Live Attenuated vs. Inactivated Vaccines: A Comprehensive Scientific Comparison of Mechanisms, Efficacy, and Applications
This article provides a detailed scientific and technical comparison of live attenuated and inactivated vaccine platforms, tailored for researchers, scientists, and drug development professionals.
Live Attenuated vs. Inactivated Vaccines: A Comprehensive Scientific Comparison of Mechanisms, Efficacy, and Applications
Abstract
This article provides a detailed scientific and technical comparison of live attenuated and inactivated vaccine platforms, tailored for researchers, scientists, and drug development professionals. It explores the foundational virology and immunology of each platform, methodologies for development and production, key challenges in safety and manufacturing, and comparative validation of immunogenicity, efficacy, and real-world application. The analysis synthesizes current data to inform platform selection for novel pathogen targets and future vaccine design.
Core Principles: Understanding the Virology and Immunology of Attenuated vs. Inactivated Vaccines
Vaccine platforms are broadly categorized by the nature of their immunogen. This guide provides a comparative analysis of the two foundational platforms: live attenuated vaccines (LAVs) and inactivated vaccines.
Fundamental Characteristics and Performance Comparison
The core distinctions between these platforms arise from the viability of the pathogen, which dictates immunogenicity, safety, and logistical profiles.
Table 1: Core Platform Characteristics and Comparative Performance
| Characteristic | Live Attenuated Vaccine (LAV) | Inactivated Vaccine |
|---|---|---|
| Pathogen State | Live, replication-competent but weakened (attenuated) virus/bacterium. | Pathogen killed (via heat, chemical, or radiation). Non-replicating. |
| Immune Response | Broad and robust. Mimics natural infection, inducing strong, long-lasting humoral (antibody) and cell-mediated immunity (cytotoxic T-cells). | Primarily humoral (antibody). Weaker or absent cytotoxic T-cell response. Often requires adjuvants. |
| Typical Dosage | Single dose often sufficient. | Multiple doses (prime-boost) usually required. |
| Onset of Immunity | Rapid (~1-2 weeks). | Slower, requires booster series to achieve high titers. |
| Duration of Immunity | Long-lasting (often decades). | Generally shorter, may require periodic boosters. |
| Safety Profile | Contraindicated in immunocompromised. Risk of reversion to virulence (theoretical). Mild, "mini-infection" symptoms possible. | Generally safer for all populations, including immunocompromised. No risk of infection. |
| Stability & Logistics | Often requires cold-chain refrigeration or freezing. Less stable. | Typically more stable, some formulations allow refrigerated storage. |
Table 2: Supporting Immunogenicity Data from Representative Studies
| Parameter (Measured Outcome) | Live Attenuated Vaccine (e.g., MMR) | Inactivated Vaccine (e.g., Inactivated Polio Vaccine - IPV) | Experimental Context (Reference) |
|---|---|---|---|
| Seroconversion Rate (%) | >95% for measles, mumps, rubella after 2 doses. | >99% for poliovirus types 1-3 after 3 doses. | Post-vaccination serology in children. |
| Geometric Mean Titer (GMT) | High, sustained GMTs over years. | High GMTs post-booster, may wane. | Longitudinal cohort studies. |
| CD8+ T-cell Response | Detectable and polyfunctional. | Negligible or absent without novel adjuvant/vector. | Intracellular cytokine staining (ICS) by flow cytometry. |
| Mucosal Immunity (IgA) | Often present (e.g., oral polio, flu mist). | Usually absent with parenteral administration. | Mucosal lavage samples post-vaccination. |
Experimental Protocols for Platform Characterization
Key methodologies for comparing these platforms involve assessing humoral and cellular immunity.
Protocol 1: Plaque Reduction Neutralization Test (PRNT) for Neutralizing Antibody Titers
- Purpose: Quantify functional, pathogen-specific neutralizing antibodies in serum.
- Method:
- Serial dilutions of heat-inactivated serum samples are prepared.
- A known quantity of live, infectious virus (e.g., 100 plaque-forming units) is added to each dilution and incubated to allow antibody-virus neutralization.
- The serum-virus mixtures are inoculated onto confluent monolayers of permissive cells (e.g., Vero cells) in multi-well plates.
- After adsorption, an overlay medium (e.g., carboxymethyl cellulose) is added to restrict viral spread, enabling plaque formation only from non-neutralized virus.
- Plates are incubated, fixed, stained (e.g., crystal violet), and plaques are counted.
- The PRNT50/PRNT90 titer is calculated as the reciprocal serum dilution that reduces plaque counts by 50% or 90% compared to virus-only controls.
Protocol 2: Intracellular Cytokine Staining (ICS) & Flow Cytometry for T-cell Responses
- Purpose: Detect antigen-specific T-cells (CD4+, CD8+) producing cytokines (IFN-γ, TNF-α, IL-2).
- Method:
- Cell Stimulation: Peripheral blood mononuclear cells (PBMCs) from vaccinated subjects are stimulated ex vivo with pathogen-specific peptide pools or whole inactivated antigen for 6-18 hours in the presence of a protein transport inhibitor (Brefeldin A/GolgiStop).
- Surface Staining: Cells are stained with fluorescent antibodies against surface markers (CD3, CD4, CD8).
- Fixation & Permeabilization: Cells are fixed (e.g., with paraformaldehyde) and permeabilized (e.g., with saponin-based buffer) to allow antibody entry.
