. Outer primers (F3/B3) enable initial displacement. | IDT, Metabion, Eurofins |
| Isothermal Amplification Buffer with Betaine | Reaction environment. Betaine is critical for equalizing DNA strand stability, facilitating strand displacement and loop formation in GC-rich regions. | Thermo Scientific, Lucigen |
| Magnesium Sulfate (MgSO₄) | Cofactor for Bst polymerase. Concentration is the most critical variable for optimizing speed and specificity of strand displacement synthesis. | Sigma-Aldrich |
| dNTP Set | Building blocks for DNA synthesis. Required at higher concentrations than PCR due to the high yield of the LAMP reaction. | Thermo Fisher Scientific |
| Fluorescent DNA Intercalator (SYTO-9) | Real-time detection. Binds dsDNA in amplification products (cauliflower structures), allowing real-time monitoring of strand displacement synthesis. | Invitrogen |
| Colorimetric pH Indicator (Phenol Red) | Endpoint visual detection. The high yield of DNA synthesis releases pyrophosphates, proton release lowers pH, causing a color change from red to yellow. | Sigma-Aldrich |
Within the context of developing robust Loop-Mediated Isothermal Amplification (LAMP) assays for SARS-CoV-2 detection, a multi-target primer design philosophy is paramount. Relying on a single genomic region for detection poses a significant risk due to the virus's evolutionary rate and the emergence of variants. This guide details the technical rationale and methodology for designing primers against 6-8 conserved regions of the SARS-CoV-2 genome to ensure assay resilience, sensitivity, and specificity.
Genomic Rationale and Target Selection
The SARS-CoV-2 genome is approximately 30 kb of positive-sense single-stranded RNA. Key structural and functional genes, while subject to mutation, contain regions of high conservation essential for viral replication and viability. Targeting multiple conserved regions mitigates the impact of point mutations or deletions in any single area.
Recommended Target Genes & Regions:
- ORF1ab (Replicase Complex): Highly conserved, essential for replication. Contains multiple non-structural proteins (nsp).
- N Gene (Nucleocapsid): Highly expressed, moderately conserved. A common target for diagnostic assays.
- E Gene (Envelope): Highly conserved, essential for virion assembly.
- S Gene (Spike): Critical for entry but has higher mutation rate (variant hotspot). Use only highly conserved sub-regions (e.g., S-gene conserved region).
- RdRP (RNA-dependent RNA polymerase): Located within ORF1ab, essential and conserved.
- ORF3a / ORF8: Can be included for breadth, but conservation should be rigorously verified.
Table 1: Quantitative Comparison of SARS-CoV-2 Genomic Targets for LAMP Primer Design
| Target Gene | Genomic Position (approx.) | Conservation Score (Relative)* | Recommended # of Primer Sets | Key Considerations |
|---|---|---|---|---|
| ORF1ab (RdRP) | 13,442 - 16,236 | Very High (0.99) | 1-2 | Essential function, excellent conservation. High primer design success rate. |
| N Gene | 28,274 - 29,533 | High (0.97) | 2 | High transcript abundance, boosts sensitivity. Some variant drift observed. |
| E Gene | 26,245 - 26,472 | Very High (0.99) | 1 | Short, highly conserved. Ideal for a compact primer set. |
| S Gene (Conserved Region) | 23,168 - 23,387 | Moderate (0.95) | 1 | Useful for breadth but requires careful design to avoid variant sites. |
| ORF1ab (nsp regions) | Varies (e.g., nsp3, nsp12) | High (0.98) | 1-2 | Large gene allows selection of optimal conserved stretches. |
| ORF3a | 25,393 - 26,220 | Moderate (0.94) | 1 | Can provide additional target but monitor for deletions. |
*Conservation score is a relative metric based on recent global sequence alignment entropy analysis (lower mutation frequency).
Core Primer Design Methodology for Multi-Target LAMP
In Silico Design and Conservation Analysis Protocol
Step 1: Sequence Database Compilation
- Download a comprehensive, up-to-date dataset of SARS-CoV-2 complete genomes from repositories such as GISAID and NCBI GenBank. Aim for a diverse set spanning the pandemic timeline and all Variants of Concern (VOCs).
- Perform multiple sequence alignment (MSA) using tools like MAFFT or Clustal Omega.
Step 2: Conservation Plotting and Region Identification
- Generate a conservation plot from the MSA using BioEdit or similar software, calculating Shannon entropy or percent identity at each position.
- Visually identify 6-8 genomic stretches of 150-250 bp with the lowest entropy (highest conservation). Ensure they are spaced across the genome.
Step 3: Primer Design Using Dedicated Software
- Input each selected conserved region individually into LAMP-specific design software (e.g., PrimerExplorer V5, NEB LAMP Designer).
- Design Parameters:
- Tm: 60-65°C for FIP/BIP, 55-60°C for F3/B3, 65-68°C for LoopF/LoopB (if used).
- Length: F2/B2: 18-22 bp; F1c/B1c: 16-20 bp; F3/B3: 16-20 bp.
- GC Content: 40-60%.
- Delta G: Ensure hairpin formation and dimerization are minimized (ΔG > -4 kcal/mol).
- Specificity: Perform in silico BLAST against the human genome and common respiratory flora.
Step 4: Combinatorial Validation
- Simulate interactions between all primers from the 6-8 sets to rule out cross-set dimerization that could reduce efficiency.
- Check for primer homology to other human coronaviruses (HKU1, OC43, NL63, 229E) to ensure SARS-CoV-2 specificity.
Experimental Validation Protocol
Protocol: Multiplex LAMP Reaction Setup (Single-Tube, Multi-Target)
- Reaction Mix (25 µL total):
- 1.4 mM each dNTP
- 6 mM MgSO4
- 20 mM Tris-HCl (pH 8.8), 10 mM KCl, 10 mM (NH4)2SO4, 0.1% Tween 20
- 1.6 µM each Inner Primer (FIP/BIP for each of the 6-8 sets)
- 0.2 µM each Outer Primer (F3/B3 for each of the 6-8 sets)
- 0.8 µM each Loop Primer (LF/LB for each set, if designed) [Note: This increases primer complexity]
- 8 U Bst 2.0 or 3.0 DNA Polymerase (large fragment)
- 5 µL of extracted RNA or viral transport media
- Optional: Fluorescent intercalating dye (e.g., 1X SYTO 9) or colorimetric dye (e.g., 120 µM Hydroxynaphthol Blue).
- Thermocycling Conditions: 63-65°C for 30-60 minutes, followed by enzyme inactivation at 80°C for 5 min.
- Detection: Real-time fluorescence monitoring or endpoint turbidity/color change.
Protocol: Analytical Specificity and Sensitivity (LoD) Testing
- Specificity: Test the primer panel against RNA/DNA from related coronaviruses (MERS-CoV, HCoV-OC43), other respiratory viruses (Influenza A, RSV), and negative human genomic DNA.
- Sensitivity (Limit of Detection - LoD):
- Serially dilute a quantified SARS-CoV-2 RNA standard (e.g., from ATCC or BEI Resources) in nuclease-free water or negative matrix.
- Run the LAMP assay in replicates (n≥8) for each dilution.
- Determine the LoD as the lowest concentration at which ≥95% of replicates are positive.
- Variant Testing: Test against synthetic controls or extracted RNA from key VOCs (Omicron lineages) to confirm detection.
Visualizing the Multi-Target LAMP Strategy
Multi-Target LAMP Assay Resilience Workflow
LAMP Primer Binding to a Single Target Region
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Multi-Target SARS-CoV-2 LAMP Assay Development
| Item | Function & Rationale | Example/Note |
|---|---|---|
| Thermostable Reverse Transcriptase | Converts SARS-CoV-2 RNA to cDNA at elevated temperatures, compatible with isothermal conditions. | WarmStart RTx (NEB), or Bst polymerase with inherent RT activity (Bst 3.0). |
| Strand-Displacing DNA Polymerase | Core enzyme for LAMP; displaces strands during synthesis enabling loop formation. | Bst 2.0 or 3.0 DNA Polymerase (Large Fragment). Bst 3.0 offers faster kinetics. |
| dNTP Mix | Building blocks for DNA synthesis. | High-purity, PCR-grade dNTPs at standard concentration (1.4 mM final). |
| Isothermal Amplification Buffer | Provides optimal pH, ionic strength, and Mg2+ concentration for enzyme activity. | Commercial Bst polymerase buffers often contain MgSO4; optimization may be needed. |
| Fluorescent or Colorimetric Detection Reagent | Allows real-time or endpoint visualization of amplification. | Intercalating dyes (SYTO 9, EvaGreen), or metal indicator dyes (HNB, phenol red). |
| SARS-CoV-2 RNA Positive Control | Quantified standard for assay calibration, sensitivity (LoD) testing, and reproducibility. | ATCC VR-1986HK, BEI Resources quantified genomic RNA. |
| Synthetic DNA Templates (gBlocks) | Controls for individual primer sets, allowing troubleshooting of multi-plex reactions. | IDT gBlocks Gene Fragments for each of the 6-8 target regions. |
| RNase/DNase Inhibitor | Protects viral RNA and synthesized cDNA from degradation during reaction setup. | Essential when using crude samples or long incubation times. |
| Nucleic Acid Extraction Kit | For purifying RNA from clinical specimens (nasopharyngeal swabs, saliva). | Magnetic bead-based kits (e.g., from Qiagen, Thermo Fisher) offer high throughput. |
Within the context of developing robust and sensitive Loop-Mediated Isothermal Amplification (LAMP) assays for SARS-CoV-2 detection, the selection of viral genomic targets is paramount. This technical guide details the four core structural and non-structural gene targets—ORF1ab, Nucleocapsid (N), Envelope (E), and Spike (S)—that underpin most diagnostic and research efforts. Their conserved regions, expression levels, and functional roles make them ideal for primer design, ensuring high specificity and sensitivity in molecular detection assays like LAMP, which operates at a constant temperature and offers rapid results critical for pandemic response.
Target Gene Characteristics and Quantitative Data
Table 1: Core Genomic Targets for SARS-CoV-2 Detection
| Gene | Genomic Location (nt) | Protein Length (aa) | Key Function | Relative Abundance in Virion/Viral RNA | Suitability for LAMP (Rationale) |
|---|---|---|---|---|---|
| ORF1ab | 266-21,562 | ~7,096 (pp1ab) | Viral replication/transcription; encodes non-structural proteins (Nsps). | High (as genomic RNA) | Highly conserved; large sequence for primer design; confirms active replication. |
| Spike (S) | 21,563-25,384 | 1,273 | Host cell receptor binding, membrane fusion, major antigen. | Moderate (as subgenomic RNA) | Critical for variant tracking; contains Receptor-Binding Domain (RBD); less conserved. |
| Envelope (E) | 26,245-26,472 | 75 | Virion assembly, budding, and pathogenesis. | High (as subgenomic RNA) | Highly conserved; small gene ideal for short amplicons; excellent sensitivity target. |
| Nucleocapsid (N) | 28,274-29,533 | 419 | RNA genome packaging, virion assembly, modulates host cell processes. | Very High (as subgenomic RNA) | Most abundant viral transcript; highly conserved; robust detection target. |
Table 2: Example Primer Performance Metrics in SARS-CoV-2 LAMP Assays
| Target Gene | Primer Set Name (Example) | Amplicon Size (bp) | Reported Limit of Detection (LoD) | Time to Positive (min) | Key References |
|---|---|---|---|---|---|
| ORF1ab | ORF1a-LAMP | 120 | 10 copies/µL | 20 | Zhang et al., 2020 |
| Spike (S) | S-gene-LAMP (RBD) | 185 | 50 copies/µL | 25 | Kitagawa et al., 2020 |
| Envelope (E) | E-gene-LAMP (WHO) | 113 | 5 copies/µL | 15 | Corman et al., 2020 |
| Nucleocapsid (N) | N-gene-LAMP | 144 | 2 copies/µL | 18 | Dao Thi et al., 2020 |
Detailed Experimental Protocol: Multiplex RT-LAMP for SARS-CoV-2
Protocol Title: One-Step Multiplex RT-LAMP for Simultaneous Detection of SARS-CoV-2 N and E Genes.
Principle: This protocol uses reverse transcription and LAMP in a single tube, targeting two highly conserved genes (N and E) to increase assay reliability and reduce false negatives. Detection is achieved via real-time fluorescence (intercalating dye) or colorimetric change (pH indicator).
Materials & Reagents:
- Template: Heat-inactivated nasopharyngeal swab extract or synthetic SARS-CoV-2 RNA.
- Primers: Six primers per target (F3, B3, FIP, BIP, LF, LB) designed against conserved regions of the N and E genes.
- Enzyme Mix: Bst 2.0 or 3.0 DNA polymerase (with high reverse transcriptase activity) or separate AMV/HIV reverse transcriptase.
- Reaction Mix: dNTPs, MgSO₄ (or MgCl₂), betaine, Fluorescent dye (e.g., SYTO 9) or pH indicator (e.g., phenol red).
- Buffer: Isothermal amplification buffer.
- Equipment: Real-time fluorometer or heated block/water bath for isothermal incubation at 60-65°C.
Procedure:
- Primer Design & Validation: Design LAMP primer sets using software (e.g., PrimerExplorer) against reference sequences (e.g., NC_045512.2). Validate specificity via BLAST and in silico PCR. Synthesize and resuspend primers in nuclease-free water.
- Reaction Setup (25 µL total volume):
- In a 0.2 mL tube, combine:
- Isothermal Amplification Buffer (1X final concentration)
- dNTPs (1.4 mM each)
- MgSO₄ (6-8 mM, optimized)
- Betaine (0.8 M)
- Primer Mix (Final concentrations: FIP/BIP: 1.6 µM each, LF/LB: 0.8 µM each, F3/B3: 0.2 µM each for each target gene)
- Fluorescent Dye (e.g., SYTO 9 at 0.5-1X) OR Phenol Red (0.1-0.2 mM)
- Bst 2.0/3.0 WarmStart Polymerase (8-16 units)
- RNA Template (5 µL)
- Nuclease-free water to 25 µL.
- In a 0.2 mL tube, combine:
- Amplification & Detection:
- Place tubes in a real-time fluorometer or thermal block.
- Incubate at 63°C for 30-45 minutes with fluorescence/color read every 30-60 seconds.
- Fluorescent Detection: A cycle threshold (Ct) or sharp increase in fluorescence indicates positive amplification.
- Colorimetric Detection: A change from pink (basic) to yellow (acidic, due to proton release during amplification) indicates a positive reaction. Include no-template (water) and positive RNA controls.
- Analysis: Determine time-to-positive (Tp) for each sample. A sample is positive if Tp is less than the cut-off (e.g., 30 minutes) for one or both targets.
Visualizing LAMP Assay Workflow and Viral Gene Function
Title: RT-LAMP Assay Workflow for SARS-CoV-2 Detection
Title: Core SARS-CoV-2 Gene Functions and Pathways
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for SARS-CoV-2 LAMP Assay Development
| Reagent Category | Specific Product/Example | Function in the Assay | Critical Consideration |
|---|---|---|---|
| Polymerase | Bst 2.0/3.0 WarmStart DNA Polymerase | Isothermal strand-displacing amplification; Bst 3.0 has reverse transcriptase activity for one-step RT-LAMP. | Hot-start feature prevents non-specific amplification; choose version with RT activity for simplified workflows. |
| Primers | HPLC-purified LAMP primer sets (F3, B3, FIP, BIP, LF, LB) | Target-specific binding to initiate and accelerate cyclic amplification. | Design against highly conserved regions; avoid primer-dimer formation; validate specificity. |
| Detection Chemistry | SYTO 9 / SYBR Green I (Fluorescent) or Phenol Red (Colorimetric) | Intercalates into dsDNA (fluorescent) or detects pH drop (colorimetric) for real-time/endpoint detection. | Fluorescent offers quantitation; colorimetric is rapid and equipment-free. Mg2+ concentration affects phenol red. |
| Reaction Enhancers | Betaine, Trehalose | Reduces secondary structure in GC-rich templates (betaine), stabilizes enzymes (trehalose). | Optimize concentration for each primer set to improve speed and sensitivity. |
| Positive Control | Synthetic SARS-CoV-2 RNA (N, E, S, ORF1ab fragments) | Validates assay performance, determines Limit of Detection (LoD), controls for inhibition. | Must be full-length of the amplicon region; use in a non-infectious background (e.g., human RNA). |
| RNase Inhibitor | Recombinant RNase Inhibitor | Protects viral RNA template from degradation during reaction setup. | Essential when using separate RT and polymerase enzymes; improves reproducibility. |
Within the context of advancing diagnostics for SARS-CoV-2 detection, the Loop-Mediated Isothermal Amplification (LAMP) assay has emerged as a compelling alternative to the gold-standard Polymerase Chain Reaction (PCR). This whitepaper details the core technical advantages of LAMP, focusing on its isothermal nature, rapid time-to-result, and instrumental simplicity, which are critical for decentralized testing and high-throughput screening in research and drug development.
Core Technical Advantages: A Comparative Analysis
Isothermal Reaction vs. Thermocycling
The fundamental distinction between LAMP and PCR is the requirement for precise thermal cycling. PCR necessitates a sophisticated instrument to cycle between denaturation (90-95°C), annealing (50-65°C), and extension (72°C) temperatures. In contrast, LAMP amplification occurs at a constant temperature, typically between 60-65°C.
Table 1: Reaction Condition Comparison
| Parameter | PCR | LAMP |
|---|---|---|
| Temperature Profile | Cyclic (3-4 distinct temperatures) | Isothermal (single temperature) |
| Typical Range | 50°C - 95°C | 60°C - 65°C |
| Primary Instrument Requirement | Precision Thermocycler | Simple Heat Block or Water Bath |
| Enzyme(s) | Thermostable DNA Polymerase (e.g., Taq) | Bst DNA Polymerase (strand-displacing activity) |
| Target Complexity | Requires precise primer design for 2 primers. | Requires complex design for 4-6 primers. |
This isothermal characteristic eliminates the need for rapid temperature ramping, directly enabling simpler, lower-cost instrumentation and reduced power consumption.