- Intracellular Staining: Cells are stained with fluorescent antibodies against intracellular cytokines (IFN-γ, TNF-α, IL-2).
- Flow Cytometry & Analysis: Cells are acquired on a flow cytometer. Antigen-specific T-cells are identified as live, CD3+, CD4+/CD8+ cells positive for cytokine expression above the unstimulated control threshold.
Visualizations of Key Concepts
Title: Immune Activation Pathways for Two Vaccine Platforms
Title: Experimental Workflow for Neutralizing Antibody Assay
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for Comparative Vaccine Immunology Studies
| Reagent / Material | Function / Application | Key Considerations |
|---|---|---|
| Vero E6 / MDCK Cells | Permissive cell lines for viral culture, plaque assays (PRNT), and vaccine virus propagation. | Cell passage number and mycoplasma-free status are critical for assay consistency. |
| Pathogen-Specific Peptide Pools | Overlapping peptides spanning viral/bacterial proteins used to stimulate antigen-specific T-cells in ICS/ELISpot assays. | Class I (CD8+) vs. Class II (CD4+) peptide libraries must be selected appropriately. |
| Protein Transport Inhibitors (Brefeldin A, Monensin) | Block cytokine secretion during ex vivo T-cell stimulation, allowing intracellular accumulation for ICS detection. | Concentration and incubation time must be optimized to avoid cell toxicity. |
| Fluorochrome-conjugated Antibodies (Anti-CD3, CD4, CD8, IFN-γ, etc.) | Critical for phenotyping and detecting functional immune cells via multicolor flow cytometry. | Panel design must account for spectral overlap; titration is required for optimal signal-to-noise. |
| Commercial Neutralization Assay Kits (e.g., Pseudovirus-based) | Standardized, safer (BSL-2) alternative to wild-type virus PRNT for measuring neutralizing antibodies against enveloped viruses. | Must be validated against "gold standard" live-virus neutralization assays. |
| Adjuvant Controls (e.g., Alum, AS01) | Essential for inactivated vaccine studies to dissect adjuvant-specific from antigen-specific immune effects. | Choice depends on the desired immune polarization (Th1 vs. Th2). |
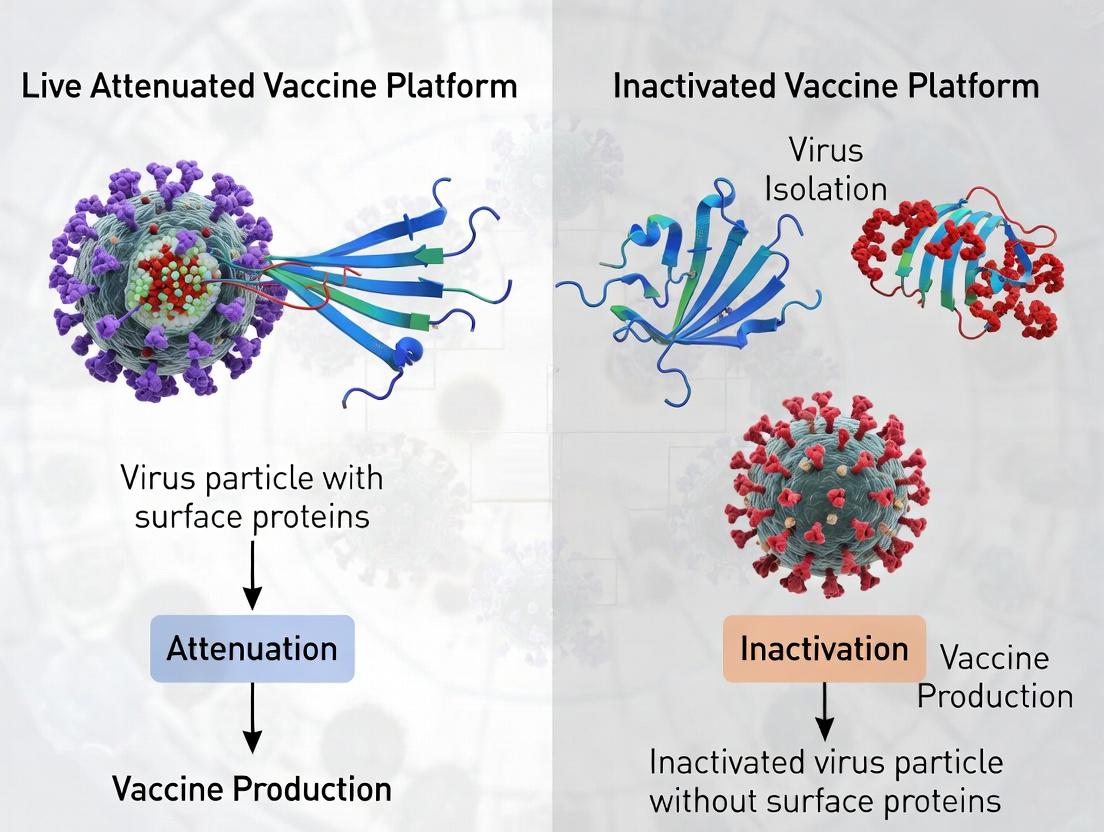
This guide, framed within broader research comparing vaccine platforms, objectively compares the mechanisms by which Live Attenuated Vaccines (LAVs) and Inactivated Vaccines mimic natural infection and present antigens to the immune system. The distinction lies fundamentally in the replication competence of LAVs versus the non-replicating nature of inactivated antigens.