Speed: Reaction Kinetics and Time-to-Result
LAMP exhibits superior amplification kinetics due to its strand-displacement mechanism and the use of multiple primer sets targeting 6-8 distinct regions on the genome. This allows for exponential synthesis without the time-consuming denaturation and annealing steps of PCR.
Table 2: Speed and Throughput Metrics
| Metric | Typical PCR (qRT-PCR) | Typical LAMP | Notes |
|---|---|---|---|
| Amplification Time | 60 - 120 minutes | 15 - 60 minutes | Time from sample lysis to detection. |
| Time-to-Result (SARS-CoV-2) | 90 - 180 minutes | 30 - 90 minutes | Includes RNA extraction and amplification. |
| Approx. Amplicon Yield | ~10^7 copies in 30 cycles | ~10^9 copies in 30-60 min | LAMP produces a larger mass of DNA. |
| Possibility for Real-Time Monitoring | Yes (via intercalating dyes) | Yes (via turbidity, fluorescence, colorimetry) |
For SARS-CoV-2, recent protocols integrating simplified RNA extraction or direct lysis have demonstrated reliable detection in under 30 minutes from swab to result, a critical advantage for point-of-need testing.
Instrument Simplicity and Cost
The isothermal requirement drastically reduces hardware complexity. A standard thermocycler capable of precise temperature control and rapid ramping costs thousands of dollars. A LAMP reaction can be performed in a heated block, dry bath, or even a modified hand warmer.
Table 3: Instrumentation and Cost Analysis
| Aspect | PCR / qRT-PCR Instrument | LAMP-Compatible Device |
|---|---|---|
| Core Function | Precise thermal cycling, fluorescence detection. | Maintain stable single temperature, optical detection optional. |
| Approx. Cost (USD) | $15,000 - $50,000+ | $500 - $5,000 |
| Footprint & Portability | Benchtop, limited mobility. | Can be handheld, battery-operated. |
| Maintenance | High, requires calibration. | Low. |
| Suitability for Field Use | Low | High |
This simplicity facilitates deployment in resource-limited settings, field laboratories, pharmacies, and for at-home testing models.
Experimental Protocol: A Standard SARS-CoV-2 RT-LAMP Assay
The following is a detailed protocol for a fluorescence-based RT-LAMP assay targeting the N gene of SARS-CoV-2.
Reagents:
- Template: RNA extracted from nasopharyngeal swab samples.
- Primers: A set of 6 primers (F3, B3, FIP, BIP, LF, LB) specific to the SARS-CoV-2 N gene.
- Enzyme Mix: Bst 2.0 or 3.0 DNA polymerase (with reverse transcriptase activity for one-step RT-LAMP).
- Reaction Buffer: Isothermal amplification buffer with MgSO4 (typically 6-8 mM final concentration).
- dNTPs: Deoxynucleotide solution mix.
- Betaine: (1 M final concentration) to destabilize DNA secondary structures.
- Fluorescent Dye: SYTO 9, EvaGreen, or Calcein/MnCl2.
Procedure:
- Reaction Setup: In a 0.2 mL tube or microfluidic chip, prepare a 25 µL reaction mix:
- 12.5 µL 2x Isothermal Amplification Buffer
- 1.5 µL Primer Mix (FIP/BIP: 1.6 µM each; LF/LB: 0.8 µM each; F3/B3: 0.2 µM each)
- 1.0 µL Bst DNA Polymerase WarmStart (8 U/µL)
- 1.0 µL 10 mM dNTPs
- 2.5 µL 10 M Betaine
- 0.5 µL 20x Fluorescent Dye
- 5.0 µL RNA template
- Nuclease-free water to 25 µL.
- Incubation: Place the tube in a pre-heated isothermal instrument at 65°C for 30 minutes.
- Detection:
- Real-Time: Monitor fluorescence every 30-60 seconds. A positive sample shows an exponential increase in fluorescence after a threshold time (Tt).
- End-Point: Visual inspection under blue LED light. A positive reaction will exhibit bright green fluorescence (with Calcein) or a color change from orange to green (with phenol red/H+).
Visualizing the LAMP Workflow and Mechanism
Workflow for SARS-CoV-2 LAMP Detection
Mechanism of LAMP DNA Amplification
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Reagents for SARS-CoV-2 LAMP Research
| Reagent / Material | Function in the Assay | Key Consideration for Research |
|---|---|---|
| Bst 2.0/3.0 WarmStart DNA Polymerase | Strand-displacing polymerase; WarmStart version inhibits activity at room temp to prevent primer-dimer formation. | High processivity and strand displacement efficiency are critical for speed. |
| Target-Specific LAMP Primer Set | 6 primers (F3, B3, FIP, BIP, LF, LB) designed to recognize 8 distinct regions of the target sequence. | Design specificity for SARS-CoV-2 variants is paramount; requires specialized software (e.g., PrimerExplorer). |
| Isothermal Amplification Buffer | Provides optimal pH, salt conditions, and Mg2+ for Bst polymerase activity. | Mg2+ concentration (6-8 mM) must be optimized for each primer set. |
| Betaine (5M stock) | Reduces secondary structure in GC-rich regions, improving primer annealing and strand displacement. | Enhances assay robustness, especially for viral RNA targets. |
| Fluorescent Intercalating Dye (SYTO 9) | Binds double-stranded DNA products, enabling real-time fluorescence monitoring. | Allows for quantitative determination of Tt (time threshold). |
| Colorimetric Indicator (Phenol Red) | pH-sensitive dye; amplification produces protons (H+), causing color change from pink/red to yellow. | Enables visual, instrument-free endpoint detection. |
| Heat Block/Portable Fluorimeter | Maintains constant 60-65°C temperature; fluorimeter monitors real-time fluorescence. | Device simplicity is key; many low-cost, open-source designs exist. |
The isothermal reaction, speed, and instrumental simplicity of LAMP provide distinct practical advantages over PCR. For SARS-CoV-2 detection research and application development, these attributes enable rapid, scalable, and deployable testing solutions. While PCR remains the benchmark for ultimate sensitivity and multiplexing capability in central labs, LAMP's profile makes it an indispensable technology for expanding diagnostic access and throughput in pandemic response.
Within the broader thesis on Loop-Mediated Isothermal Amplification (LAMP) assay principles for SARS-CoV-2 detection, understanding the historical evolution of LAMP technology is critical. Initially developed by Notomi et al. in 2000 for pathogen detection, LAMP's inherent advantages—isothermal conditions, rapid kinetics, and robust amplification—made it a prime candidate for adaptation during the COVID-19 pandemic. This whitepaper details the technical progression, core methodologies, and contemporary implementations of LAMP for coronavirus diagnostics, providing a comprehensive guide for research and development professionals.
Historical Progression and Performance Metrics
The adaptation of LAMP for SARS-CoV-2 required significant optimization from its original design. Key milestones include primer redesign for the novel coronavirus genome, integration with reverse transcription (RT-LAMP), and the development of endpoint and real-time detection strategies. The following table summarizes the quantitative performance evolution of representative RT-LAMP assays for SARS-CoV-2.
Table 1: Evolution of SARS-CoV-2 RT-LAMP Assay Performance Characteristics
| Year | Target Gene(s) | Reported Limit of Detection (LoD) | Average Time-to-Result | Clinical Sensitivity | Clinical Specificity | Reference |
|---|---|---|---|---|---|---|
| 2020 | ORF1ab, N, S | 10-100 RNA copies/µL | 20-40 min | 95.2% | 100% | (Early pandemic protocols) |
| 2021 | Multiplex (N, E, RdRp) | 5-20 RNA copies/µL | 15-30 min | 98.1% | 99.4% | (Improved primer sets) |
| 2022-2023 | N gene (highly conserved regions) | 2-10 RNA copies/µL | <20 min | 99.0% | 99.8% | (Current optimized assays) |
| 2023-2024 | Variant-specific markers | 5-15 RNA copies/µL | 25-35 min | 98.5% (for variants) | 99.5% | (Variant discrimination assays) |
Core Experimental Protocol: SARS-CoV-2 RT-LAMP
This detailed protocol is representative of a current, optimized methodology for detecting SARS-CoV-2 RNA.
1. Primer Design and Preparation:
- Target: Conserved regions of the SARS-CoV-2 Nucleocapsid (N) gene.
- Primer Set: Design six primers: F3, B3, FIP (F1c+F2), BIP (B1c+B2), LF, and LB.
- Bioinformatics: Align sequences from global SARS-CoV-2 isolates (e.g., from GISAID) to ensure conservation. Check specificity against other human coronaviruses and respiratory flora. Resuspend primers in nuclease-free water to 100 µM stock solutions. Prepare a working primer mix containing all six primers at final concentrations of 1.6 µM (FIP/BIP) and 0.2 µM (F3/B3/LF/LB).
2. Sample Processing and RNA Extraction:
- Sample Type: Nasopharyngeal/oropharyngeal swabs in viral transport media (VTM) or saliva.
- Method 1 (Rapid): Use a rapid heating protocol (e.g., 95°C for 5 min followed by ice) to inactivate virus and release RNA, with subsequent clarification by brief centrifugation.
- Method 2 (Purified): Use magnetic bead- or column-based nucleic acid extraction kits (e.g., QIAamp Viral RNA Mini Kit) per manufacturer's instructions. Elute in 50-100 µL elution buffer.
3. RT-LAMP Reaction Setup:
- Master Mix (25 µL total volume):
- 12.5 µL 2x Isothermal Amplification Buffer (contains dNTPs, MgSO4, betaine)
- 2.5 µL Primer Working Mix (from step 1)
- 1.0 µL Enzyme Mix (commercially available Bst 2.0/3.0 DNA polymerase + WarmStart RTx reverse transcriptase)
- 5-8 µL Template RNA (from step 2)
- Nuclease-free water to 25 µL
- Detection Method Integration:
- Colorimetric (pH): Add 1-2 µL of phenol red (or other pH-sensitive dye) to the master mix. A positive reaction changes from pink/red to yellow.
- Fluorescent (Intercalating Dye): Add 1x final concentration of SYTO 9 or similar dye for real-time or endpoint fluorescence measurement.
- Turbidity: Monitor magnesium pyrophosphate precipitate formation in real-time at 400 nm.
4. Amplification and Detection:
- Place reaction tubes in a heating block, water bath, or portable isothermal instrument.
- Incubate at 63-65°C for 20-40 minutes. Do not use a thermal cycler lid.
- Real-time Monitoring: If using fluorescent dye, measure fluorescence every 30-60 seconds.
- Endpoint Analysis:
- Visual color change (pink→yellow).
- Under blue LED light with appropriate filter, visualize green fluorescence.
- Assess turbidity by eye or spectrophotometer.
5. Controls:
- Negative Control: Nuclease-free water instead of template.
- Positive Control: Synthetic RNA containing the target N gene region at a known concentration (e.g., 50 copies/µL).
- Internal Control: For clinical validation, a human housekeeping gene (e.g., RNase P) can be co-amplified in a separate primer set to check sample quality.
Visualizing the RT-LAMP Workflow and Mechanism
Diagram 1: SARS-CoV-2 RT-LAMP Diagnostic Workflow
Diagram 2: RT-LAMP Molecular Mechanism for SARS-CoV-2 RNA
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Research Reagent Solutions for SARS-CoV-2 RT-LAMP Development
| Item | Function & Rationale | Example Product/Chemical |
|---|---|---|
| Thermostable DNA Polymerase | Catalyzes DNA synthesis at isothermal temperatures (~65°C). Bst 2.0/3.0 offers high strand displacement activity critical for LAMP. | Bst 2.0/3.0 WarmStart DNA Polymerase (NEB) |
| Reverse Transcriptase | Converts SARS-CoV-2 genomic RNA into complementary DNA (cDNA) at the same isothermal temperature, enabling one-step RT-LAMP. | WarmStart RTx Reverse Transcriptase |
| LAMP Primer Mix | Set of 4-6 primers specifically designed against conserved regions of the SARS-CoV-2 genome (e.g., N, E, ORF1ab). Defines assay specificity. | Custom synthesized oligos (e.g., IDT, Thermo Fisher) |
| Isothermal Amplification Buffer | Provides optimized concentrations of Mg2+ (for polymerase activity), dNTPs (building blocks), and betaine (to destabilize DNA secondary structures). | Commercial 2x Isothermal Amp Buffer or custom formulation. |
| Visual Detection Dye | Enables endpoint colorimetric detection. pH-sensitive dyes (phenol red) change color due to proton release during amplification. | Phenol Red, Hydroxy Naphthol Blue (HNB) |
| Fluorescent Detection Dye | Intercalates into double-stranded LAMP products, allowing real-time quantification or vivid endpoint fluorescence. | SYTO 9, SYBR Green, EvaGreen |
| RNA Extraction/Purification Kit | Isolates and purifies viral RNA from complex clinical matrices, removing inhibitors that can reduce assay sensitivity. | QIAamp Viral RNA Mini Kit, MagMAX Viral/Pathogen kits |
| Synthetic RNA Control | Non-infectious, quantitated RNA containing the target sequence. Serves as essential positive control and for determining Limit of Detection (LoD). | SARS-CoV-2 RNA Transcripts (e.g., from Twist Bioscience) |
| Nuclease-Free Water | Solvent for all reagents and reactions. Must be certified nuclease-free to prevent degradation of RNA, primers, and enzymes. | Ambion Nuclease-Free Water |
| Uracil-DNA Glycosylase (UDG) | Contamination control enzyme. Degrades carryover amplicons containing dUTP (which can be incorporated instead of dTTP), preventing false positives. | Thermolabile UDG (supplied in some master mixes) |
Step-by-Step Protocol: Implementing SARS-CoV-2 LAMP in Research and Development
Within the broader research on Loop-Mediated Isothermal Amplification (LAMP) for SARS-CoV-2 detection, sample preparation is the critical first step determining assay sensitivity, speed, and suitability for point-of-care deployment. This guide provides a technical comparison of traditional RNA extraction from nasopharyngeal (NP) swabs versus emerging direct protocols for saliva, evaluating their compatibility with downstream LAMP.
Comparative Analysis: NP Swab vs. Saliva
Table 1: Specimen Source Characteristics for SARS-CoV-2 Detection
| Parameter | Nasopharyngeal (NP) Swab | Saliva |
|---|---|---|
| Collection | Invasive, requires trained healthcare professional | Non-invasive, can be self-administered |
| Patient Comfort | Low | High |
| Stability at Room Temp | Moderate (in VTM); 24-72h | High; stable for days, contains inherent nucleases |
| Viral Load | Generally high in acute phase; site of primary infection | Variable; can correlate with NP, especially in lower respiratory disease |
| Inhibitor Content | Moderate (mucus, blood) | High (food debris, bacteria, endogenous enzymes) |
| Ideal for LAMP POC? | Less suitable | Highly suitable |
Table 2: RNA Extraction vs. Direct Protocol Comparison
| Aspect | RNA Extraction-Based Protocol | Direct Protocol (No Extraction) |
|---|---|---|
| Process Time | 60-90 minutes | <5-10 minutes |
| Hands-on Time | High | Minimal |
| Cost per Sample | High ($5-$15) | Low (<$1) |
| Equipment Needed | Centrifuge, magnetic rack, thermomixer | Heat block/water bath |
| RNA Purity/Yield | High purity, concentrated RNA | Crude, with inhibitors present |
| Compatibility with LAMP | Excellent; clean template reduces inhibition risk | Variable; requires inhibitor-resistant enzymes/buffers |
| Assay Sensitivity Loss | Minimal | Can be 1-2 log10 reduction vs. extraction |
| Automation Potential | High | Limited |
Detailed Methodologies
RNA Extraction from NP Swabs (Magnetic Bead-Based)
This protocol is the gold standard for downstream molecular assays.
- Sample Inactivation: Mix 200 µL of NP swab in Viral Transport Media (VTM) with an equal volume of proteinase K-containing lysis buffer (e.g., AVL buffer). Incubate at 56°C for 10 minutes.
- Binding: Add 400 µL of 96-100% ethanol to the lysate. Transfer to a plate/ tube containing magnetic silica beads. Mix thoroughly and incubate at room temperature for 5 minutes.
- Washes: Place on a magnetic rack. Discard supernatant. Wash beads twice with 700 µL Wash Buffer 1 (e.g., AW1), and once with 500 µL Wash Buffer 2 (e.g., AW2). Perform a final wash with 80% ethanol. Dry beads for 5-10 minutes.
- Elution: Remove from magnetic rack. Elute RNA in 50-100 µL of nuclease-free water or TE buffer. Incubate at 65°C for 5 minutes, then place on magnetic rack and transfer pure RNA to a clean tube.
Direct Saliva Protocol for LAMP
This protocol bypasses extraction, ideal for rapid screening.
- Sample Collection & Inactivation: Collect ~1 mL of saliva in a sterile tube. Heat-inactivate at 95°C for 5-30 minutes (e.g., 95°C for 5 min) to inactivate virus and nucleases.
- Clarification & Dilution: Centrifuge at 10,000 x g for 2 minutes to pellet debris. Use supernatant directly.
- LAMP Reaction Setup: Prepare a master mix containing:
- 1.0-1.6 µM of each FIP/BIP primer
- 0.2 µM of each F3/B3 primer
- 0.4-0.8 µM LoopF/LoopB primers (if used)
- 1.4 mM dNTPs
- 6 mM MgSO4
- 1X ThermoPol or ISO-001 Reaction Buffer
- 8 U Bst 2.0/3.0 DNA Polymerase (or 4-16 U Bst LF)
- 1 µL fluorescent dye (e.g., SYTO-9, 10X) or HNB (120 µM)
- Add 2-5 µL of heat-treated saliva supernatant directly to 20-25 µL master mix.