Core Mechanistic Comparison
Antigen Presentation Pathways
Live Attenuated Vaccines (LAVs) replicate within host cells, mimicking a natural infection. This leads to:
- Endogenous Antigen Presentation: Viral proteins are synthesized de novo in the cytosol of infected cells. These proteins are processed by the proteasome, and peptides are transported into the endoplasmic reticulum (ER) via TAP transporters. They are loaded onto MHC Class I molecules for presentation to CD8+ cytotoxic T lymphocytes (CTLs), inducing robust cellular immunity.
- Cross-Presentation: Antigens from infected cells can be taken up and presented by professional antigen-presenting cells (APCs) via the MHC Class I pathway, amplifying CTL responses.
- MHC Class II Presentation: Infected APCs or cells that take up debris from dead infected cells can also process antigens via the endolysosomal pathway, loading them onto MHC Class II for CD4+ T helper cell activation.
Inactivated Vaccines contain pathogens killed by heat or chemicals. They cannot replicate and primarily enter APCs via phagocytosis.
- Exogenous Antigen Presentation: The inactivated virus particles are internalized into endosomes, which fuse with lysosomes. Antigens are degraded into peptides within this endolysosomal compartment and loaded onto MHC Class II molecules. This potently activates CD4+ T helper cells, which support B cell maturation and antibody production.
- Limited MHC Class I Presentation: Without endogenous protein synthesis, inactivated vaccines are generally poor at entering the cytosolic pathway for MHC Class I presentation, resulting in weaker CD8+ T cell responses.
Diagram: Antigen Presentation Pathways for LAVs vs. Inactivated Vaccines
Comparative Immune Response Data
The mechanistic differences result in quantifiable disparities in immune responses, as supported by experimental data from model pathogens like influenza and measles.
Table 1: Comparative Immune Outcomes of LAV vs. Inactivated Vaccine Platforms
| Immune Parameter | Live Attenuated Vaccine (LAV) | Inactivated Vaccine | Key Supporting Experimental Evidence (Example) |
|---|---|---|---|
| CD8+ T Cell Response | High frequency, polyfunctional (IFN-γ+, TNF-α+, IL-2+). Establishes long-lived memory pool. | Weak or absent. Primarily cross-presentation dependent, low magnitude. | Intracellular cytokine staining (ICS) & MHC-I tetramer staining in murine influenza models show 10-100x higher antigen-specific CD8+ T cells post-LAV vs. inactivated. |
| CD4+ T Cell Response | Robust Th1 and often broader cytokine profiles. Supports both cellular & humoral arms. | Strong, often Th2-skewed. Excellent helper function for antibodies. | ELISpot assays measuring IL-4 (Th2) vs. IFN-γ (Th1) reveal Th1 bias for LAV (e.g., measles) vs. Th2 bias for inactivated (e.g., whole-cell pertussis). |
| Antibody Response | Typically high-affinity, durable IgG with strong mucosal IgA. | High-titer, systemic IgG. May require adjuvants for durability. | Plaque reduction neutralization tests (PRNT) show LAV (e.g., measles-mumps-rubella) induces lifelong neutralizing titers, while inactivated (e.g., influenza) titers wane within months. |
| Breadth of Response | Targets multiple internal and external antigens due to full viral replication. | Primarily targets surface/structural antigens present in the preparation. | Protein microarray analysis shows broader antibody epitope recognition following LAV vaccination. |
| Duration of Immunity | Often lifelong due to establishment of effector memory T cells and long-lived plasma cells. | Usually requires multiple boosters to maintain protective titers. | Longitudinal cohort studies of measles (LAV) vs. inactivated polio vaccine (IPV) demonstrate the superior durability of single-dose LAV immunity. |
Detailed Experimental Protocols
Protocol: Assessing MHC Class I vs. Class II-Restricted T Cell Responses
Objective: To quantify and characterize antigen-specific CD8+ and CD4+ T cell responses post-vaccination. Materials: See "The Scientist's Toolkit" below. Method:
- Immunization: Administer LAV or inactivated vaccine to mouse model (C57BL/6) or collect PBMCs from vaccinated humans.
- Antigen Re-stimulation (Day 7-14): Isolate splenocytes or PBMCs. Plate cells in 96-well plates.
- For CD8+ T cells: Stimulate with immunodominant MHC-I restricted peptide (e.g., NP366 for influenza).
- For CD4+ T cells: Stimulate with whole inactivated virus or MHC-II restricted peptides.
- Intracellular Cytokine Staining (ICS): Add protein transport inhibitor (Brefeldin A) for 4-6 hours. Fix, permeabilize, and stain for surface markers (CD3, CD8, CD4) and intracellular cytokines (IFN-γ, TNF-α, IL-2).
- Flow Cytometry: Acquire data on a flow cytometer. Gate on live, single CD3+CD8+ or CD3+CD4+ cells to quantify cytokine-positive populations.
- MHC Tetramer Staining: As a parallel approach, stain cells with fluorochrome-conjugated MHC-I peptide tetramers to directly quantify antigen-specific CD8+ T cells without stimulation.
Protocol: Evaluating Antibody Quality and Durability
Objective: To compare neutralizing antibody titers and affinity over time. Method:
- Serum Collection: Collect serial serum samples pre-vaccination and at defined intervals post-vaccination (e.g., day 28, month 6, year 1).