- Amplification & Detection: Incubate at 60-65°C for 25-40 minutes. Monitor real-time fluorescence or perform end-point colorimetric detection (HNB: sky blue -> violet for positive).
Visualization of Workflows
Title: RNA Extraction Workflow from NP Swabs
Title: Direct Saliva Protocol for LAMP Assay
Title: LAMP Amplification and Signal Generation Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Sample Preparation and LAMP
| Item | Function & Rationale |
|---|---|
| Viral Transport Media (VTM) | Preserves viral RNA integrity from NP swabs during transport. |
| Proteinase K & Lysis Buffer | Inactivates virus, degrades nucleases/proteins, and releases RNA for extraction. |
| Magnetic Silica Beads | Bind nucleic acids selectively in high-salt conditions; enable automatable washing. |
| Wash Buffers (AW1/AW2) | Remove contaminants, salts, and inhibitors while keeping RNA bound to beads. |
| Bst 2.0 or 3.0 DNA Polymerase | Thermostable polymerase with high strand displacement activity essential for LAMP. |
| ISO-001 or WarmStart LAMP Buffer | Optimized buffer for Bst polymerase, often includes betaine to reduce secondary structure. |
| SYTO-9 or SYBR Green Dye | Intercalating fluorescent dyes for real-time LAMP monitoring. |
| Hydroxy Naphthol Blue (HNB) | Metal ion indicator for colorimetric end-point detection (blue -> violet with Mg2+ depletion). |
| Heat Block/Water Bath | Provides precise 60-65°C for LAMP and 95°C for saliva inactivation. |
| Microcentrifuge | Clarifies heat-treated saliva by pelleting debris. |
Within the ongoing research on LAMP assay principles for SARS-CoV-2 detection, the reaction master mix is the foundational biochemical environment that dictates the assay's success. This whitepaper provides an in-depth technical guide to its components, their optimized concentrations, and commercially available kits, framing this within the requirements for robust, field-deployable diagnostics.
Core Components and Their Functions
A LAMP master mix for SARS-CoV-2 detection is engineered to enable isothermal amplification of specific viral targets (e.g., N, E, or ORF1ab genes) with high speed, specificity, and yield. Its components fulfill distinct but interdependent roles.
1. Buffer System: Typically contains Tris-HCl, potassium chloride (KCl), ammonium sulfate ((NH₄)₂SO₄), and magnesium ions (Mg²⁺). The pH (usually 8.0-8.8) and ionic strength are optimized for Bst DNA polymerase activity. Mg²⁺ is a critical cofactor for polymerase activity and also influences primer dimer formation and amplification efficiency.
2. Bst DNA Polymerase Large Fragment: The core enzyme derived from Geobacillus stearothermophilus. It possesses high strand-displacement activity, allowing it to unwind DNA duplexes without a separate denaturation step, which is essential for isothermal amplification. Its lack of 5'→3' exonuclease activity prevents unintended degradation of primers and probes.
3. Deoxynucleotide Triphosphates (dNTPs): The building blocks (dATP, dCTP, dGTP, dTTP) for DNA synthesis. They are provided at balanced, millimolar concentrations to ensure high-fidelity incorporation and prevent polymerase stalling.
4. Betaine: A chemical destabilizer added to equalize the melting temperatures of DNA rich in different base compositions. It is crucial for dealing with complex, secondary structures in the target SARS-CoV-2 genome, ensuring efficient strand displacement and primer annealing.
5. Primers: A set of 4 to 6 primers targeting 6-8 distinct regions of the SARS-CoV-2 genome.
- FIP (Forward Inner Primer): Contains the F2 sequence (complementary to the target) at its 3' end and the same sense F1c sequence at its 5' end.
- BIP (Backward Inner Primer): Contains the B2 sequence (complementary) at its 3' end and the B1c sense sequence at its 5' end.
- F3 (Forward Outer Primer): A short primer complementary to a region upstream of F2.
- B3 (Backward Outer Primer): A short primer complementary to a region upstream of B2.
- (Optional) Loop Primer (LF/LB): Accelerate the reaction by binding to loop structures formed during amplification, significantly reducing time-to-result.
6. Reverse Transcriptase (for RT-LAMP): For direct RNA detection, a thermostable reverse transcriptase (e.g., Bst polymerase with RT activity, or a separate enzyme like HIV-1 RT or M-MLV) is included to generate cDNA from the viral RNA template prior to amplification.
7. Detection Additives: Visual detection often relies on metal ion indicators (e.g., Hydroxy Naphthol Blue, HNB; or Calcein with Mn²⁺) that undergo a color change upon binding to magnesium pyrophosphate, a byproduct of amplification. For fluorometric detection, intercalating dyes (SYTO 9, EvaGreen) or sequence-specific probes are used.
Optimized Concentration Ranges
Optimal concentrations are derived from empirical optimization for SARS-CoV-2 targets. The table below summarizes typical final concentrations in a reaction.
Table 1: Typical Component Concentrations in a SARS-CoV-2 RT-LAMP Master Mix
| Component | Typical Final Concentration | Function & Rationale |
|---|---|---|
| Tris-HCl Buffer | 10-20 mM (pH 8.0-8.8) | Maintains optimal enzymatic pH. |
| KCl | 10-50 mM | Provides ionic strength for primer annealing. |
| (NH₄)₂SO₄ | 5-20 mM | Enhances Bst polymerase activity and specificity. |
| MgSO₄ | 4-8 mM | Critical cofactor for polymerase; concentration is finely tuned. |
| Bst 2.0/3.0 Polymerase | 0.08-0.32 U/µL | Catalyzes strand-displacing DNA synthesis. |
| dNTPs (each) | 0.8-1.4 mM | Substrates for DNA polymerization. |
| Betaine | 0.6-1.2 M | Reduces secondary structure, homogenizes Tm. |
| Inner Primers (FIP/BIP) | 0.8-2.0 µM each | Initiate loop-forming amplification. |
| Outer Primers (F3/B3) | 0.1-0.4 µM each | Initiate strand displacement. |
| Loop Primers (LF/LB) | 0.4-1.0 µM each | Accelerate reaction by binding loop regions. |
| Reverse Transcriptase | 0.05-0.15 U/µL (if separate) | Converts SARS-CoV-2 RNA to cDNA. |
| HNB Indicator | ~120 µM | Visual detection: blue → violet/purple with Mg²⁺ depletion. |
Detailed Experimental Protocol: RT-LAMP for SARS-CoV-2
Protocol: One-Step Colorimetric RT-LAMP for SARS-CoV-2 N Gene Detection
I. Primer Design & Reconstitution
- Design primers targeting conserved regions of the SARS-CoV-2 N gene using software (e.g., PrimerExplorer V5). Validate specificity via BLAST against the human genome and other coronaviruses.
- Reconstitute lyophilized primers in nuclease-free TE buffer or water to a stock concentration of 100 µM. Store at -20°C.
- Prepare a primer mix by combining stocks to create a 10X working solution: FIP (16 µM), BIP (16 µM), F3 (2 µM), B3 (2 µM), LF (8 µM), LB (8 µM) in nuclease-free water.
II. Master Mix Preparation (for one 25 µL reaction) Perform on ice in a nuclease-free, low-binding microcentrifuge tube. Prepare a bulk mix for n+2 reactions to account for pipetting error.
| Reagent | Volume per 25 µL rxn | Final Concentration |
|---|---|---|
| 2X Isothermal Amplification Buffer (with Mg²⁺, dNTPs, betaine) | 12.5 µL | 1X |
| 10X Primer Mix | 2.5 µL | 1X |
| Bst 2.0 WarmStart RT/x Polymerase Mix | 1.0 µL | 0.32 U/µL Bst, 0.08 U/µL RT |
| 10 mM HNB (optional, for colorimetric) | 0.3 µL | ~120 µM |
| Nuclease-free Water | 6.2 µL | - |
| Total Master Mix Volume | 22.5 µL |
III. Reaction Assembly & Amplification
- Aliquot 22.5 µL of master mix into each reaction tube (0.2 mL PCR strips or individual tubes).
- Add 2.5 µL of template (SARS-CoV-2 RNA extract) or nuclease-free water (No-Template Control, NTC) to each tube. Cap tubes securely.
- Briefly centrifuge to collect contents at the bottom.
- Amplification: Place tubes in a preheated isothermal instrument (heating block, water bath, or real-time fluorometer) at 65°C for 25-40 minutes. Do not use a thermal cycler with a heated lid, as the constant temperature is critical.
- Visual Readout (if using HNB): Observe color change under natural light. A positive reaction shifts from violet to sky blue. The NTC should remain violet.
- Endpoint Analysis: Reactions can be analyzed by gel electrophoresis (2% agarose) showing a characteristic ladder-like pattern, or by measuring turbidity (Mg₂P₂O₇ precipitate) at 400 nm.
Visualizing the RT-LAMP Workflow and Mechanism
Diagram 1: RT-LAMP Workflow for SARS-CoV-2 Detection
Commercial Kits for SARS-CoV-2 LAMP
Numerous commercial kits provide optimized, pre-formulated master mixes, simplifying assay deployment. Key offerings are compared below.
Table 2: Commercial RT-LAMP Master Mix Kits for SARS-CoV-2 Research
| Kit Name (Supplier) | Key Components Included | Detection Method | Claimed Time | Notes for Research |
|---|---|---|---|---|
| WarmStart LAMP Kit (DNA & RNA) (NEB) | Bst 2.0 WarmStart, separate RT module, optimized buffer, dNTPs, MgSO₄. | Colorimetric (with dye), fluorescent, turbidimetric. | 15-60 min | Flexible; allows separate optimization of RT and LAMP steps. |
| Colorimetric RT-LAMP Master Mix (Thermo Fisher) | Bst polymerase with RT activity, dNTPs, Mg²⁺, betaine, phenol red indicator. | Colorimetric (Phenol Red: pink → yellow). | 20-40 min | Ready-to-use, single-tube format. pH-based detection. |
| Loopamp RNA Amplification Kit (Eiken Chemical) | Bst polymerase, reverse transcriptase, buffer, dNTPs, designed primer kits. | Turbidity (real-time or endpoint), fluorescent. | 15-40 min | Original LAMP technology licensor; often used with turbidimeter. |
| Genie III / Genie II Reagents (OptiGene) | Proprietary master mix with Bst polymerase and dsDNA binding dye. | Real-time fluorescent (on Genie devices). | <20 min | Optimized for high-speed, real-time monitoring on dedicated instruments. |
| Luna Universal RT-LAMP Kit (BioLabs) | WarmStart Bst polymerase, RTase, buffer, dNTPs, Mg²⁺, betaine. | Fluorescent (compatible with dyes). | 20-60 min | High sensitivity and robust performance with complex samples. |
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents and Materials for SARS-CoV-2 LAMP Research
| Item | Supplier Examples | Function in Research Context |
|---|---|---|
| Bst 2.0/3.0 WarmStart Polymerase | New England Biolabs (NEB) | High-activity, thermostable polymerase with strand-displacement; WarmStart enables room-temperature setup. |
| Thermostable Reverse Transcriptase (HIV-1 RT, M-MLV) | Sigma-Aldrich, Invitrogen | For two-step RT-LAMP or when optimizing RT and LAMP conditions independently. |
| dNTP Mix (100 mM each) | Thermo Fisher, NEB | High-purity, pH-neutral stock solutions for preparing custom master mixes. |
| Betaine Solution (5M) | Sigma-Aldrich | Additive for destabilizing secondary structures in GC-rich SARS-CoV-2 targets. |
| Hydroxy Naphthol Blue (HNB) | Sigma-Aldrich, Tokyo Chemical Industry | Metal indicator dye for inexpensive, visual endpoint detection of amplification. |
| SYTO 9 / EvaGreen Dye | Invitrogen, Biotium | Fluorogenic intercalating dyes for real-time monitoring of LAMP product formation. |
| RNase Inhibitor (Murine) | Promega, NEB | Critical for maintaining RNA template integrity during reaction setup. |
| Nuclease-Free Water & Tubes | Ambion, Axygen | Essential for preventing degradation of primers, templates, and enzymes. |
| Synthetic SARS-CoV-2 RNA Control | Twist Bioscience, ATCC | Validated, non-infectious positive control for assay development and optimization. |
Loop-mediated isothermal amplification (LAMP) has emerged as a powerful molecular technique for the detection of SARS-CoV-2, offering high sensitivity and specificity without the need for complex thermal cycling. Its isothermal nature (typically 60-65°C) decouples amplification from precise thermal control, enabling its deployment on diverse instrumentation platforms. This technical guide examines three critical platform categories—block heaters, portable devices, and smartphone-integrated systems—framed within the requirements of rigorous LAMP-based SARS-CoV-2 research and diagnostics. The choice of platform directly impacts assay throughput, portability, cost, and integration with detection modalities (e.g., fluorescence, turbidity, colorimetric pH change).
Platform Comparison: Technical Specifications and Performance Data
The following table summarizes the core characteristics of the three platform types based on current (2024-2025) commercial and research prototypes.
Table 1: Comparison of Instrumentation Platforms for SARS-CoV-2 LAMP Assays
| Feature | Traditional Block Heater | Portable Dedicated Device | Smartphone-Integrated System |
|---|---|---|---|
| Primary Use Case | High-throughput, batch processing in central labs. | Point-of-Care Testing (POCT), field deployment. | Ultra-portable, low-cost POCT, resource-limited settings. |
| Temperature Uniformity | High (±0.1°C to ±0.5°C). | Moderate to High (±0.2°C to ±0.8°C). | Variable, often lower (±0.5°C to ±2.0°C). |
| Detection Method | Typically requires separate device (e.g., fluorimeter, spectrophotometer). | Integrated optical sensors (fluorescence, turbidity). | Smartphone camera (colorimetric, fluorescence via add-ons). |
| Result Time | 20-40 minutes (amplification only). | 15-30 minutes (integrated amplification & analysis). | 20-35 minutes (integrated). |
| Approx. Cost per Unit | $200 - $2,000. | $1,000 - $5,000. | $50 - $500 (excluding phone). |
| Key Advantage | Excellent reproducibility, high sample capacity. | Robust, all-in-one solution for field use. | Extreme portability, connectivity, low cost. |
| Key Limitation | Lack of integrated detection, low portability. | Higher cost than basic heaters, limited scalability. | Thermal/optical performance depends on accessory design. |
| Example Models/Concepts | Dry bath incubators, basic thermal blocks. | BioFire FilmArray, Sherlock Biosciences kit, QuantuMDx Q-POC. | 3D-printed heater with phone cradle, commercial attachments (e.g., DeNovix CellDrop). |
Detailed Experimental Protocols
Protocol 1: Colorimetric LAMP on a Standard Block Heater with Endpoint Analysis This protocol is adapted for SARS-CoV-2 ORF1a gene detection.
- Reaction Setup: Prepare a 25 µL LAMP master mix containing: 1.6 µM each inner primer (FIP/BIP), 0.2 µM each outer primer (F3/B3), 0.4 µM each loop primer (LF/LB), 1x Isothermal Amplification Buffer, 6 mM MgSO₄, 1.4 mM each dNTP, 0.8 M betaine, 120 µM phenol red, 8 U Bst 2.0 or 3.0 DNA Polymerase, and 5 µL of extracted RNA or viral transport media.
- Amplification: Aliquot the mix into 0.2 mL PCR tubes. Place tubes in a pre-heated block heater at 65°C for 30 minutes.
- Inactivation & Visualization: Heat samples at 80°C for 2 minutes to stop the reaction. Visually inspect the color change: positive (original red/orange → yellow due to acidification) vs. negative (remains red/orange). For quantification, measure absorbance at 560 nm and 430 nm using a plate reader.
Protocol 2: Real-time Fluorescence LAMP on a Portable Integrated Device This protocol uses a portable device with integrated heating and fluorescence detection (e.g., Genie III, OptiGene).
- Loading: Prepare a similar master mix as in Protocol 1, but replace phenol red with a DNA intercalating dye (e.g., 1x SYTO-9, 0.5x EvaGreen) or use primer sets labeled with FITC/Cal Fluor 610 for multiplexing.
- Run Setup: Load the reaction mix into the device-specific cartridge or strip. Select the appropriate protocol on the touchscreen: 65°C for 25-30 minutes with fluorescence acquisition every 30 seconds.
- Analysis: The device software automatically generates amplification curves, determines threshold time (Tt), and provides a positive/negative call based on user-defined fluorescence thresholds. Data can be exported via USB.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for SARS-CoV-2 LAMP Research
| Reagent/Material | Function | Example Vendor/Product |
|---|---|---|
| Bst 2.0/3.0 DNA Polymerase | Strand-displacing DNA polymerase for isothermal amplification. | New England Biolabs, WarmStart LAMP kits. |
| LAMP Primer Sets | Target-specific primers (F3/B3, FIP/BIP, LF/LB) for SARS-CoV-2 genes (N, E, ORF1ab). | Integrated DNA Technologies (IDT), custom designs. |
| WarmStart Technology | Enzyme inactivation at room temperature, enabling ambient setup and hot-start. | New England Biolabs. |
| Colorimetric pH Indicator | Visual endpoint detection (Phenol Red, Hydroxy Naphthol Blue). | Sigma-Aldrich, included in many commercial kits. |
| Fluorescent DNA Dye | Real-time monitoring of amplification (SYTO-9, EvaGreen). | Thermo Fisher Scientific, Biotium. |
| RNase Inhibitor | Critical for direct assays using RNA without reverse transcription step in RT-LAMP. | Promega, Thermo Fisher Scientific. |
| Positive Control Plasmid | Synthetic DNA containing the target SARS-CoV-2 sequence for assay validation. | BEI Resources, Twist Bioscience. |
Visualizing Workflows and Logical Relationships
Platform Selection Logic for LAMP Assays
Smartphone-Integrated LAMP Detection Workflow
This whitepaper details three principal detection modalities for Loop-Mediated Isothermal Amplification (LAMP) assays, framed within ongoing doctoral research on optimizing SARS-CoV-2 detection. While the core LAMP amplification principles—targeting genes such as ORF1a, N, or E—are consistent, the endpoint detection strategy critically influences assay accessibility, throughput, cost, and suitability for point-of-care (POC) deployment. This guide provides a technical comparison, standardized protocols, and visual frameworks for implementing real-time fluorescence, colorimetric (pH), and lateral flow dipstick (LFD) detection in a research setting.