- Plaque Reduction Neutralization Test (PRNT): (For viruses that form plaques)
- Mix serial dilutions of heat-inactivated serum with a fixed amount of live, pathogenic virus.
- Incubate (1 hour, 37°C) and inoculate onto confluent cell monolayers (e.g., Vero cells).
- Overlay with semi-solid medium (e.g., carboxymethyl cellulose). Incubate until plaques form.
- Fix, stain, and count plaques. The PRNT50/PRNT90 titer is the serum dilution that reduces plaques by 50%/90% compared to virus control.
- Enzyme-Linked Immunosorbent Assay (ELISA):
- Coat plates with purified viral antigen.
- Add serial serum dilutions, followed by enzyme-conjugated anti-species secondary antibody and substrate.
- The endpoint titer is the highest dilution giving an optical density above a pre-defined cut-off.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Mechanistic Vaccine Studies
| Reagent / Material | Function in Experiment | Example Product / Assay |
|---|---|---|
| MHC Tetramers | Direct ex vivo staining and quantification of antigen-specific T cells via flow cytometry. | NIH Tetramer Core Facility; MBL International Peptide-MHC Tetramers. |
| Intracellular Cytokine Staining (ICS) Kits | Detect cytokine production (IFN-γ, TNF-α, IL-2, etc.) at the single-cell level after antigen re-stimulation. | BD Cytofix/Cytoperm; BioLegend True-Nuclear Transcription Factor Buffer Set. |
| ELISpot Kits | Enumerate individual cytokine-secreting cells (e.g., IFN-γ ELISpot for Th1/CTL response). | Mabtech IFN-γ ELISpotPRO; R&D Systems Human IL-4 ELISpot. |
| Protein Transport Inhibitors | Block cytokine secretion during ICS to allow intracellular accumulation (Brefeldin A, Monensin). | GolgiPlug (Brefeldin A); GolgiStop (Monensin) from BD Biosciences. |
| Fluorochrome-conjugated Antibodies | Surface and intracellular staining for flow cytometry (anti-CD3, CD4, CD8, CD19, CD69, etc.). | Clone-specific antibodies from BD Biosciences, BioLegend, Thermo Fisher. |
| Synthetic Peptide Pools | Cover entire viral proteome for broad T cell re-stimulation in ICS/ELISpot. | JPT PepMix; BEI Resources Peptide Arrays. |
| Neutralization Assay Components | Live virus, susceptible cell lines, and overlay media for PRNT/MN assays. | ATCC for virus/cell lines; Sigma for methylcellulose/carboxymethyl cellulose. |
| Adjuvant Controls | Essential for inactivated vaccine studies to separate antigen effects from adjuvant-driven immunity. | InvivoGen aluminum salts (Alum); Sigma CpG ODN 1826. |
Diagram: Experimental Workflow for Comparing T Cell Immunity
The mechanistic dichotomy between LAVs and inactivated vaccines—rooted in replication and antigen presentation pathways—dictates distinct immunological outcomes. LAVs, by mimicking natural infection, induce comprehensive, durable immunity involving strong CD8+ T cell responses. Inactivated vaccines efficiently drive antibody responses via MHC Class II presentation but typically fail to engage cytotoxic T cells. The choice of platform depends on the pathogen and the correlate of protection required, guiding rational vaccine design.
The development of vaccine platforms represents a cornerstone of modern medicine. This guide, framed within a thesis comparing live attenuated versus inactivated vaccine platforms, objectively traces their historical evolution, compares their performance, and details key experimental methodologies.
Historical Milestones: A Comparative Timeline
| Year | Live Attenuated Vaccine Milestone | Inactivated/Killed Vaccine Milestone |
|---|---|---|
| 1798 | - | Edward Jenner uses cowpox material (a live, heterologous virus) to create immunity to smallpox. |
| 1885 | Louis Pasteur develops the first lab-attenuated vaccine for rabies. | - |
| 1896 | - | Almroth Wright develops a killed whole-cell typhoid vaccine. |
| 1937 | Max Theiler develops the 17D yellow fever vaccine (live attenuated). | - |
| 1955 | - | Jonas Salk's formalin-inactivated polio vaccine (IPV) is licensed. |
| 1960s | Albert Sabin's oral polio vaccine (OPV, live attenuated) licensed. | - |
| 1970s | Live attenuated measles, mumps, and rubella (MMR) vaccines introduced. | - |
| 1980s | - | Advancements in purification lead to improved subunit (e.g., acellular pertussis) and polysaccharide-conjugate vaccines. |
| 2000s | Live attenuated influenza vaccine (LAIV) introduced. | Cell-culture based inactivated influenza vaccines developed. |
| 2020s | Intranasal COVID-19 vaccines (live attenuated) in clinical trials. | mRNA and adenovirus-vectored COVID-19 vaccines (functionally "inactivated" gene delivery) achieve global use. |
Performance Comparison: Key Parameters
The following table summarizes comparative performance data from immunological and clinical studies.
| Performance Parameter | Live Attenuated Vaccines | Inactivated Vaccines | Supporting Experimental Data / Meta-Analysis Findings |
|---|---|---|---|
| Immunogenicity Duration | Typically long-lasting (>10 years for many). | Often requires booster doses. | MMR vaccine shows >90% seropositivity after 20+ years. Tetanus toxoid (inactivated) requires boosters every 10 years. |
| Onset of Immunity | Rapid (1-2 weeks). | Slower, often requires multiple doses for full effect. | Single OPV dose induces intestinal immunity within days. IPV requires ≥2 doses for high seroconversion. |
| Mucosal Immunity | Strong. Induces secretory IgA at portals of entry. | Weak. Primarily systemic IgG response. | LAIV showed 55% greater efficacy than IIV in children due to mucosal immunity (meta-analysis, Pediatrics). |
| Cold Chain Requirement | Stringent (lyophilized forms help). | Less stringent (more stable). | Measles vaccine thermostability is a key logistic challenge in LMICs. |
| Safety Profile (General) | Contraindicated in immunocompromised. Rare reversion risk. | Generally safe for immunocompromised. No replication risk. | Vaccine-associated paralytic polio (VAPP) rate: ~1 case per 2.4 million OPV doses distributed. |
| Cellular Immune Response (CD8+ T cells) | Potent. Due to intracellular replication. | Weak/absent without novel adjuvants or vectors. | Yellow fever vaccine induces polyfunctional, long-lasting CD8+ T cell memory. |
| Manufacturing Complexity | Complex (viable organism, consistency of attenuation). | Relatively straightforward (kill, purify). | Consistency of attenuation for influenza LAIV is a key process control. |
Experimental Protocols for Key Comparisons
Protocol 1: Assessing Mucosal IgA Response
Objective: Compare mucosal immunogenicity of LAIV vs. Inactivated Influenza Vaccine (IIV). Methodology:
- Animal Model/Grouping: BALB/c mice (n=10/group). Groups: LAIV (intranasal), IIV (intramuscular), placebo.
- Immunization: Day 0 and Day 21.
- Sample Collection: Nasal washes and bronchoalveolar lavage (BAL) fluid collected on Day 35.
- Analysis: Mucosal IgA specific to influenza hemagglutinin (HA) measured by ELISA. Titers reported as endpoint dilution.
Protocol 2: Evaluation of CD8+ T Cell Memory
Objective: Quantify memory T cell responses induced by live attenuated vs. inactivated vaccine platforms. Methodology:
- Vaccine & Model: LCMV-based models (attenuated LCMV vs. UV-inactivated LCMV) in C57BL/6 mice.
- Immunization: Single dose.
- Analysis (Day 60+):
- Intracellular Cytokine Staining (ICS): Splenocytes stimulated with viral peptides, stained for IFN-γ, TNF-α, IL-2 in CD8+ T cells.
- Tetramer Staining: Direct quantification of antigen-specific CD8+ T cells.
- Challenge: Mice challenged with wild-type virus; viral load in organs quantified via plaque assay.
Visualization: Comparative Immunological Pathways
Title: Immune Activation Pathways of LAV vs Inactivated Vaccines
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Function in Comparative Vaccine Research |
|---|---|
| ELISpot Kits (IFN-γ, IL-4) | Quantify antigen-specific T cell responses at the single-cell level. Critical for comparing cellular immunity between platforms. |
| Multiplex Cytokine Assays (Luminex/MSD) | Profile broad panels of cytokines/chemokines from serum or culture supernatant to compare immune polarization. |
| MHC Tetramers/Dextramers | Precisely identify and sort antigen-specific T cells for functional analysis or transcriptomics. |
| Neutralization Assay Reagents (e.g., Reporter Viruses, Cell Lines) | Gold-standard for measuring functional, neutralizing antibody titers induced by different vaccines. |
| Pathogen-Specific Peptide Pools | Stimulate T cells in ICS or ELISpot assays to measure recall responses. |
| Mucosal Sampling Kits (e.g., Saliva/BAL Collection) | Standardize collection of mucosal secretions for IgA measurement. |
| Adjuvant Systems (e.g., Alum, AS01, CpG) | Essential for enhancing immunogenicity of inactivated/subunit vaccines in comparative studies. |
| Next-Generation Sequencing Kits | For BCR/TCR repertoire sequencing and transcriptional profiling of immune cells post-vaccination. |
Within the broader research on live attenuated versus inactivated vaccine platforms, understanding the distinct and complementary roles of cellular (T-cell) and humoral (B-cell) memory is paramount. This guide provides a comparative analysis of these two pillars of adaptive immunity, focusing on their induction, longevity, and functional outcomes, with supporting experimental data relevant to vaccine design.