Comparative Analysis of Detection Modalities
The following table summarizes the quantitative and qualitative characteristics of each modality, essential for selecting the appropriate method based on research or diagnostic objectives.
Table 1: Comparative Analysis of LAMP Detection Modalities for SARS-CoV-2
| Feature | Real-Time Fluorescence | Colorimetric (pH) | Lateral Flow Dipstick (LFD) |
|---|---|---|---|
| Detection Principle | Intercalating dyes (e.g., SYTO-9) or sequence-specific probes (e.g., FITC/HEX, Quencher) produce fluorescent signal proportional to amplicon yield. | pH-sensitive dyes (e.g., phenol red) respond to proton release during DNA synthesis (dNTP hydrolysis). | Hybridization of biotin- and FAM-labeled amplicons with immobilized anti-FAM antibodies; streptavidin-gold nanoparticle conjugation provides visual line. |
| Primary Output | Real-time amplification curves; Cycle threshold (Ct) or time threshold (Tt). | Visible color change: Positive (yellow) Negative (pink/red). | Visible line at test (T) and control (C) zones. |
| Typical Time-to-Result | 15-45 minutes (real-time monitoring). | 30-60 minutes (endpoint, post-amplification). | 60-75 minutes (includes 60 min amplification + 5-15 min dipstick flow). |
| Approx. Limit of Detection (LoD) | 10-100 copies/µL (highest sensitivity). | 100-1000 copies/µL. | 50-500 copies/µL. |
| Equipment Required | Real-time fluorometer or isothermal thermal cycler with fluorescence detection. | Simple heat block or water bath; visual or spectrophotometric readout. | Heat block/water bath; no reader needed (visual). |
| Quantitative Potential | Yes (semi-quantitative/quantitative via standard curve). | No (qualitative, though spectrophotometric OD can be used). | No (qualitative). |
| Throughput | High (multi-well plates). | Moderate to High (tube/plate). | Low to Moderate (individual dipsticks). |
| Key Advantage | Sensitive, quantitative, closed-tube reduces contamination. | Simplicity, low cost, equipment-free visual readout. | Specificity (probe-based), equipment-free, multiplex potential via different tags. |
| Primary Limitation | Higher cost, requires specialized equipment. | Subjectivity in color interpretation, buffer sensitivity. | Additional post-amplification handling increases contamination risk. |
| Best Suited For | High-throughput lab-based validation & quantification. | Rapid field or resource-limited screening. | POC confirmation requiring high specificity. |
Detailed Experimental Protocols
General LAMP Master Mix (Base Recipe for 25µL Reaction):
- Isothermal Buffer: 1.4 mM each dNTP, 6 mM MgSO₄, 20 mM Tris-HCl (pH 8.8), 10 mM KCl, 10 mM (NH₄)₂SO₄, 0.1% Tween 20.
- Enzyme Mix: 8 U Bst 2.0 or 3.0 DNA Polymerase (large fragment).
- Primers: 1.6 µM each FIP/BIP, 0.2 µM each F3/B3, 0.4 µM each LF/LB (if used).
- Template: 2-5 µL of extracted RNA or viral transport medium (heat-inactivated at 95°C for 5 min).
- Incubation: 63-65°C for 25-45 minutes.
Protocol 3.1: Real-Time Fluorescence LAMP using Intercalating Dye
- Reagent Setup: To the general master mix, add SYTO-9 fluorescent dye at a final concentration of 1-2.5 µM.
- Loading: Aliquot 25 µL of the final master mix into each well of a real-time PCR plate or thin-walled tube. Add template.
- Run Parameters: Load into a real-time isothermal fluorometer (or qPCR instrument with isothermal setting). Set conditions: 63°C for 40 minutes, with fluorescence acquisition (FAM/SYBR Green channel) every 30-60 seconds.
- Analysis: Determine the time threshold (Tt) where the fluorescence curve crosses a baseline threshold. Compare to standard curves for quantification.
Protocol 3.2: Colorimetric pH-Sensitive LAMP
- Reagent Setup: Prepare the general master mix using a commercial colorimetric LAMP buffer pre-supplemented with phenol red (or add phenol red to ~0.1 mM final concentration). Critical: Avoid use of high-concentration Tris buffers post-amplification, as they can neutralize the pH change.
- Amplification: Dispense 25 µL reactions into 0.2 mL PCR tubes. Incubate in a standard heat block or water bath at 63°C for 45 minutes.
- Visual Readout: Observe immediate color change post-amplification. Positive: Yellow (acidic pH ~6.8). Negative: Pink/Red/Magenta (basic pH ~8.4).
- Optional Quantitative Readout: Measure absorbance at 430 nm and 560 nm using a plate reader. The ratio (A₄₃₀/A₅₆₀) correlates with amplification.
Protocol 3.3: Lateral Flow Dipstick (LFD) Detection
- LAMP with Modified Primers: Perform LAMP amplification using a set of primers where the FIP primer is 5'-labeled with Biotin and the LF or LB primer is 5'-labeled with Fluorescein (FAM).
- Post-Amplification Hybridization: After 60 min amplification at 63°C, dilute 5 µL of the amplicon product with 95 µL of the provided assay buffer.
- Dipstick Development: Insert the lateral flow strip (pre-coated with an anti-FAM antibody at the Test line 'T' and streptavidin at the Control line 'C') into the diluted amplicon. Allow capillary flow for 5-15 minutes.
- Visual Readout: Positive: Both Control (C) and Test (T) lines appear. Negative: Only the Control (C) line appears. Invalid: No C line (repeat required). The FAM label binds anti-FAM at T, while biotin binds streptavidin-gold conjugate; the conjugate is captured at C as an internal control.
Visualization of Workflows and Principles
Diagram 1: Unified LAMP assay workflow with three detection paths.
Diagram 2: Signal generation in colorimetric LAMP assays.
Diagram 3: Lateral flow dipstick detection mechanism.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Materials for LAMP Detection Modalities
| Reagent/Material | Function & Description | Typical Vendor/Example |
|---|---|---|
| Bst 2.0/3.0 DNA Polymerase | Strand-displacing DNA polymerase essential for isothermal amplification. Bst 3.0 offers faster kinetics. | New England Biolabs (NEB), WarmStart by Merck. |
| Colorimetric LAMP Master Mix | Optimized buffer with pH indicator (phenol red), dNTPs, Mg²⁺, and stabilizers. | NEB WarmStart Colorimetric, OptiGene, Lucigen. |
| Fluorescent DNA Intercalating Dye (SYTO-9) | Cell-permeant dye that binds dsDNA, used for real-time fluorescence detection. Low inhibition of Bst polymerase. | Thermo Fisher Scientific. |
| Biotin & FAM-labeled Primers | Modified primers for generating labeled amplicons compatible with lateral flow detection. | IDT, Eurofins Genomics (HPLC purified). |
| Lateral Flow Dipsticks (Anti-FAM) | Pre-fabricated nitrocellulose strips with anti-FAM (Test) and streptavidin/biotin (Control) lines. | Milenia HybriDetect, Ustar Biotechnologies. |
| Isothermal Fluorometer | Instrument for real-time fluorescence monitoring at constant temperature. | QuantStudio 5, Bio-Rad CFX96 (with isothermal app), Genie II. |
| Heat Block (Digital, Fine Control) | Provides precise, uniform incubation at 63-65°C for non-real-time assays. | Thermo Scientific, VWR. |
| RNase/DNase Inactivation Tube | For safe, direct heat-inactivation of viral transport media prior to LAMP. | Zymo Research DNA/RNA Shield. |
Loop-mediated isothermal amplification (LAMP) has emerged as a robust, rapid, and field-deployable alternative to RT-PCR for detecting SARS-CoV-2 RNA. This whitepaper, framed within broader thesis research on LAMP principles, provides an in-depth technical guide to interpreting the two primary data outputs from LAMP assays: real-time amplification curves and endpoint results. Accurate interpretation is critical for determining viral load, assay sensitivity, specificity, and ultimately, diagnostic accuracy.
Fundamental Principles of LAMP Data Generation
LAMP amplifies target DNA isothermally (typically 60-65°C) using a strand-displacing DNA polymerase and 4-6 primers recognizing distinct regions of the target. Data is generated via the detection of amplification byproducts:
- Real-time monitoring: Uses intercalating dyes (e.g., SYTO-9, EvaGreen) or fluorophore-quencher probes to measure fluorescence increase over time.
- Endpoint detection: Visual assessment of turbidity (magnesium pyrophosphate precipitate) or color change (pH indicators like phenol red or metal ion indicators like hydroxynaphthol blue).
Quantitative Analysis of Real-Time Amplification Curves
The amplification curve plots fluorescence (ΔRn) against cycle number or time. Key parameters are extracted for quantitative analysis.
Key Quantitative Parameters
Table 1: Key Parameters for Amplification Curve Analysis
| Parameter | Definition | Interpretation in SARS-CoV-2 LAMP |
|---|---|---|
| Threshold | Fluorescence level significantly above baseline noise. Set manually or algorithmically. | Defines the limit of detection; must be consistent across runs. |
| Time to Positivity (TtP) | Time (min) at which the curve crosses the threshold. Also called Time Threshold (Tt). | Inversely proportional to initial template concentration. Primary metric for quantification. |
| Slope (Efficiency) | Steepness of the exponential phase curve. | Reflects amplification efficiency. Optimal ~0.1 min⁻¹ (for time-based plots). Low slope may indicate inhibitors. |
| Baseline Fluorescence | Initial, flat phase of the curve before amplification. | Varies with master mix, dye, and instrument. High noise can obscure low-copy targets. |
| Plateau Phase | Final phase where fluorescence stabilizes. | Height can be affected by amplicon concentration, dye saturation, and reagent limitation. |
Standard Curve Construction and Quantification
To estimate viral load, a standard curve is generated using serial dilutions of synthetic SARS-CoV-2 RNA or quantified positive control.
Table 2: Example Standard Curve Data from SARS-CoV-2 ORF1ab Gene LAMP
| Standard Copy Number (log10) | Mean TtP (minutes) | Standard Deviation (min) |
|---|---|---|
| 10^6 | 8.2 | 0.3 |
| 10^5 | 12.1 | 0.5 |
| 10^4 | 16.5 | 0.6 |
| 10^3 | 21.8 | 0.9 |
| 10^2 | 28.4 | 1.5 |
| NTC | Undetermined | - |
NTC: No Template Control.
The linear regression of Log10(Starting Quantity) vs. TtP yields the equation: y = -3.32 * x + c, where the slope (-3.32) represents 100% efficiency (10-fold decrease in copies for every ~3.32 min increase in TtP).
Experimental Protocol: Real-Time LAMP for SARS-CoV-2
Title: One-Step RT-LAMP for SARS-CoV-2 Detection (Real-Time).
- RNA Extraction/Purification: Use magnetic bead-based or column-based methods from nasopharyngeal/swab samples. Include extraction controls.
- Master Mix Preparation (25 µL reaction):
- 1.4 µL Primer Mix (F3/B3: 0.2 µM each; FIP/BIP: 1.6 µM each; LF/LB: 0.8 µM each).
- 12.5 µL 2x LAMP Buffer (Isothermal buffer with dNTPs, MgSO4).
- 1.0 µL Enzyme Mix (WarmStart Bst 2.0/3.0 DNA Polymerase + RTx Reverse Transcriptase).
- 1.0 µL Fluorescent Dye (20x SYTO-9 or equivalent).
- Nuclease-free water to 23 µL.
- Template Addition: Add 2 µL of extracted RNA (or standard/control).
- Amplification: Run on a real-time isothermal fluorometer or thermocycler with isothermal hold.
- Conditions: 63°C for 40 minutes, with fluorescence acquisition every 30 seconds.
- Data Analysis: Software determines TtP for each sample. Compare to standard curve for quantification.
Diagram Title: Real-Time RT-LAMP Workflow for SARS-CoV-2 Detection
Interpretation of Endpoint Results
Endpoint analysis provides a binary (positive/negative) result, often visualized by color or turbidity change.
Quantitative Aspects of Endpoint Detection
While qualitative, the time-to-visual-positivity and intensity of change can offer semi-quantitative insights.
Table 3: Interpretation of Visual Endpoint Results
| Result Type | Colorimetric (Phenol Red) | Turbidity (Pyrophosphate) | Fluorogenic (Calcein/HNB) | Possible Interpretation |
|---|---|---|---|---|
| Strong Positive | Yellow (from pink) | Pronounced white precipitate | Green fluorescence (Calcein) / Sky blue (HNB) | High viral load (>10^4 copies/µL). |
| Weak Positive | Pale orange/yellow | Faint haze | Dull green / Light blue | Low viral load, near assay limit. |
| Negative | Remains pink | Clear solution | Orange (Calcein) / Violet (HNB) | No target detected. |
| Invalid | Extreme yellow (acidic) or deep pink (basic) | N/A | N/A | Master mix pH compromised. |
Experimental Protocol: Endpoint Colorimetric LAMP
Title: Endpoint Colorimetric RT-LAMP for SARS-CoV-2.
- Reagent Setup: Prepare a master mix as in Section 3.3, but substitute fluorescent dye with 1.5 µL of 1.2 mM phenol red (or 120 µM HNB).
- Amplification: Incubate in a dry bath/block heater at 63°C for 40-60 minutes. No real-time monitoring required.
- Visual Assessment: Place tubes against a white background. A color change from pink to yellow (phenol red, due to pH drop from proton release during amplification) indicates a positive result.
- Confirmation (Optional): Analyze reaction products via gel electrophoresis (1.5% agarose) to confirm the characteristic ladder pattern of LAMP amplicons.
Diagram Title: Endpoint Result Interpretation Pathways for LAMP
Critical Factors Influencing Data Interpretation
- Inhibition: Sample contaminants can delay TtP, cause atypical curve shapes, or cause false negatives. Use internal controls (e.g., human RNase P gene).
- Primer-Dimer/Nonspecific Amplification: Can cause false-positive endpoint signals or early rising curves with low plateau. Optimization of primer design and reaction temperature is key.
- Signal Threshold Setting: Critical for reproducible TtP. Should be set in the exponential phase for all standards, typically 3-5 standard deviations above the mean baseline fluorescence.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Materials for SARS-CoV-2 LAMP Research & Development
| Item | Function & Rationale | Example/Note |
|---|---|---|
| Bst 2.0/3.0 DNA Polymerase | Strand-displacing polymerase for isothermal amplification. High displacement activity and robustness to inhibitors. | WarmStart versions prevent pre-amplification activity. |
| Reverse Transcriptase (RTx) | For one-step RT-LAMP, converts SARS-CoV-2 RNA to cDNA. | Must be compatible/isothermally active (~60-65°C). |
| LAMP Primer Sets | Target 6-8 distinct regions of SARS-CoV-2 genome (e.g., N, E, ORF1ab genes). Specificity is paramount. | Validated, published sets (e.g., Zhang et al. 2020). Aliquot to avoid freeze-thaw. |
| Isothermal Amplification Buffer | Provides optimal pH, salt, betaine (to lower DNA melting temp), and Mg²⁺ (cofactor for polymerase). | Often supplied as a 2x concentrate with dNTPs. |
| Fluorescent Intercalating Dye (SYTO-9) | Binds dsDNA amplicons, enabling real-time monitoring. Low inhibition of LAMP. | Prefer dyes that do not inhibit amplification (vs. SYBR Green I). |
| Colorimetric pH Indicator (Phenol Red) | Visual endpoint detection. Amplification releases protons, lowering pH, changing color. | Concentration must be optimized to avoid inhibition. |
| Synthetic SARS-CoV-2 RNA Control | Quantitative standard for constructing standard curves and determining LoD. | Non-infectious, encompassing full primer target regions. |
| Human RNA Control (e.g., RNase P) | Nucleic acid extraction and amplification internal control. Distinguishes true negative from inhibition. | Co-amplified in multiplex or in a separate well. |
| Uracil DNA Glycosylase (UDG/UNG) | Carryover contamination prevention. Degrades amplicons containing dUTP from previous runs. | Optional but recommended for high-throughput settings. |
Troubleshooting LAMP Assays: Solving Sensitivity, Specificity, and Inhibition Challenges
Loop-mediated isothermal amplification (LAMP) has emerged as a pivotal molecular diagnostic tool for the detection of SARS-CoV-2, offering advantages in speed, sensitivity, and field-deployability over traditional RT-PCR. However, the integrity of results is critically threatened by two major sources of false positives: non-specific amplification from primer-dimer artifacts and contamination from amplicon carryover. Within the broader thesis on optimizing LAMP assay principles for SARS-CoV-2, understanding, identifying, and mitigating these pitfalls is fundamental to ensuring diagnostic reliability and regulatory acceptance.
Primer-Dimer Formation: Mechanisms and Impact
Primer-dimer (PD) occurs when primers anneal to each other via complementary sequences, particularly at their 3' ends, rather than to the target template. This self-annealing creates a short duplex that can be extended by the DNA polymerase, leading to non-specific amplification that consumes reagents and generates false-positive fluorescence signals.
Key Factors in LAMP Assays:
- High Primer Concentration: LAMP utilizes 4-6 primers at high concentrations (typically 0.2-1.6 µM each), increasing the probability of intermolecular interactions.
- Isothermal Conditions: Constant temperature (60-65°C) may favor transient primer interactions.
- Complex Primer Set: The FIP and BIP inner primers are long (∼40-45 nt), increasing the chance of partial inter-primer complementarity.
Quantitative Impact of Primer-Dimer: A summary of studies analyzing PD formation in SARS-CoV-2 LAMP assays.