Core Functional Comparison: T-cell vs. B-cell Memory
| Feature | Cellular Memory (T-cells) | Humoral Memory (B-cells) |
|---|---|---|
| Primary Effector Cells | Memory CD8+ Cytotoxic T Lymphocytes (CTLs), Memory CD4+ Helper T cells (Th1, Th2, Tfh). | Memory B cells, Long-lived Plasma Cells (LLPCs). |
| Molecular Mediator | Cytokines (e.g., IFN-γ, TNF, IL-2), cytotoxic granules (perforin, granzymes). | High-affinity, class-switched antibodies (IgG, IgA, IgE). |
| Primary Function | Direct killing of infected host cells (CD8+), orchestration of immune responses (CD4+). | Neutralization of pathogens/toxins, opsonization, complement activation. |
| Key Induction Signal | T-cell Receptor (TCR) engagement with peptide-MHC + co-stimulation (e.g., CD28:B7). | B-cell Receptor (BCR) engagement with antigen + T-cell help (CD40L:CD40, cytokines). |
| Memory Cell Lifespan | Long-lived, but may require intermittent antigen or cytokine signals. | Memory B cells: long-lived. LLPCs: can persist for decades in survival niches (e.g., bone marrow). |
| Response Kinetics | Rapid proliferation and effector function upon re-encounter. | Rapid differentiation into antibody-secreting plasma cells; pre-existing antibodies provide immediate protection. |
| Role in Vaccine Platforms | Live Attenuated: Strongly induced. Inactivated: Often weak/poorly induced without adjuvants. | Both: Induced. Live Attenuated: Often broader, durable antibody responses. Inactivated: Relies heavily on robust antibody induction. |
Quantitative Data from Comparative Studies
Table 1: Representative Data from Prime-Boost Vaccine Studies in Murine Models
| Parameter | Live Attenuated Vaccine Platform | Inactivated/Subunit Vaccine Platform |
|---|---|---|
| Antigen-Specific CD8+ T cell Frequency (IFN-γ+) | High (e.g., 5-15% of CD8+ T cells) | Low to undetectable (e.g., <1% of CD8+ T cells) |
| Memory CD4+ T cell (Tfh) Induction | Robust Germinal Center (GC) Tfh response | GC response variable, often adjuvant-dependent |
| Serum Neutralizing Antibody Titer (GMT) | High, sustained (e.g., log10 titer >3 for >6 months) | Moderate, may wane faster (e.g., log10 titer 2-3 at 6 months) |
| Long-Lived Plasma Cells in Bone Marrow | Abundant, stable (e.g., 100-500 cells/million) | Present, but may be fewer (e.g., 50-200 cells/million) |
| Protection Against Heterologous Challenge | Often strong due to broad T-cell recognition | May be limited, focused on antibody epitopes |
Note: GMT = Geometric Mean Titer. Data is illustrative, compiled from studies on influenza, measles, and SARS-CoV-2 vaccine platforms.
Experimental Protocols for Assessing Immunological Memory
Protocol 1: Intracellular Cytokine Staining (ICS) for Memory T-cell Function
- Objective: Quantify antigen-specific memory T-cells by cytokine production.
- Methodology:
- Isolate PBMCs or splenocytes from vaccinated hosts.
- Stimulate cells with peptide pools from the target antigen (e.g., 6-18 hours) in the presence of a protein transport inhibitor (e.g., Brefeldin A).
- Surface stain for CD3, CD4, CD8, CD44 (memory marker).
- Permeabilize cells, stain intracellularly for cytokines (IFN-γ, TNF, IL-2).
- Analyze by flow cytometry. Memory T-cells are identified as CD44high, cytokine-positive.
Protocol 2: Enzyme-Linked Immunosorbent Spot (ELISpot) for Antibody-Secreting Cells
- Objective: Enumerate antigen-specific memory B cells and antibody-secreting cells.
- Methodology:
- Coat ELISpot plates with the target antigen or anti-immunoglobulin capture antibody.
- Seed isolated lymphocytes (from blood, spleen, or bone marrow) into plates.
- Incubate (e.g., 24-48 hours) to allow antibody secretion and capture.
- Detect with enzyme-conjugated anti-IgG/IgA/IgM and a precipitating substrate.
- Count spots, each representing an antigen-specific antibody-secreting cell (ASC) or memory B cell (if polyclonally reactivated prior to assay).
Protocol 3: Adoptive Transfer & Challenge for Memory Function
- Objective: Functionally test the protective capacity of memory lymphocytes.
- Methodology:
- Purify memory phenotype T or B cells (e.g., CD44high for T cells) from vaccinated donor mice.
- Adoptively transfer cells into naïve, immunodeficient recipient mice.
- Challenge recipients with a live pathogen.
- Monitor outcomes: survival, pathogen load, or clinical score compared to controls.
Visualizations
T-cell Memory Generation Pathway
B-cell Memory & Antibody Production Pathway
Vaccine Platform Impact on Immune Memory
The Scientist's Toolkit: Key Research Reagents
Table 2: Essential Reagents for Immunological Memory Research
| Reagent/Solution | Function/Application | Example Targets |
|---|---|---|
| Fluorochrome-conjugated Antibodies | Phenotypic and intracellular staining for flow cytometry. | CD3, CD4, CD8, CD19, CD38, CD27, CD45RA/RO, CD44, CD62L, Cytokines. |
| Peptide Megapools / MHC Tetramers | Specific identification of antigen-reactive T-cells. | Viral peptide libraries, peptide-MHC I/II tetramers. |
| ELISpot Kits (IFN-γ, IgG) | Quantification of antigen-specific cytokine-secreting or antibody-secreting cells. | Coating antigen or capture antibody. |
| Cell Stimulation Cocktails | Polyclonal activation of T or B cells for functional assays. | PMA/Ionomycin, anti-CD3/CD28 beads, CpG + anti-Ig. |
| Cell Isolation Kits (Magnetic Beads) | Positive or negative selection of specific lymphocyte subsets. | CD4+ T cells, CD8+ T cells, Naïve/Memory B cells. |
| Adjuvants (for in vivo studies) | Enhance immunogenicity of inactivated/subunit vaccines. | Alum, MF59, AS01, CpG ODN. |
| Cell Trace Proliferation Dyes | Track cell division history upon re-stimulation. | CFSE, CellTrace Violet. |
This guide objectively compares the performance of major licensed live attenuated (LAV) and inactivated vaccines. The data is framed within ongoing research on platform immunogenicity, durability, and safety.