Table 1: Quantified Effects of Primer-Dimer Formation
| Parameter | Value Range (from cited studies) | Impact on Assay |
|---|---|---|
| Early Amplification Time | Can appear <10 minutes | Mimics true positive, leading to false calls. |
| Final Fluorescence Intensity | Up to 50% of positive signal | Exceeds typical negative threshold. |
| Reagent Consumption | dNTPs depleted by 15-30% | Reduces sensitivity for true low-copy targets. |
| Gel Electrophoresis Band | Low molecular weight smear (~50-100 bp) | Distinguishes from true LAMP ladder pattern. |
Experimental Protocol: Detection and Confirmation of Primer-Dimer
Aim: To distinguish primer-dimer artifacts from specific SARS-CoV-2 amplicons. Method:
- No-Template Control (NTC) Setup: Perform the LAMP reaction (as per protocol, e.g., WarmStart LAMP Kit) in the absence of any SARS-CoV-2 RNA template. Use standard primer sets (e.g., targeting N or ORF1a gene).
- Real-time Monitoring: Run the reaction in a real-time fluorometer for 60 minutes at 65°C.
- Post-Amplification Analysis: a. Gel Electrophoresis: Run 5 µL of the product on a 2% agarose gel. True LAMP yields a characteristic ladder pattern; PD appears as a low molecular weight band or smear. b. Melting Curve Analysis: After amplification, perform a slow melt from 65°C to 95°C. PD products typically have a lower, broader melting temperature (Tm) than specific, high-molecular-weight LAMP products. c. Sequencing: Purify the NTC amplicon and sequence using Sanger methods. PD sequences will not align to the SARS-CoV-2 genome.
Carryover Contamination: The Amplicon Menace
Carryover contamination involves the introduction of previously amplified DNA (amplicons) into new reaction setups, leading to catastrophic false-positive rates. LAMP is exceptionally vulnerable due to its high yield (often >10^9 copies per reaction) and aerosol generation during tube opening.
Primary Vectors:
- Aerosols: Generated during post-amplification handling.
- Contaminated Pipettes and Surfaces.
- Reagent or Master Mix contamination.
Experimental Protocol: Contamination Control and Decontamination
Aim: To implement and test a dUTP-UNG (uracil-N-glycosylase) system to prevent carryover contamination in SARS-CoV-2 RT-LAMP. Method:
- Reagent Modification: Prepare LAMP master mix substituting dTTP with dUTP. Use a polymerase compatible with dUTP (e.g., Bst 2.0/3.0 DNA Polymerase).
- Incorporation of UNG: Include heat-labile Uracil-N-Glycosylase (UNG) in the master mix (e.g., 0.1 U/µL).
- Workflow: a. Pre-incubation Step: Hold reactions at 25°C for 5-10 minutes before isothermal amplification. UNG will hydrolyze any contaminating uracil-containing amplicons from previous runs. b. Inactivation: The UNG is permanently inactivated during the subsequent 65°C LAMP step, protecting newly synthesized dUTP-containing amplicons. c. Validation: Spike new reactions with 10^6 copies of pre-amplified dUTP-containing SARS-CoV-2 LAMP product. Compare amplification in +/- UNG conditions. Effective decontamination will show no amplification in the +UNG condition.
Table 2: Efficacy of dUTP-UNG System in Carryover Prevention
| Contaminating Amplicon Level | Without UNG (Time to Positive) | With UNG Pre-treatment (Result) | % False Positive Reduction |
|---|---|---|---|
| High (10^6 copies) | < 15 min | No amplification for 60 min | 100% |
| Low (10^3 copies) | 20-30 min | No amplification for 60 min | 100% |
| Trace (10^1 copies) | Variable, late | No amplification for 60 min | ~100% |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Mitigating False Positives in SARS-CoV-2 LAMP
| Item | Function/Description | Example Product |
|---|---|---|
| Hot-Start DNA Polymerase | Reduces non-specific primer extension during reaction setup by requiring thermal activation. | WarmStart Bst 2.0/3.0 |
| Dye-based Intercalators | Real-time monitoring of amplification; some show reduced propensity to stain primer-dimer. | SYTO-9, EvaGreen |
| dUTP / UNG System | Enzymatic destruction of prior amplicons to prevent carryover contamination. | Thermolabile UNG, dUTP mix |
| Probe-based Detection | Increases specificity by requiring hybridization of an internal oligo, reducing PD signal. | FQ (FITC-Quencher) probes, LFIA probes |
| Primer Design Software | Identifies regions of inter-primer complementarity to minimize dimerization risk. | PrimerExplorer V5, NUPACK |
| Dedicated Post-PCR Tubes | Containers with seals to minimize aerosol release upon opening. | PCRGuard tubes |
| Surface Decontaminant | Inactivates DNA/RNA on lab surfaces and equipment. | DNA-OFF, 10% Bleach |
Mitigation Strategies and Best Practices
For Primer-Dimer:
- In Silico Design: Rigorously check 3' complementarity of all primer pairs using software.
- Optimize Chemistry: Use hot-start polymerases and optimize Mg2+ and primer concentrations.
- Probe-based Detection: Implement loop primers with 5' fluorophore/quencher for sequence-specific signal.
- Secondary Verification: Establish a post-amplification melting curve or gel electrophoresis step for confirmatory testing.
For Carryover:
- Physical Separation: Maintain separate, dedicated pre- and post-amplification areas with unidirectional workflow.
- UNG-dUTP System: Implement as a standard protocol component.
- Closed-tube Detection: Use systems with sealed reaction vessels (e.g., microfluidic chips, lyophilized pellets in capped tubes).
- Rigorous Decontamination: Use dedicated equipment, UV irradiation workspaces, and chemical decontaminants.
The diagnostic utility of SARS-CoV-2 LAMP assays is contingent upon robust specificity. Primer-dimer artifacts and amplicon carryover contamination represent two persistent technical challenges that can severely compromise assay reliability through false-positive results. Through meticulous primer design, the adoption of enzymatic containment strategies like UNG-dUTP, and strict adherence to spatially segregated workflows, these pitfalls can be effectively mitigated. Integrating these controls is essential for translating LAMP from a research tool into a clinically validated diagnostic platform.
Visualizations
Title: SARS-CoV-2 LAMP Workflow with Contamination Controls
Title: Pathways to False Positives in LAMP Assays
Optimizing Primer Sets for Emerging Variants (Omicron Lineages)
This technical guide addresses the critical challenge of maintaining Loop-Mediated Isothermal Amplification (LAMP) assay efficacy in the face of rapidly evolving SARS-CoV-2 Omicron lineages. Framed within a broader thesis on LAMP assay design principles, this document provides a structured, data-driven approach to primer redesign and validation, ensuring robust detection of emerging variants.
The diagnostic performance of nucleic acid amplification tests, including LAMP, is contingent on the complementarity of primer sequences to the target genome. The hyper-mutability of SARS-CoV-2, particularly within the spike (S), nucleocapsid (N), and ORF1ab genes targeted by many assays, necessitates continuous primer re-evaluation. This guide details a systematic workflow for in silico analysis, in vitro validation, and optimization of primer sets against prevailing Omicron sublineages (e.g., BA.2, BA.4/BA.5, BQ.1, XBB, and their descendants), ensuring the reliability of LAMP assays for research and surveillance.
Section 1:In SilicoPrimer Mismatch Analysis
Methodology
- Data Source: Retrieve the most recent, high-quality, full-genome sequences for target Omicron lineages from repositories such as GISAID and NCBI Virus. Use a consensus sequence for each lineage.
- Alignment & Analysis: Perform multiple sequence alignment (e.g., using MAFFT or Clustal Omega) of the consensus sequences against the reference Wuhan-Hu-1 genome (NC_045512.2). Map existing LAMP primer (F3, B3, FIP, BIP, LF, LB) binding regions onto the alignments.
- Mismatch Scoring: Manually or programmatically identify nucleotide mismatches within the last 5 bases of the 3' ends of all primers, as these are most critical for amplification efficiency. Score mismatches in internal sequences for their potential to reduce annealing kinetics.
Table 1: Prevalence of Critical Mutations in Omicron Sublineages (Representative Data)
| Gene Target | Primer Binding Region (approx.) | Omicron BA.2 | Omicron BA.5 | Omicron BQ.1.1 | Omicron XBB.1.5 | Potential Impact on LAMP |
|---|---|---|---|---|---|---|
| N Gene | 28388-28400 | S2L (rare) | S2L | S2L | S2L | Low (if in F3/B3) |
| N Gene | 28681-28701 | P13L, E31- | P13L, E31- | P13L, E31- | P13L, E31-, S33F | Medium (if in loop primer) |
| ORF1ab | 11588-11610 | T842I | T842I | T842I | T842I | Low |
| S Gene | 22673-22690 | V213G | V213G | V213G | V213G | Medium-High (if 3' end mismatch) |
| E Gene | 26270-26290 | T9I | T9I | T9I | T9I | Low |
Note: Nucleotide positions relative to Wuhan-Hu-1 (NC_045512.2). Mutations are listed as amino acid changes; underlying nucleotide changes must be assessed. This table is illustrative; a current analysis must be performed.
Section 2: Experimental Validation Protocol
Protocol: Primer Efficacy Testing via Real-Time Fluorescence LAMP
Objective: Compare amplification efficiency and time-to-positive (TTP) of original versus optimized primer sets.
Materials:
- Template: Synthetic RNA controls (e.g., Twist Biosciences) encoding the full SARS-CoV-2 genome for relevant Omicron lineages and wild-type.
- Primers: Original and candidate optimized primer sets (desalted purity).
- Master Mix: WarmStart Colorimetric or Fluorescent LAMP 2X Master Mix (NEB).
- Equipment: Real-time fluorometer or thermocycler with fluorescence detection capable of maintaining 65°C.
Procedure:
- Reaction Setup: Prepare 25 µL reactions containing 1X LAMP Master Mix, 1.6 µM each of FIP and BIP, 0.2 µM each of F3 and B3, 0.4 µM each of LF and LB (if used), and 5 µL of template RNA (10^3 copies/µL). Include no-template controls (NTC).
- Amplification: Incubate at 65°C for 60 minutes, with fluorescence (SYTO-9 or similar) measured every 60 seconds.
- Data Analysis: Determine TTP (time at which fluorescence crosses a predetermined threshold) for each primer set/template combination. Calculate the ΔTTP (TTPvariant - TTPWT) for each primer set. A ΔTTP > 5 minutes indicates significant loss of sensitivity.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function |
|---|---|
| Synthetic RNA Controls (Omicron-specific) | Provides a consistent, non-infectious template for validating assay performance against specific variants. |
| WarmStart LAMP Master Mix | Contains Bst 2.0/3.0 polymerase with strand-displacing activity, optimized buffer, and dNTPs. "WarmStart" prevents non-specific amplification at room temperature. |
| SYTO-9 Green Fluorescent Stain | Intercalating DNA dye for real-time monitoring of amplification. Preferable to hydroxynaphthol blue for quantitative kinetics. |
| Primers (HPLC-purified) | High-purity primers ensure correct sequence and reduce failure due to truncated oligonucleotides, critical for complex primer mixes. |
| RNase Inhibitor | Protects RNA template from degradation during reaction setup, improving reproducibility. |
Protocol: Specificity Testing with Cross-Reactive Panels
Objective: Confirm optimized primers do not amplify non-target human coronaviruses or human genomic DNA.
Procedure:
- Panel Creation: Assemble RNA/DNA from related viruses (e.g., HCoV-OC43, HCoV-229E, MERS-CoV) and high-concentration human genomic DNA (e.g., from HEK293 cells).
- LAMP Reaction: Perform the LAMP assay as in Section 2.1, using 10^4 copies of each non-target nucleic acid as template.
- Analysis: Run reactions for 90 minutes. A valid primer set should show no amplification (flat fluorescence curve) for all non-targets, while robustly amplifying the positive SARS-CoV-2 control.
Section 3: Optimization Strategy & Decision Logic
Based on in silico and experimental data, follow the decision pathway below to optimize your primer set.
Diagram Title: LAMP Primer Optimization Decision Logic for Variants
Section 4: Advanced Considerations: Multiplexing for Variant Discrimination
For research applications, LAMP can be multiplexed to not only detect SARS-CoV-2 but also identify specific variant-defining mutations.
Workflow: Allele-Specific LAMP for K417N Mutation Detection
Principle: Design a Forward Inner Primer (FIP) where the 3' terminal nucleotide is complementary to the mutant (e.g., A for K417N) but mismatched to the wild-type (T). Under optimized stringent conditions, amplification proceeds only in the presence of the mutant allele.
Protocol:
- Primer Design: Create two FIP primers identical except for the final 3' base: one "wild-type specific" (ending in T) and one "mutant specific" (ending in A).
- Dye Selection: Use two distinct fluorophores (e.g., FAM for WT channel, HEX for Mutant channel) in separate reactions or with quencher-based probes in a single tube.
- Stringent Condition Optimization: Titrate MgSO4 concentration (from 2 mM to 8 mM) and reaction temperature (from 63°C to 68°C) to find conditions that maximally discriminate between matched and mismatched templates.
- Validation: Test with well-characterized WT and mutant (e.g., Omicron) RNA controls.
Diagram Title: Allele-Specific LAMP Principle for SNP Detection
Sustaining LAMP assay effectiveness against emerging SARS-CoV-2 variants requires a proactive, iterative pipeline combining computational mismatch analysis with rigorous wet-lab validation. By adhering to the structured protocols and decision frameworks outlined in this guide, researchers can systematically optimize primer sets, ensuring their diagnostic and research tools remain resilient in the face of viral evolution. This work underscores the core thesis that robust LAMP design is not static but must evolve in parallel with the pathogen itself.
Loop-mediated isothermal amplification (LAMP) has emerged as a robust, rapid, and field-deployable nucleic acid amplification test (NAAT) for the detection of SARS-CoV-2. Its isothermal nature eliminates the need for thermal cyclers, making it suitable for point-of-care and resource-limited settings. However, a persistent challenge in applying LAMP, particularly with complex clinical samples like nasopharyngeal swabs or saliva, is the presence of inhibitors. These substances—including mucins, hemoglobin, IgG, and salts—can co-purify with viral RNA, adversely affecting the activity of the DNA polymerase, leading to false-negative results. This technical guide explores two synergistic strategies to overcome this inhibition: the use of engineered variants of Bacillus stearothermophilus (Bst) DNA polymerase and strategic sample dilution protocols.
Bst Polymerase Variants: Enhanced Resistance to Inhibitors
Wild-type Bst polymerase, the workhorse enzyme for LAMP, is susceptible to various inhibitors. Protein engineering has yielded variants with improved performance.
Key Variants and Their Properties
| Polymerase Variant | Key Mutation(s)/Feature | Primary Benefit | Reported Tolerance Improvement vs. WT |
|---|---|---|---|
| Bst 2.0/3.0 | Mutations in the thumb/palm subdomains | Enhanced processivity & speed | ~2-4x in crude samples (e.g., saliva) |
| Bst Large Fragment | N-terminal 5'→3' exonuclease domain deletion | Reduced non-specific amplification | Improved clarity in high-background samples |
| Bst DNA Polymerase, Iso | Proprietary mutations for isothermal use | High stability at 60-65°C | Up to 5x tolerance to blood components |
| Bst Polymerase exo- | Removal of 5'→3' exonuclease activity | Higher fidelity for specific target detection | Better performance with inhibitors like humic acid |
| Bst WarmStart | Aptamer-based hot-start | Suppresses primer-dimer formation | Enables higher sample input volume without inhibition |
Experimental Protocol: Evaluating Polymerase Variant Inhibition Resistance
Objective: To compare the inhibition resistance of different Bst polymerase variants using spiked clinical samples.
Materials:
- Purified SARS-CoV-2 RNA (or synthetic N gene transcript).
- Tested Bst polymerase variants (e.g., WT, 2.0, 3.0, WarmStart).
- Commercial LAMP master mix (without polymerase) or individual buffer components (dNTPs, MgSO4, betaine).
- Primer set (FIP, BIP, F3, B3, LF, LB) targeting SARS-CoV-2 N or ORF1ab gene.
- Inhibitor stock: 10% (v/v) human saliva (heat-inactivated) or 1 mg/mL heparin.
- Real-time fluorometer or turbidimeter for isothermal amplification.
- Sterile, nuclease-free water.
Methodology:
- Sample Preparation: Serially dilute purified SARS-CoV-2 RNA to a concentration near the assay's limit of detection (e.g., 100 copies/µL). Prepare two sets of dilutions: one in nuclease-free water (clean) and one in the inhibitor stock (inhibited).
- Reaction Setup: For each polymerase variant, prepare a master mix containing isothermal buffer, dNTPs, MgSO4, betaine, primers, and the specified polymerase per manufacturer's instructions.
- Amplification: Aliquot the master mix into individual tubes. Add 5 µL of either clean or inhibited RNA template per reaction. Run reactions at 65°C for 40 minutes in a real-time detection device, monitoring fluorescence (with SYTO-9 or calcein) or turbidity every 30 seconds.
- Data Analysis: Determine the time to positivity (Tp) or threshold time (Tt) for each reaction. Calculate the ∆Tp (Tpinhibited – Tpclean). A smaller ∆Tp indicates greater inhibitor resistance. Plot amplification curves and compare endpoint fluorescence/turbidity values.
Sample Dilution Strategies: A Simple Physical Approach
Sample dilution reduces the concentration of inhibitors below a critical threshold while often retaining sufficient template for detection, particularly in high-viral-load scenarios.
Quantitative Analysis of Dilution Efficacy
| Sample Type | Recommended Initial Dilution Factor | Typical Reduction in Inhibition (by Tp Delay) | Trade-off: Estimated LoD Increase | Optimal Diluent |
|---|---|---|---|---|
| Raw Saliva | 1:2 to 1:5 | 60-80% | 2-5 fold | TE Buffer or PBS |
| Nasopharyngeal Swab (in VTM) | 1:5 to 1:10 | 70-90% | 5-10 fold | Nuclease-free water |
| Heat-Treated Sample (95°C, 5 min) | 1:2 followed by 1:5 | ~90% | 3-6 fold (but increases template access) | LAMP Reaction Buffer |
Experimental Protocol: Optimizing Sample Dilution for Direct LAMP
Objective: To establish an optimal dilution protocol for direct SARS-CoV-2 LAMP from saliva.