Comparative Performance Data: Key Licensed Vaccines
Table 1: Immunogenicity & Efficacy of Representative LAV vs. Inactivated Vaccines
| Vaccine (Platform) | Target Pathogen | Typical Seroconversion Rate (%) | Estimated Clinical Efficacy (%) | Durability of Protection | Key Efficacy Data Source |
|---|---|---|---|---|---|
| MMR (LAV) | Measles, Mumps, Rubella | >95 (Measles) | 97 (Measles) after 2 doses | Decades, often lifelong | CDC based on outbreak studies |
| Varicella (LAV) | Varicella-Zoster Virus | ~95 after 2 doses | 98 against severe disease | >10 years, likely longer | Kuter et al., 2004 (Pediatrics) |
| Polio (Salk) (Inactivated) | Poliovirus (Types 1-3) | >99 after 3 doses | 99-100 against paralytic disease | Long-term (>10 years) | Salk et al., 1954 (Field Trial) |
| Hepatitis A (Inactivated) | Hepatitis A Virus | ~100 after 2-dose series | >95 | At least 20-30 years | Werzberger et al., 1992 (NEJM) |
| Seasonal Influenza (IIV) (Inactivated) | Influenza Virus | 40-60 (strain-dependent) | 40-60% VE annually | 6-8 months | Annual CDC estimates |
| Influenza (LAIV) (LAV) | Influenza Virus | Induces mucosal IgA | Variable by season/age | Seasonal | Often comparable to IIV in children |
Table 2: Safety & Reactogenicity Profile Comparison
| Vaccine Platform | Common Local Reactions | Systemic Reactions | Rare Severe Risks | Contraindications |
|---|---|---|---|---|
| Live Attenuated (e.g., MMR, Varicella) | Injection site pain, rash | Fever, rash, mild viral symptoms | Vaccine-strain disease (immunocompromised), Encephalitis (extremely rare) | Pregnancy, immunodeficiency, recent antibody products |
| Inactivated (e.g., Polio (IPV), Hep A) | Pain, erythema, swelling | Fever, malaise, headache | Anaphylaxis (very rare) | Severe allergic reaction to prior dose or component |
Experimental Protocols: Key Methodologies for Platform Comparison
Protocol 1: Measuring Neutralizing Antibody Titers (Primary Immunogenicity Readout)
- Sample Collection: Obtain serum samples pre-vaccination (Day 0) and post-vaccination (e.g., Day 28, 1 year).
- Virus & Cell Culture: Prepare working stocks of target virus (e.g., measles Edmonston strain, poliovirus Sabin strain) and susceptible cell lines (e.g., Vero cells).
- Serum Serial Dilution: Perform 2-fold serial dilutions of heat-inactivated serum in duplicate in a 96-well plate.
- Virus Neutralization: Mix an equal volume of virus suspension (containing ~100 TCID50) with each serum dilution. Incubate at 37°C for 1-2 hours.
- Cell Inoculation: Transfer the virus-serum mixture to a monolayer of cells. Include virus-only and cell-only controls.
- Incubation & Detection: Incubate plates for an appropriate period (5-7 days). Monitor for cytopathic effect (CPE).
- Titer Calculation: The neutralizing antibody titer is the reciprocal of the highest serum dilution that inhibits CPE in 50% of wells (TCID50), calculated using the Spearman-Kärber method.
Protocol 2: Assessment of Cell-Mediated Immunity (ELISpot for IFN-γ)
- PBMC Isolation: Isplicate peripheral blood mononuclear cells (PBMCs) from heparinized blood via Ficoll-Paque density gradient centrifugation.
- Antigen Stimulation: Seed PBMCs into an IFN-γ pre-coated ELISpot plate. Stimulate with vaccine-specific peptides, pooled viral lysate (for inactivated platforms), or live attenuated virus at low MOI (for LAV platforms). Include positive (PHA) and negative (media) controls.
- Incubation: Incubate plates at 37°C, 5% CO2 for 24-48 hours.
- Detection: Follow manufacturer's protocol for detection of captured IFN-γ (typically biotinylated detection Ab, streptavidin-enzyme conjugate, and precipitating substrate).
- Analysis: Enumerate antigen-specific spot-forming cells (SFCs) per 10^6 PBMCs using an automated ELISpot reader.