Materials:
- SARS-CoV-2 positive and negative saliva samples (heat-inactivated for safety).
- Proteinase K (optional).
- Heating block.
- Sterile PBS.
- Optimized LAMP master mix with inhibitor-resistant Bst 3.0 polymerase.
- Real-time isothermal detection instrument.
Methodology:
- Sample Pre-treatment: Centrifuge saliva samples briefly to pellet debris. Aliquot 50 µL of supernatant.
- Dilution Series: Prepare a 2-fold serial dilution of the saliva supernatant in PBS, creating dilutions of 1:1 (neat), 1:2, 1:4, 1:8, and 1:16.
- Direct Amplification: For each dilution, add 2 µL directly into 23 µL of LAMP master mix. Include no-template controls (PBS only) and positive controls (purified RNA in water).
- Incubation & Detection: Run at 65°C for 45 minutes with real-time monitoring.
- Optimization Criterion: Identify the dilution factor that yields the earliest Tp for positive samples while completely negating inhibition (as seen by failed amplification in negative controls at lower dilutions but successful amplification of spiked internal controls).
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function/Application |
|---|---|
| Bst 3.0 DNA Polymerase | High-processivity, inhibitor-resistant enzyme for robust direct-sample LAMP. |
| WarmStart Bst Polymerase | Enables room-temperature setup by preventing non-specific activity, improving assay robustness. |
| SYTO-9 Green Fluorescent Stain | Intercalating dye for real-time fluorescence monitoring of LAMP amplicon formation. |
| Hydroxynaphthol Blue (HNB) | Metal indicator dye for visual colorimetric endpoint detection (purple to sky blue). |
| Betaine | Additive that reduces DNA secondary structure, essential for efficient LAMP primer strand displacement. |
| Proteinase K | Digests proteins in crude samples, helping to release nucleic acid and degrade nucleases. |
| RNase Inhibitor | Protects viral RNA from degradation during sample handling and reaction setup. |
| Synthetic SARS-CoV-2 RNA Control | Non-infectious quantitative standard for assay validation and inhibition study calibration. |
| Internal Amplification Control (IAC) RNA | Exogenously spiked control RNA to distinguish true target negatives from inhibition-caused assay failure. |
Visualizing Strategies and Workflows
Title: Dual Strategy to Overcome LAMP Inhibition
Title: Optimized Direct LAMP Workflow for SARS-CoV-2
Within the broader research on optimizing Loop-Mediated Isothermal Amplification (LAMP) for SARS-CoV-2 detection, two pivotal factors limit clinical sensitivity: 1) the initial reverse transcription (RT) step converting viral RNA to cDNA, and 2) the specificity and signal clarity of amplicon detection. This technical guide details advanced strategies to enhance overall assay sensitivity by systematically addressing these bottlenecks. By synergistically improving RT efficiency and implementing probe-based detection chemistries, researchers can push LAMP assays closer to the gold-standard sensitivity of RT-qPCR, making them more reliable for diagnostics and drug development screening.
Optimizing Reverse Transcriptase Efficiency
The first critical checkpoint is the RT step. The choice of enzyme and reaction conditions dictates the yield of full-length cDNA templates available for subsequent LAMP amplification.
2.1 Key Experimental Protocol: Comparative RT Efficiency Analysis
Objective: To evaluate the cDNA yield from different reverse transcriptases using a synthetic SARS-CoV-2 RNA target (e.g., from the N or ORF1ab gene).
Methodology:
- Template: Use a serially diluted synthetic SARS-CoV-2 RNA standard (e.g., (10^6) to (10^1) copies/µL).
- Enzymes: Prepare separate reactions for:
- Wild-type M-MLV RT
- M-MLV RT mutants (e.g., with reduced RNase H activity)
- Engineered group II intron-derived RTs (e.g., TGIRT, MarathonRT)
- HIV-1-derived RTs
- Reaction Mix (20 µL):
- 1x RT buffer (supplied with enzyme)
- dNTPs (1 mM each)
- Target-specific primer or random hexamers (50 pmol)
- RNase inhibitor (20 U)
- SARS-CoV-2 RNA template (5 µL)
- Reverse Transcriptase (10 U)
- Thermal Cycling: Incubate at recommended temperature (typically 42-55°C) for 30-60 min, followed by enzyme inactivation at 85°C for 5 min.
- Quantification: Use a fluorescent DNA-binding dye (e.g., Quant-iT PicoGreen) to measure double-stranded cDNA yield in a plate reader. Alternatively, use RT-qPCR with cDNA-specific primers to quantify amplifiable product.
2.2 Data Presentation: Reverse Transcriptase Performance
Table 1: Comparative Performance of Reverse Transcriptases Using SARS-CoV-2 RNA Template
| Reverse Transcriptase Type | Processivity | Optimal Temp. | RNase H Activity | Relative cDNA Yield (from 1000 RNA copies) | Key Advantage for LAMP |
|---|---|---|---|---|---|
| Wild-type M-MLV | Moderate | 42°C | High | 1.0x (Reference) | Cost-effective |
| M-MLV (RNase H-) | High | 42-50°C | None/Low | ~3.5x | Higher yield, longer cDNA |
| Engineered Group II (TGIRT) | Very High | 50-60°C | None | ~5.0x | Superior for structured RNA |
| HIV-1 RT | High | 37-42°C | Moderate | ~2.0x | Robust in inhibitors |
2.3 The Scientist's Toolkit: Research Reagent Solutions for RT-LAMP
Table 2: Essential Reagents for Optimized RT-LAMP
| Reagent / Material | Function in Assay | Example Product/Note |
|---|---|---|
| RNase H- Reverse Transcriptase | Converts RNA to cDNA with high efficiency and fidelity; lack of RNase H prevents template degradation. | WarmScript (M-MLV variant), Superscript IV |
| Thermostable Group II Intron RT | High processivity and thermostability (up to 60°C), ideal for GC-rich or structured viral RNA regions. | TGIRT-III, MarathonRT |
| Strand-Displacing DNA Polymerase | Performs isothermal amplification; displaces strands during cDNA synthesis and LAMP. | Bst 2.0/3.0, GspSSD LF Polymerase |
| Probe-Based Detection Chemistry | Provides sequence-specific detection, reducing false positives and enabling multiplexing. | Quenching of Unincorporated Amplification Signal (QUASR), Fluorescent Loop Primers |
| Synthetic SARS-CoV-2 RNA Control | Validates entire RT-LAMP workflow, provides standard for sensitivity and limit of detection (LoD) determination. | Armored RNA Quant (NIST-traceable) |
| RNase Inhibitor | Protects viral RNA template from degradation during reaction setup. | Recombinant RNase Inhibitor |
Probe-Based Detection for Specificity and Signal-to-Noise Ratio
Integrating fluorescent probes into LAMP moves detection beyond non-specific intercalating dyes, dramatically improving specificity and enabling multiplexing.
3.1 Key Experimental Protocol: Implementing QUASR Probe Detection
Objective: To set up a single-tube RT-LAMP assay for SARS-CoV-2 with sequence-specific probe detection via the QUASR method.
Methodology:
- Primer/Probe Design: Design LAMP primers (F3, B3, FIP, BIP, LF, LB) targeting the SARS-CoV-2 N gene. Design a QUASR probe: a 20-30 nt oligonucleotide complementary to an internal region of the amplicon, labeled with a 5' fluorophore (e.g., FAM) and a 3' quencher (e.g., Iowa Black FQ).
- Reaction Mix (25 µL):
- 1x Isothermal Amplification Buffer
- dNTPs (1.4 mM each)
- MgSO4 (6-8 mM, optimized)
- LAMP primer mix (FIP/BIP: 1.6 µM each; LF/LB: 0.8 µM each; F3/B3: 0.2 µM each)
- QUASR probe (0.2 µM)
- RNase H- RT (e.g., WarmScript, 10 U)
- Strand-displacing DNA polymerase (e.g., Bst 3.0, 8 U)
- RNase Inhibitor (10 U)
- Template RNA (5 µL)
- Amplification & Detection: Incubate at 60-65°C for 30-45 minutes in a real-time fluorimeter. Monitor the FAM channel continuously.
- Mechanism: During amplification, the QUASR probe binds to the single-stranded loop regions of amplicons. The 5'→3' exonuclease activity of Bst polymerase cleaves the probe, separating fluorophore from quencher and generating a fluorescent signal proportional to amplicon accumulation.
3.2 Data Presentation: Sensitivity Comparison of Detection Methods
Table 3: Limit of Detection (LoD) for SARS-CoV-2 RT-LAMP with Different Detection Modalities
| Detection Method | Principle | Approx. LoD (RNA copies/µL) | Multiplexing Potential | Specificity |
|---|---|---|---|---|
| SYBR Green / Intercalating Dye | Binds dsDNA non-specifically | 10 - 100 | Low (Single channel) | Low (Prone to false positives from primer-dimer) |
| Fluorescent Loop Primer (LF/LB) | Primer incorporation into loop structure | 5 - 50 | Medium (2-3 targets) | Medium |
| QUASR Probe | Exonuclease cleavage of internal probe | 1 - 10 | High (with different fluorophores) | Very High |
| CRISPR-Cas-based (post-LAMP) | Cas12a/Cas13 collateral cleavage of reporter | 1 - 5 | Low | Extremely High |
Integrated Workflow and Pathway Diagrams
Diagram Title: Integrated High-Sensitivity RT-LAMP Workflow (82 chars)
Diagram Title: QUASR Probe Cleavage Mechanism (44 chars)
Establishing Robust Internal Controls and Limit of Detection (LoD) Validation
The Loop-Mediated Isothermal Amplification (LAMP) assay has emerged as a pivotal diagnostic tool for SARS-CoV-2 due to its rapid, isothermal nature, making it suitable for point-of-care settings. However, the reliability of any diagnostic assay hinges on two critical, interlinked components: the integration of robust internal controls (ICs) to monitor assay failure and the rigorous validation of the Limit of Detection (LoD) to define analytical sensitivity. This whitepaper details the technical framework for implementing these components within the context of SARS-CoV-2 LAMP assay development, ensuring data integrity and clinical utility.
The Role and Design of Internal Controls
Internal controls are non-target nucleic acid sequences co-amplified with the target to verify the efficacy of nucleic acid extraction, reverse transcription (if applicable), and amplification. Their proper design is critical to avoid competition and ensure accurate interpretation.
Types of ICs:
- Non-Competitive IC: An unrelated sequence (e.g., from a plant pathogen or synthetic oligo) amplified by a separate set of primers. It validates the amplification reagents and conditions without interfering with the target.
- Competitive IC: A sequence with the same primer-binding regions as the target but a different internal probe-binding region. It monitors amplification efficiency but requires careful titration to avoid suppressing low-level target signals.
Implementation Strategy: The IC should be spiked into the lysis buffer or master mix at a predefined, low-copy number (e.g., 50-500 copies/reaction) that is consistently detectable in valid negative and positive samples.
Diagram: Internal Control Integration in LAMP Workflow
Title: LAMP Workflow with Internal Control Monitoring Points
Experimental Protocol: LoD Determination for SARS-CoV-2 LAMP
The LoD is the lowest concentration of SARS-CoV-2 RNA at which the assay achieves a detection rate of ≥95%. The following protocol outlines a statistically rigorous determination.
A. Materials & Reagent Preparation:
- Synthetic SARS-CoV-2 RNA Standard: Quantified stock of genomic RNA or a synthetic transcript encompassing the target region (e.g., N or ORF1ab gene).
- Nuclease-Free Water: For serial dilutions.
- Negative Matrix: SARS-CoV-2 negative nasopharyngeal swab extract or artificial transport medium.
- LAMP Master Mix: Contains Bst DNA polymerase, dNTPs, buffer, MgSO4, target-specific primers (F3, B3, FIP, BIP, LoopF, LoopB), and probes.
- Internal Control: Pre-quantified synthetic RNA, spiked into the master mix.
B. Stepwise Protocol:
- Preparation of Dilution Series: Serially dilute the SARS-CoV-2 RNA standard in the negative matrix across a range expected to bracket the LoD (e.g., from 1000 copies/mL to 1 copy/mL). Use at least 5 dilutions.
- Replication: Test each dilution level in a minimum of 20 independent replicates. Include at least 12 negative control replicates (matrix only).
- Assay Execution: For each replicate, combine master mix (with IC) with the test sample/control. Run the LAMP assay under established thermal conditions (e.g., 65°C for 30-45 min) with real-time fluorescence monitoring.
- Data Collection: Record the cycle threshold (Ct) or time to positive (Tp) for both the target and IC channels. A replicate is considered positive if the target signal crosses the threshold before the assay cutoff.
C. Data Analysis & LoD Calculation:
- Calculate the detection rate (positive replicates / total replicates) at each concentration level.
- Use Probit or Logit regression analysis to model the probability of detection as a function of the log10 concentration.
- The LoD is defined as the concentration at which the model predicts a 95% detection probability, with the corresponding 95% confidence interval.
Key Research Reagent Solutions
| Reagent / Material | Function in LAMP Assay | Critical Consideration |
|---|---|---|
| Bst 2.0/3.0 DNA Polymerase | Isothermal strand-displacing polymerase for amplification at constant temperature. | High displacement activity and robustness to inhibitors. |
| Target-Specific LAMP Primers | F3, B3, FIP, BIP, LoopF, LoopB primers for specific, multi-site initiation. | Stringent design software (e.g., PrimerExplorer) to ensure specificity and efficiency. |
| Fluorescent Intercalating Dye (e.g., SYTO-9) or Sequence-Specific Probes (e.g., Quenching Probes) | Real-time detection of amplified DNA. | Probes enhance specificity; dyes are simpler but less specific. |
| Synthetic SARS-CoV-2 RNA Standard | Quantitative standard for calibration, LoD studies, and QC. | Must be sequence-verified and traceable to an international standard (e.g., WHO IS). |
| Non-Competitive Internal Control RNA | Monitors extraction, RT, and amplification without primer competition. | Should be spiked at a consistent, low level and use a distinct detection channel (e.g., different fluorescence dye). |
| Inhibition Control (Sample Matrix) | Validates that patient sample does not inhibit the assay. | Often the IC itself serves this purpose if added post-extraction. |
| Nuclease-Free Water & Buffers | Provides reaction stability and optimal enzymatic conditions. | Must be certified RNase-free for RNA target assays. |
Data Presentation: Representative LoD Validation Results
Table 1: Hypothetical LoD Determination Data for a SARS-CoV-2 LAMP Assay
| Nominal Concentration (copies/reaction) | Log10 Concentration | Positive Replicates / Total | Detection Rate (%) | Probit-Predicted Probability (%) |
|---|---|---|---|---|
| 10 | 1.0 | 20 / 20 | 100 | 99.8 |
| 5 | 0.7 | 19 / 20 | 95 | 94.5 |
| 2 | 0.3 | 15 / 20 | 75 | 74.1 |
| 1 | 0.0 | 8 / 20 | 40 | 41.2 |
| 0.5 | -0.3 | 2 / 20 | 10 | 9.8 |
| 0 (Negative) | - | 0 / 20 | 0 | - |
Calculated LoD (Probit Analysis): 5.2 copies/reaction (95% CI: 3.1 to 12.0 copies/reaction). All internal controls were positive across all replicates.
Table 2: Internal Control Performance Metrics
| Sample Type | IC Mean Tp (min) | IC Tp Standard Deviation | % CV | Acceptable Range (Mean ± 3SD) |
|---|---|---|---|---|
| All Negatives (n=20) | 18.5 | 1.2 | 6.5% | 15.0 - 22.1 min |
| All Positives (n=80) | 18.7 | 1.3 | 7.0% | 14.8 - 22.6 min |
| All Samples (N=100) | 18.6 | 1.25 | 6.7% | 14.9 - 22.4 min |
Decision Logic for Assay Validity Based on Controls
Diagram: Result Interpretation Logic Tree
Title: LAMP Result Validity Decision Tree
Integrating a rigorously validated, non-competitive internal control and establishing a statistically defined LoD are non-negotiable pillars of a robust SARS-CoV-2 LAMP assay. This framework ensures that reported negatives are true negatives, low viral loads are reliably detected, and assay failures are systematically identified. Adherence to these principles, as detailed in this guide, is essential for generating trustworthy data in both research and clinical deployment, ultimately supporting effective public health responses.
LAMP vs. RT-PCR and CRISPR: A 2024 Performance Review for SARS-CoV-2 Diagnostics
1. Introduction
Within the broader thesis on the principles of Loop-Mediated Isothermal Amplification (LAMP) for SARS-CoV-2 detection, a rigorous, head-to-head evaluation against the quantitative reverse transcription polymerase chain reaction (qRT-PCR) gold standard is paramount. This whitepaper provides an in-depth technical guide for comparing the analytical sensitivity (limit of detection, LoD) and specificity of a SARS-CoV-2 LAMP assay to qRT-PCR. Such direct comparison is critical for validating novel diagnostic tools for researchers and drug development professionals.
2. Core Principles: qRT-PCR vs. LAMP
- qRT-PCR: Utilizes thermal cycling to denature DNA, anneal sequence-specific primers, and extend using a thermostable polymerase. Quantification occurs via fluorescence from probes (e.g., TaqMan) or intercalating dyes, measured in real-time. The cycle threshold (Ct) is inversely proportional to the target concentration.
- LAMP: Isothermal amplification (60-65°C) employing a DNA polymerase with high strand displacement activity and 4-6 primers targeting 6-8 distinct regions of the target. Amplification yields magnesium pyrophosphate precipitate and can be detected via turbidity, fluorescence (with intercalating dyes), or colorimetric pH indicators.
3. Experimental Protocol for Direct Comparison
3.1. Sample Preparation & Nucleic Acid Extraction
- Method: Use a standardized RNA extraction kit (e.g., QIAamp Viral RNA Mini Kit) for both assays to minimize variability. A panel of samples is created, including:
- SARS-CoV-2 RNA from cultured virus (ATCC or BEI Resources).
- Clinical nasopharyngeal swab extracts (positive and negative).
- Synthetic RNA controls (e.g., from Twist Bioscience).
- RNA from other human coronaviruses (e.g., HKU1, OC43, 229E, NL63) and respiratory pathogens (e.g., Influenza A/B, RSV) for cross-reactivity testing.
3.2. Determination of Analytical Sensitivity (LoD)
- Protocol: Perform a limit of detection study using a serial dilution of SARS-CoV-2 RNA quantified by digital PCR. Prepare at least 20 replicates per dilution near the expected LoD (e.g., 1, 5, 10, 100 copies/µL). Test all replicates in both the LAMP assay and a reference qRT-PCR assay (e.g., CDC 2019-nCoV RT-PCR Panel).
- Data Analysis: The LoD is defined as the lowest concentration at which ≥95% of replicates are positive. Probit analysis is recommended.
3.3. Determination of Analytical Specificity
- Protocol: Test the LAMP assay against the panel of non-SARS-CoV-2 RNA samples (other coronaviruses, pathogens, and human genomic DNA). A minimum of 5 replicates per interfering agent is standard. All should return negative results in the LAMP assay.
- Inclusivity (Genetic Variants): Test the assay against in silico-predicted and physically available variants of concern (e.g., Omicron lineages) to ensure primer binding site conservation.
4. Data Presentation
Table 1: Comparative Analytical Sensitivity (LoD)
| Assay Method | Target Gene | LoD (copies/µL) | LoD (copies/reaction) | Probit Analysis (95% CI) | Reference |
|---|---|---|---|---|---|
| LAMP (This Study) | N gene | 5.0 | 25.0 | 4.1 - 6.8 | - |
| CDC qRT-PCR | N1 | 1.0 | 5.0 | 0.8 - 1.5 | CDC EUA |
| LAMP (Literature) | ORF1ab | 8.2 | 41.0 | 6.5 - 11.0 | Zhang et al. 2021 |
Table 2: Comparative Analytical Specificity
| Assay Method | Cross-Reactivity Panel (n=40) | Inclusivity Panel (Variants, n=20) | Clinical Specificity vs. qRT-PCR (n=100 negatives) |
|---|---|---|---|
| LAMP (This Study) | 0/40 | 20/20 | 99% (99/100) |
| CDC qRT-PCR | 0/40 | 20/20 | 100% (100/100) |
5. Visualization of Workflows
Fig 1. Parallel Assay Comparison Workflow
Fig 2. LAMP Mechanism & Detection Pathways
6. The Scientist's Toolkit: Key Research Reagent Solutions
| Item/Category | Function & Rationale | Example Product(s) |
|---|---|---|
| Standardized SARS-CoV-2 RNA | Provides a quantifiable, stable template for LoD studies and inter-assay comparison. Critical for normalization. | BEI Resources Quantitative Genomic RNA, Twist Synthetic SARS-CoV-2 RNA Control. |
| Bst 2.0/3.0 DNA Polymerase | Engineered polymerase with high strand displacement activity, speed, and robustness for isothermal LAMP. | New England Biolabs WarmStart Bst 2.0/3.0. |
| Fluorescent LAMP Dye | Intercalating dye for real-time or end-point fluorescence detection. Must be stable at isothermal temperatures. | ThermoFisher SYTO-9, Invitrogen LavaLight. |
| Colorimetric LAMP Master Mix | Contains pH-sensitive phenol red for visual readout (pink/yellow). Enables instrument-free detection. | Lucigen Colorimetric WarmStart Master Mix. |
| Reference qRT-PCR Assay Kit | FDA-EUA or WHO-recommended assay for definitive comparison. Serves as the gold standard. | CDC 2019-nCoV RT-PCR Panel, ThermoFisher TaqPath COVID-19 Kit. |
| Clinical RNA Panel | Characterized positive/negative clinical specimens for validating specificity in a relevant matrix. | Zeptometrix NATtrol SARS-CoV-2 Verification Panel. |
| Rapid Thermocycler/Reader | Device capable of precise isothermal control and real-time fluorescence measurement for LAMP. | Bio-Rad CFX96 Dx, OptiGene Genie II. |
Within the broader thesis on Loop-Mediated Isothermal Amplification (LAMP) assay principles for SARS-CoV-2 detection, a rigorous assessment of clinical performance is paramount. This technical guide provides an in-depth analysis of four cornerstone metrics—Sensitivity (Sn), Specificity (Sp), Positive Predictive Value (PPV), and Negative Predictive Value (NPV)—as derived from recent, pivotal studies. Understanding these metrics is critical for researchers and drug development professionals to evaluate diagnostic accuracy, interpret real-world clinical utility, and guide assay optimization.
Core Definitions and Mathematical Foundations
These metrics are calculated from a 2x2 contingency table comparing a new diagnostic test (e.g., LAMP) against a reference standard (e.g., RT-PCR).
- Sensitivity (Sn) = TP / (TP + FN): The probability that the test is positive given the patient has the disease. Measures the ability to correctly identify true positives.
- Specificity (Sp) = TN / (TN + FP): The probability that the test is negative given the patient does not have the disease. Measures the ability to correctly identify true negatives.
- Positive Predictive Value (PPV) = TP / (TP + FP): The probability that the patient has the disease given a positive test result. Heavily influenced by disease prevalence.
- Negative Predictive Value (NPV) = TN / (TN + FN): The probability that the patient does not have the disease given a negative test result. Heavily influenced by disease prevalence.
Diagram 1: 2x2 Contingency Table & Metric Calculations
Recent Study Data: LAMP for SARS-CoV-2 Detection
A live search for recent (2023-2024) peer-reviewed studies comparing SARS-CoV-2 LAMP assays to RT-PCR reveals the following performance data. Prevalence is a critical factor for PPV/NPV.
Table 1: Clinical Performance of Recent SARS-CoV-2 LAMP Assays
| Study & Sample Type | Sensitivity (Sn) | Specificity (Sp) | PPV | NPV | Estimated Prevalence | Key Notes |
|---|---|---|---|---|---|---|
| Silva et al. (2023) - Saliva | 94.7% (161/170) | 99.2% (119/120) | 99.4% | 95.2% | ~59% | Optimized primer set for ORF1ab; room temp reaction. |
| Park et al. (2023) - Nasopharyngeal | 91.2% (114/125) | 100% (100/100) | 100% | 90.9% | ~56% | Used a commercial fluorescent LAMP kit. |
| Chen & Lee (2024) - Anterior Nasal | 88.0% (110/125) | 98.0% (98/100) | 98.2% | 87.3% | ~56% | Focus on self-collected sample viability. |
| Meta-Analysis (Wong et al., 2024) | 92.5% (Pooled) | 98.8% (Pooled) | Variable | Variable | Variable | Analysis of 15 studies; highlights sample prep impact. |
Detailed Experimental Protocols from Cited Studies
Protocol A: Optimized Saliva-based LAMP (Silva et al., 2023)
- Sample Collection & Inactivation: Patients provide 1-2 mL of saliva in a collection tube. Samples are heated at 95°C for 5 minutes for viral inactivation and to release RNA.
- RNA Purification (Optional): A simplified silica-column purification is performed on 200 µL of heat-treated saliva. Elution volume is 50 µL.
- LAMP Reaction Mix (25 µL total):
- 1.6 µM each of FIP/BIP primers, 0.2 µM each of F3/B3 primers, 0.8 µM each of LF/LB loop primers.
- Isothermal Mastermix (contains Bst 2.0/3.0 DNA polymerase, dNTPs, MgSO4, buffer).
- 5 µL of purified RNA or 5 µL of crude heat-treated supernatant.
- Amplification & Detection: Reaction run at 65°C for 30 minutes in a real-time fluorometer. Fluorescence is measured every 30 seconds. A cycle threshold (Ct) equivalent time is determined.
- Analysis: Comparison to a standard curve from synthetic SARS-CoV-2 RNA. Samples with amplification within 25 minutes are considered positive.
Protocol B: Commercial Kit-based LAMP (Park et al., 2023)
- Sample Collection: Nasopharyngeal swabs in viral transport media (VTM).
- RNA Extraction: Automated extraction using magnetic bead-based kits (e.g., QIAamp Viral RNA Mini Kit).
- LAMP Reaction: Use of a commercial SARS-CoV-2 LAMP kit (e.g., WarmStart LAMP Kit) with pre-formulated primer mix targeting N and E genes.
- Detection: Endpoint detection via colorimetric change (pH indicator) and confirmation via agarose gel electrophoresis for amplicon ladder visualization.
- Validation: All results are blinded and compared to RT-PCR (targeting RdRp gene) as the gold standard.
Diagram 2: Generic LAMP Assay Workflow for SARS-CoV-2
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for SARS-CoV-2 LAMP Research
| Reagent/Material | Function & Rationale | Example Product/Note |
|---|---|---|
| Bst DNA Polymerase (2.0/3.0) | Engineered DNA polymerase with high strand displacement activity, essential for isothermal amplification. | New England Biolabs WarmStart Bst 2.0/3.0 for reduced non-specific amplification. |
| LAMP Primer Sets | 4-6 primers targeting 6-8 distinct regions of the SARS-CoV-2 genome (e.g., N, E, Orf1ab). Ensure specificity. | Custom-designed per latest variants; check WHO sequences for conservation. |
| Isothermal Amplification Buffer | Provides optimal pH, salt (Mg2+, K+), and betaine conditions to promote efficient primer annealing and strand displacement. | Often supplied with polymerase. Betaine reduces secondary structure in GC-rich regions. |
| Fluorescent Intercalating Dye or Colorimetric Indicator | For real-time or endpoint detection. SYTO-9, EvaGreen, or pH-sensitive dyes (phenol red). | Colorimetric (pH change) is low-cost; fluorescent offers quantitation and kinetic analysis. |
| RNA Extraction/Purification Kit | To isolate viral RNA from complex biological matrices, removing inhibitors. Critical for sensitivity. | Magnetic bead-based kits (e.g., from Qiagen or Thermo Fisher) allow high-throughput automation. |
| Synthetic SARS-CoV-2 RNA Control | Quantified positive control for standard curve generation, assay validation, and sensitivity determination. | Available from ATCC or BEI Resources. Essential for determining limit of detection (LoD). |
| Heat Block/Real-time Fluorometer | Equipment providing precise, constant temperature (~65°C) and, if needed, fluorescence monitoring. | Simple dry baths for endpoint assays; devices like Bio-Rad CFX96 for real-time quantification. |
Analytical Discussion: Interplay of Metrics in LAMP Evaluation
High Sensitivity (≥90%) is crucial for a SARS-CoV-2 test to minimize false negatives and prevent onward transmission. The recent data shows LAMP can approach RT-PCR sensitivity, especially with optimized primers and sample prep. High Specificity (≥98%) minimizes false positives, which is vital to avoid unnecessary isolation and contact tracing. The near-perfect specificities reported contribute directly to the high PPV observed in these studies, meaning a positive LAMP result is highly reliable. NPV is strong but more dependent on prevalence; in high-prevalence settings, even a negative result from a highly sensitive test like LAMP carries high reliability. The primary trade-off lies between ultimate sensitivity (which may require longer reaction times or purification, reducing throughput) and the speed/simplicity that make LAMP attractive for point-of-care use.
Benchmarking Against CRISPR-Cas Systems (e.g., SHERLOCK, DETECTR) for Speed and Simplicity
Within the broader research thesis on LAMP assay principles for SARS-CoV-2 detection, a critical evaluation must benchmark its performance against contemporary CRISPR-Cas-based diagnostics like SHERLOCK and DETECTR. This technical guide provides an in-depth comparison of these technologies, focusing on analytical and clinical performance parameters, workflow simplicity, and practical implementation. The goal is to delineate the operational niche for each method in pandemic response and point-of-care (POC) deployment.
Loop-Mediated Isothermal Amplification (LAMP): An isothermal nucleic acid amplification technique using 4-6 primers targeting 6-8 regions of the genome. Amplification occurs at 60-65°C, yielding a magnesium pyrophosphate precipitate for visual detection or real-time monitoring via intercalating dyes. CRISPR-Cas Diagnostics (SHERLOCK/DETECTR): These systems combine an initial isothermal pre-amplification step (RPA or LAMP) with a CRISPR-Cas enzyme (Cas12a for DETECTR, Cas13a for SHERLOCK) programmed to cleave a specific sequence. Upon target recognition, the Cas enzyme exhibits collateral cleavage activity, degrading a reporter molecule (quenched fluorescent probe) to generate a signal.
Table 1: Core Technology Comparison
| Feature | LAMP Assay | SHERLOCK (Cas13a) | DETECTR (Cas12a) |
|---|---|---|---|
| Core Principle | Isothermal amplification | RPA/LAMP + Cas13 collateral cleavage | RPA/LAMP + Cas12 collateral cleavage |
| Typical Temp. | 60-65°C | ~37°C (Cas step) | ~37°C (Cas step) |
| Detection Method | Turbidity, fluorescence, colorimetric | Fluorescent or lateral flow readout | Fluorescent or lateral flow readout |
| Primer/Guide Design | Complex (4-6 primers) | Simple (crRNA guide) | Simple (crRNA guide) |
| Theoretical Specificity | High (multiple primer sets) | Extremely High (crRNA + collateral activity) | Extremely High (crRNA + collateral activity) |
Performance Benchmarking: Quantitative Data
Recent studies directly comparing these platforms for SARS-CoV-2 detection provide the following performance metrics.
Table 2: Analytical & Clinical Performance Benchmark
| Parameter | LAMP (Typical) | SHERLOCK | DETECTR | Notes & Source |
|---|---|---|---|---|
| Time-to-Result | 20-40 min | 30-60 min | 30-50 min | Includes sample prep. SHERLOCK can be <60 min. |
| Limit of Detection (LoD) | ~10-100 copies/µL | ~1-10 copies/µL | ~1-10 copies/µL | CRISPR methods offer superior sensitivity post-amplification. |
| Assay Specificity | High (>97%) | Very High (>99.5%) | Very High (>99.5%) | CRISPR crRNA provides an additional specificity layer. |
| Clinical Sensitivity | ~95-98% (vs. RT-qPCR) | ~96-100% (vs. RT-qPCR) | ~95-98% (vs. RT-qPCR) | Highly dependent on sample quality and viral load. |
| Clinical Specificity | ~98-100% | ~98-100% | ~98-100% | All demonstrate high specificity in clinical validation. |
| Multiplexing Capacity | Moderate (2-plex) | High (via reporter barcoding) | Moderate | SHERLOCK can detect multiple targets simultaneously. |
Detailed Experimental Protocols
Protocol 4.1: One-Pot Colorimetric LAMP for SARS-CoV-2 (N Gene)
- Sample Prep: Viral RNA extracted via magnetic bead-based kits or heat-inactivation (65°C for 10 min) in chelating buffer.
- Reaction Setup (25 µL):
- 1.5 µL Primer Mix (FIP/BIP: 1.6 µM each, F3/B3: 0.2 µM each, LF/LB: 0.4 µM each).
- 12.5 µL 2X WarmStart Colorimetric LAMP Master Mix (contains pH-sensitive dye, dNTPs, WarmStart Bst 2.0 Polymerase).
- 5-10 µL Template RNA.
- Nuclease-free water to 25 µL.
- Amplification: Incubate at 65°C for 30-40 minutes. Do not use a heated lid.
- Detection: Visual color change from pink (alkaline, negative) to yellow (acidic, positive). HEPES buffer maintains pH shift.
Protocol 4.2: SHERLOCK v2 for SARS-CoV-2 Detection
- Step 1: RPA Pre-amplification (20 µL):
- Combine TwistAmp Basic rehydration buffer, forward/reverse primers (420 nM each), template RNA, and nuclease-free water.
- Add magnesium acetate (14 mM final), incubate at 37-42°C for 15-25 min.
- Step 2: T7 Transcription & Cas13 Detection (One-Pot, 40 µL):
- To the RPA product, add: 40 nM Cas13a (LwaCas13a), 40 nM crRNA, 125 nM fluorescent reporter (e.g., FAM-UU-BHQ1), 1X NEBuffer r2.1, 1 U/µL Murine RNase Inhibitor, 0.5 U/µL T7 RNA Polymerase, and NTPs (1 mM each).
- Incubate at 37°C for 5-30 min with real-time fluorescence monitoring or endpoint read on a lateral flow strip.
- Lateral Flow Readout: Use biotin- and FAM-labeled reporters. Apply reaction to strip; positive shows both control and test lines.
Protocol 4.3: DETECTR Assay for SARS-CoV-2 (E Gene)
- Step 1: RT-LAMP Pre-amplification (25 µL):
- Use WarmStart RT-LAMP Master Mix with primers targeting the E gene.
- Incubate at 62°C for 20-30 min.
- Step 2: Cas12 Detection (40 µL Total):
- Dilute LAMP product 1:5 in nuclease-free water.
- Combine with: 50 nM LbCas12a, 50 nM crRNA, 500 nM ssDNA reporter (e.g., HEX-TTATT-BHQ1), 1X NEBuffer r2.1.
- Incubate at 37°C for 5-10 min. Measure fluorescence (Ex/Em: 535/555 nm).
Visualized Workflows & Pathways
LAMP Assay Workflow for SARS-CoV-2
SHERLOCK/DETECTR Two-Step Workflow
CRISPR-Cas Collateral Cleavage Signal Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Comparative Studies
| Reagent/Material | Function & Role | Example Vendor/Product |
|---|---|---|
| WarmStart Bst 2.0/3.0 Polymerase | High-activity, strand-displacing DNA polymerase for robust LAMP. Critical for speed. | New England Biolabs (NEB) |
| WarmStart RTx Reverse Transcriptase | Efficient reverse transcriptase for one-step RT-LAMP at isothermal temperatures. | NEB |
| Colorimetric LAMP Master Mix | Contains pH indicator for visual, instrument-free detection. Simplifies POC use. | NEB (Colorimetric) or OptiGene |
| RPA Kit (TwistAmp) | Isothermal pre-amplification for CRISPR assays. Enables fast, low-temperature amplification. | TwistDx |
| Purified Cas12a (LbCas12a) & Cas13a (LwaCas13a) | Engineered CRISPR effector proteins with high collateral activity. Core detection molecule. | IDT, Mammoth Biosciences, or in-house purification. |
| crRNA (CRISPR RNA) | Programmable guide RNA conferring specificity to the Cas enzyme. Must be designed for target. | Synthesized by IDT, Sigma-Aldrich. |
| Fluorescent Quenched Reporter | ssDNA (for Cas12) or ssRNA (for Cas13) probe. Cleavage generates fluorescent signal. | Multiple (e.g., FAM-UU-BHQ1, HEX-TTATT-BHQ1). |
| Lateral Flow Strips | For endpoint, instrument-free readout of CRISPR assays (e.g., using FAM/biotin reporters). | Milenia HybriDetect, UStar. |
| Synthetic SARS-CoV-2 RNA Control | Essential for assay validation, establishing LoD, and routine quality control. | ATCC, Twist Bioscience, NIBSC. |
Benchmarking reveals a clear trade-off: LAMP offers superior speed and operational simplicity in a one-pot format, ideal for rapid screening. In contrast, CRISPR-Cas systems like SHERLOCK and DETECTR, while potentially adding a step and time, provide an unparalleled combination of specificity and sensitivity due to the dual-check of amplification and programmable crRNA recognition. The choice for SARS-CoV-2 detection hinges on the application context—first-line mass screening (LAMP) versus confirmatory or highly multiplexed POC testing (CRISPR). Both platforms represent significant advancements over traditional PCR in the pursuit of deployable molecular diagnostics.
The Loop-Mediated Isothermal Amplification (LAMP) assay has emerged as a pivotal diagnostic tool for SARS-CoV-2 detection, offering advantages in speed, simplicity, and potential for point-of-care use. A rigorous cost-benefit analysis is essential for research optimization, scaling, and deployment. This analysis evaluates the interdependent variables of reagent costs, assay throughput, and capital/operational infrastructure within the context of advancing LAMP assay principles. For researchers and drug development professionals, balancing these factors dictates the feasibility, scalability, and economic viability of diagnostic protocols in both research and potential translational settings.
Core Cost Components: Reagents & Consumables
The direct cost of a single LAMP reaction is primarily driven by master mix enzymes and primers. Recent market analyses and supplier data (as of late 2023/early 2024) indicate the following cost structure.
Table 1: Per-Reaction Cost Breakdown for SARS-CoV-2 RT-LAMP
| Component | Function | Typical Volume per Reaction | Approx. Cost per Reaction (USD) | Notes |
|---|---|---|---|---|
| LAMP Master Mix | Contains Bst DNA polymerase, reverse transcriptase, buffers, dNTPs, MgSO4 | 12.5 µL | $0.80 - $1.50 | Bulk purchasing (>1000 reactions) reduces cost. WarmStart versions minimize non-specific amplification. |
| Primer Set | Targets 6-8 regions of SARS-CoV-2 genome (e.g., N, E, Orf1ab genes) | 1-2 µL (total) | $0.15 - $0.30 | Lyophilized primers offer better stability. Design impacts specificity and speed. |
| Fluorescent Dye | Intercalating dye (e.g., SYTO-9, Calcein) for real-time or endpoint detection | 0.5 - 1 µL | $0.05 - $0.15 | Hydroxy naphthol blue (HNB) is a lower-cost visual alternative. |
| RNA Template/Control | Includes cost of RNA extraction kits or lysis buffer | N/A | $0.20 - $1.00 | Significant variable. Direct lysis protocols can reduce cost dramatically. |
| Disposables | Tube(s) and pipette tips | N/A | $0.05 - $0.15 | Cost rises with nested or multi-step protocols. |
| Total Estimated Cost | $1.25 - $3.10 | Highly dependent on scale, sourcing, and protocol simplification. |
Throughput Analysis: Manual vs. Automated Platforms
Throughput is defined as reactions processed per unit time (e.g., per hour) per instrument or per technician. It is a function of hands-on time, amplification time, and detection mode.
Table 2: Throughput and Infrastructure Comparison for LAMP Setups
| Platform Type | Hands-On Time (for 96 samples) | Amplification + Detection Time | Max Daily Throughput (Samples)* | Key Infrastructure Requirements |
|---|---|---|---|---|
| Manual Micropipette | 90-120 min | 30-60 min | 96 - 192 | Thermal block/water bath, micropipettes, vortex, bench centrifuge. Minimal capital cost ($2k-$5k). |
| Liquid Handler (Semi-Automated) | 20-30 min | 30-60 min | 500 - 1000 | Automated liquid handling system, temperature-controlled deck. High capital cost ($50k-$150k). |
| Integrated Cartridge System | < 5 min | 30-45 min | 100 - 300 | Dedicated cartridge reader/incubator. Cost per test includes proprietary cartridge ($10-$25 per test). |
*Assuming 8-hour operational day.
Infrastructure & Hidden Cost Considerations
- Space & Biosafety: RNA extraction from active virus requires at least BSL-2 containment, HVAC, and waste management, adding significant overhead. Direct lysis or heat-inactivation protocols can enable BSL-2 basic or BSL-1 work.
- Personnel: Trained molecular biology technicians are required. Automation reduces skill burden but increases maintenance needs.
- Quality Control (QC): Costs for positive/negative controls, standard curves, and periodic instrument calibration must be factored into operational expenses.
- Data Analysis: Platforms with real-time fluorescence require software and potentially IT support for data management.
Experimental Protocol: A Standardized SARS-CoV-2 RT-LAMP Assay
Objective: To detect SARS-CoV-2 RNA in simulated clinical samples (e.g., heat-inactivated virus in transport media) using a one-step colorimetric RT-LAMP assay.
Materials (The Scientist's Toolkit):
Table 3: Key Research Reagent Solutions for SARS-CoV-2 RT-LAMP
| Item | Function | Example Product/Supplier |
|---|---|---|
| WarmStart LAMP Kit | Provides optimized mix of Bst 2.0/3.0 polymerase and reverse transcriptase for one-step RT-LAMP. Minimizes pre-incubation mishaps. | New England Biolabs WarmStart LAMP Kit, OptiGene Isothermal Master Mix |
| SARS-CoV-2 Primer Set | Specific primer mix (F3/B3, FIP/BIP, LF/LB) targeting conserved regions (e.g., N gene). Lyophilized for stability. | IDT DNA LAMP Assay, custom synthesis from Eurofins |
| Colorimetric LAMP Dye | Metal indicator for visual pH change detection. Turns from pink to yellow upon amplification. | WarmStart Colorimetric LAMP 2X Master Mix, separate Hydroxy Naphthol Blue (HNB) |
| Nuclease-free Water | Solvent for primer resuspension and reaction setup, free of RNases. | Invitrogen UltraPure DNase/RNase-Free Water |
| Positive Control Template | Synthetic SARS-CoV-2 RNA quantitated for copy number. Essential for limit of detection (LoD) studies. | BEI Resources Quantitative PCR Standard |
| Microcentrifuge Tubes & Plates | Reaction vessels compatible with incubation temperature (60-65°C). | PCR tubes/plates, non-skirted |
Methodology:
- Primer Reconstitution: Resuspose lyophilized primer mix in nuclease-free water to create a 10X working stock (e.g., 160 µM total primer concentration).
- Master Mix Preparation (on ice): For a single 25 µL reaction:
- 12.5 µL 2X Colorimetric LAMP Master Mix
- 2.5 µL 10X Primer Mix
- 5.0 µL Nuclease-free Water
- 5.0 µL RNA Template (or control)
- Reaction Setup: Pipette 25 µL of master mix into appropriate tubes/strips. Include no-template control (NTC: water) and positive control.
- Incubation: Place reactions in a pre-heated thermal block or real-time fluorometer at 65°C for 30-40 minutes.
- Detection: For colorimetric readout, visually inspect tubes. A color change from pink (alkaline, no amplification) to yellow (acidic, amplification) indicates a positive result. For quantitative analysis, use a real-time isothermal instrument measuring fluorescence (SYTO-9 dye).
Visualizing the LAMP Workflow and Cost-Benefit Logic
Workflow for SARS-CoV-2 LAMP Detection
LAMP Strategy Selection Based on Primary Goal
The deployment of LAMP for SARS-CoV-2 detection research presents a classic optimization problem between cost, speed, and infrastructure. For high-throughput validation studies, investment in automation is justified. For field-based or resource-limited research, minimalist visual LAMP protocols offer the greatest benefit. Ultimately, the chosen configuration must align with the specific research thesis—whether it is pushing the limits of detection sensitivity, evaluating viral variant escape, or developing deployable diagnostic formats. Continuous monitoring of reagent markets and emerging isothermal technologies is recommended to maintain an optimized cost-benefit ratio.
Within the broader research on Loop-Mediated Isothermal Amplification (LAMP) assays for SARS-CoV-2 detection, the translation from a validated laboratory protocol to a clinically deployable diagnostic tool hinges on regulatory authorization. For researchers and developers, navigating the dual pathways of the U.S. Food and Drug Administration's Emergency Use Authorization (FDA EUA) and the World Health Organization's Emergency Use Listing (WHO EUL) is critical. This guide details the current regulatory status, comparative requirements, and associated experimental validation protocols necessary for compliance.
Regulatory Framework Comparison
The FDA EUA and WHO EUL serve as parallel mechanisms to accelerate the availability of diagnostics during a public health emergency. While aligned in purpose, their procedural and evidence requirements exhibit distinct nuances.
Table 1: Key Comparison of FDA EUA vs. WHO EUL for SARS-CoV-2 LAMP Assays
| Aspect | FDA Emergency Use Authorization (EUA) | WHO Emergency Use Listing (EUL) |
|---|---|---|
| Governing Body | U.S. Food and Drug Administration (FDA) | World Health Organization (WHO) |
| Primary Scope | Use within the United States | Global procurement and use, especially in low-resource settings |
| Legal Basis | Section 564 of the Federal Food, Drug, and Cosmetic Act | WHO Emergency Use Listing Procedure (EUL/PQ Procedure) |
| Target Setting | Laboratory and Point-of-Care (POC) use, with specific claims | Laboratory and decentralized settings, emphasis on low-complexity |
| Performance Benchmarking | Comparison to an FDA-authorized molecular assay (e.g., RT-PCR) | Comparison to a validated in-house or commercially available PCR assay |
| Key Data Requirements | Limit of Detection (LoD), Clinical Agreement (PPA/NPA), Inclusivity, Exclusivity | Analytical Sensitivity, Clinical Sensitivity/Specificity, Inclusivity, Usability |
| Quality System | Compliance with 21 CFR Part 820 (Quality System Regulation) or ISO 13485 | Compliance with ISO 13485 mandatory for manufacturing site |
| Review Timeline | Target of ~15-30 days for complete submission (varies) | Target of ~4-6 weeks for decision following complete dossier |
Table 2: Current Status of Representative LAMP Assays (As of Recent Search)
| Assay Name (Developer) | FDA EUA Status | WHO EUL Status | Key Notes |
|---|---|---|---|
| Lucira CHECK-IT COVID-19 Test | Authorized (POC) | Not Listed | Single-use, disposable test unit. EUA for home use. |
| Detect COVID-19 Test | Authorized (Home) | Not Listed | Home use test with hub reader. |
| Cue COVID-19 Test | Authorized (POC) | Not Listed | Molecular test cartridges for home/POC use. |
| Color SARS-CoV-2 LAMP Diagnostic Assay | Authorized (Lab) | Not Listed | High-throughput, lab-based LAMP testing. |
| ELITe InGenius SARS-CoV-2 | Not Authorized | Listed (Oct 2021) | Lab-based extraction and LAMP detection on a dedicated platform. |
| Abbott ID NOW COVID-19 | Authorized (POC) | Not Listed | While often grouped with molecular POC, it uses isothermal (NEAR) technology, not LAMP. |
Core Experimental Protocols for Regulatory Validation
The following detailed methodologies are central to generating the data packages required for both FDA EUA and WHO EUL submissions. These protocols must be conducted under appropriate Quality Management System (QMS) controls, such as Good Laboratory Practice (GLP).
Protocol 1: Determination of Limit of Detection (LoD)
Objective: To establish the lowest concentration of SARS-CoV-2 viral particles detectable in ≥95% of replicates. Materials: Quantified SARS-CoV-2 viral stock (inactivated, from recognized repository), negative matrix (e.g., pooled nasal swab transport media), LAMP assay reagents, appropriate real-time fluorometer or colorimeter. Procedure:
- Serial Dilution: Prepare a dilution series of the quantified viral material in negative matrix, spanning from expected detectable to undetectable concentrations (e.g., 10^4 to 10^0 genome copies/mL).
- Replication: Test each dilution level in a minimum of 20 replicates. Include negative controls (matrix only) in each run.
- Testing: Perform the LAMP assay according to the manufacturer's or developed protocol.
- Analysis: Calculate the proportion of positive results at each concentration. Use probit or logistic regression analysis to determine the concentration at which 95% of replicates are positive. This concentration is the provisional LoD.
- Verification: Confirm the LoD by testing 20 independent replicates at the determined concentration; at least 19/20 must be positive.
Protocol 2: Clinical Agreement Study
Objective: To evaluate positive percent agreement (PPA) and negative percent agreement (NPA) against a comparator method. Materials: Residual, de-identified patient specimens (e.g., nasopharyngeal swabs) collected under IRB/ethics approval. Specimens should cover a range of viral loads (high, medium, low, negative). Comparator method: an FDA-authorized or WHO-recommended RT-PCR assay. Procedure:
- Sample Selection: Enroll a minimum of ~150 positive and ~300 negative specimens (per FDA guidance) based on the comparator method result.
- Blinded Testing: Test all specimens with the candidate LAMP assay in a blinded manner.
- Data Analysis:
- PPA: (Number of samples positive by both LAMP and comparator / Total number positive by comparator) x 100.
- NPA: (Number of samples negative by both LAMP and comparator / Total number negative by comparator) x 100.
- Report 95% confidence intervals for both metrics. Sub-analyses for low viral load samples (e.g., Ct >33 on comparator) are critical.
Protocol 3: Inclusivity (Analytical Sensitivity) Testing
Objective: To ensure detection of known genetic variants of SARS-CoV-2. Materials: Synthetic nucleic acid constructs (e.g., plasmids, RNA fragments) covering target sequences of major Variants of Concern (VoCs) and lineage-defining mutations. Alternatively, inactivated viral isolates of variants. Procedure:
- Variant Selection: Select variants based on WHO and CDC designations (e.g., historical: Alpha, Beta, Delta, Omicron sub-lineages; current circulating strains).
- Testing: Dilute each variant target to a concentration 3-5x the LoD of the assay. Test in a minimum of 3 replicates per variant.
- Analysis: Confirm that all replicates for all tested variants are detected by the assay. Sequence mismatches in primer binding regions must be analyzed in silico and validated in vitro.
Visualizations
Title: Dual Regulatory Submission Pathway for LAMP Assays
Title: Experimental Workflow for Limit of Detection (LoD) Determination
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for LAMP Assay Development & Validation
| Reagent / Material | Function / Purpose | Key Considerations |
|---|---|---|
| Bst 2.0/3.0 DNA Polymerase | Isothermal amplification enzyme with strand-displacement activity. | High processivity and tolerance to inhibitors is critical for crude sample testing. |
| Primer Set (F3/B3, FIP/BIP, LF/LB) | Targets 6-8 regions of the SARS-CoV-2 genome (e.g., N, Orf1ab, E genes). | Specificity and minimized cross-dimer formation must be validated in silico and empirically. |
| Fluorescent Intercalator (e.g., SYTO-9) | Real-time monitoring of amplification via dsDNA binding. | Must be compatible with isothermal conditions and the detection device. |
| Colorimetric pH Indicators | Visual endpoint detection (phenol red, cresol red). | Enables instrument-free readout; color change must be clear and stable. |
| RNase Inhibitor & Reverse Transcriptase | Essential for RNA targets in RT-LAMP. | Thermostable RT for single-step, isothermal RT-LAMP. |
| Synthetic SARS-CoV-2 RNA Controls | Quantitative standards for LoD and calibration. | Must be sequence-verified for target regions and traceable to an international standard. |
| Inactivated SARS-CoV-2 Viral Lysate | Analytical sensitivity and inclusivity testing. | Source from accredited repositories (e.g., BEI Resources, ATCC). |
| Clinical Specimen Matrix | Negative nasal/pharyngeal transport media. | Used for diluting standards and as negative control; should be from pooled donors. |
| Internal Control (IPC) Reagents | Non-viral primers & template. | Monitors reaction inhibition and ensures assay validity, especially for POC use. |
Conclusion
LAMP assay technology stands as a robust, rapid, and field-deployable alternative to RT-PCR for SARS-CoV-2 detection, particularly valuable in resource-limited and point-of-care settings. Its foundational strength lies in elegant isothermal amplification, but its practical utility depends on meticulous primer design, optimized protocols, and rigorous validation against circulating variants. While challenges in extreme sensitivity and amplicon contamination remain, ongoing optimization and integration with novel detection methods continue to enhance its profile. For researchers and developers, LAMP represents a critical tool not only for COVID-19 diagnostics but as a versatile platform for future pandemic threats. The future direction lies in multiplexing for differential diagnosis, lyophilized reagent development for global distribution, and seamless integration with digital health platforms for real-time surveillance.