Visualizations: Platform Mechanisms & Study Design
Diagram 1: LAV vs Inactivated Immune Activation Pathways
Diagram 2: Comparative Immunogenicity Study Workflow
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for Vaccine Platform Comparison Studies
| Reagent / Material | Function in Research | Example / Specification |
|---|---|---|
| Vero Cells (ATCC CCL-81) | Permissive cell line for culturing viruses (measles, polio, influenza) and performing virus neutralization assays. | Must be mycoplasma-free. |
| RD Cells (ATCC CCL-136) | Used specifically for poliovirus isolation and titrations. | |
| Virus Reference Reagents | Standardized virus stocks for neutralization assays (e.g., Measles Edmonston, Polio Sabin Types 1-3). | Obtained from WHO Collaborating Centers or ATCC. |
| Recombinant Viral Proteins | Antigens for ELISA to measure binding antibodies (e.g., Measles hemagglutinin (H) protein). | Ensure correct conformation for neutralizing epitopes. |
| Peptide Pools (JPT, etc.) | Overlapping peptides spanning vaccine antigens for stimulating T-cells in ELISpot or intracellular cytokine staining. | 15-mer peptides overlapping by 11 aa. |
| Human IFN-γ ELISpot Kit (Mabtech) | Pre-coated plates and paired antibodies for quantifying antigen-specific T-cell responses. | Includes capture Ab, detection Ab, streptavidin-enzyme. |
| Multiplex Cytokine Assay (Luminex) | To profile Th1/Th2/Th17 cytokine profiles (IFN-γ, IL-2, IL-4, IL-5, IL-17) post-stimulation. | 10-25 plex panels. |
| Complement (Guinea Pig Serum) | Source of complement required for some traditional neutralization assays (e.g., for measles). | Must be screened for low toxicity. |
| Standardized Human Reference Sera (NIBSC) | Positive control sera with assigned international units (IU) for neutralizing antibodies. | Critical for assay standardization. |
From Bench to Bioreactor: Development, Production, and Clinical Deployment Strategies
Within the broader research on live attenuated versus inactivated vaccine platforms, the method of pathogen attenuation is a critical determinant of safety and immunogenicity. This guide compares three core attenuation techniques—Serial Passage, Reverse Genetics, and Targeted Mutagenesis—used in the development of modern live attenuated vaccines (LAVs). The focus is on their mechanistic principles, experimental performance in generating vaccine candidates, and practical application in research.
Comparative Analysis of Attenuation Techniques
The table below summarizes the key characteristics and experimental outcomes of each technique based on recent studies (2023-2024).
Table 1: Comparison of Attenuation Techniques for Live Attenuated Vaccine Development
| Feature | Serial Passage | Reverse Genetics | Targeted Mutagenesis (e.g., Codon Deoptimization) |
|---|---|---|---|
| Underlying Principle | Empirical adaptation to suboptimal growth conditions; accumulation of random mutations. | Rational assembly of viral genome from cloned cDNA; introduction of specific attenuating mutations. | Rational redesign of genomic regions to reduce translational efficiency via synonymous codon changes. |
| Time to Candidate (Typical) | 12-36 months | 3-9 months | 6-12 months |
| Genetic Stability | Variable; can revert or compensate. | High, if mutations are stable. | Very High; reversion is statistically negligible. |
| Example Vaccine/Strain | Historical: Sabin Polio, MMR. Recent: CAF-dengue (CYD-TDV) foundation. | FluMist (AAV), RSV LID/ΔM2-2 candidates, COVI-VAC (NDV-based COVID-19 candidate). | eOPV2 (type 2 novel oral polio vaccine), SARS-CoV-2 candidate CV2. |
| Reported Immunogenicity (Animal Model) | Strong, broad immune response often mimicking natural infection. | Can be tailored; sometimes requires balancing attenuation and immunogenicity. | Strong humoral and cellular responses; antigenicity preserved. |
| Safety Profile (Reversion Risk) | Moderate to High (risk of reversion to virulence). | Low to Moderate (controlled by known mutations). | Very Low (genetic "firewalls" prevent precise reversion). |
| Key Quantitative Data (Recent Studies) | Passage 50: 4-log reduction in wild-type virulence titer in vivo. | Single gene segment swap (PB2) led to 1000x reduction in lung titer vs. wild-type in mice. | 564 synonymous mutations yielded a 10^5 reduction in replication in neural cells. |
Detailed Experimental Protocols
Protocol 1: Serial Passage for Attenuation
Aim: To attenuate a virulent virus by sequential propagation in non-human cells or at suboptimal temperatures. Methodology:
- Starting Material: Isolate wild-type virus from a clinical specimen.
- Host System: Inoculate the virus into a non-natural host system (e.g., Vero cells for arboviruses, primary chicken embryo fibroblasts for influenza).
- Passaging: Harvest virus from infected cells/supernatant at peak cytopathic effect (CPE) or a fixed time post-infection (e.g., 72h). Use a defined dilution (e.g., 1:100 to 1:1000) to inoculate fresh cells. Repeat for 50-100 passages.
- Monitoring: Sequence the viral genome at every 10th passage to track mutation accumulation. Assess attenuation by comparing pathogenicity (LD50/TCID50) of passaged virus vs. wild-type in an animal model (e.g., ferrets, mice) at passages 0, 30, 50, and 100.
- Candidate Selection: Select a passage number where virulence is significantly reduced but replication in vitro remains robust (e.g., >1x10^6 PFU/ml).
Protocol 2: Reverse Genetics for Attenuated Strain Rescue
Aim: To generate a live attenuated virus by rescuing an infectious virus from plasmid DNA encoding designed attenuating mutations. Methodology:
- Plasmid System: Use an 8-plasmid or 12-plasmid system (for segmented viruses like influenza) where each viral genomic segment is cloned under a polymerase I (or II) promoter.
- Mutagenesis: Introduce specific, known attenuating mutations (e.g., deletions in NS1, mutations in polymerase complex genes) into relevant plasmids via site-directed mutagenesis.
- Transfection: Co-transfect the full set of plasmids into a permissive cell line (e.g., HEK-293T or Vero). Include plasmids expressing viral polymerase proteins if required.
- Rescue & Harvest: Incubate for 48-72 hours. Supernatant is then harvested and used to inoculate embryonated chicken eggs or a high-density cell culture (e.g., MDCK) for amplification.
- Phenotypic Validation: Sequence the entire rescued virus genome to confirm mutations. Titrate via plaque assay. Assess attenuation in vitro (e.g., plaque size reduction) and in vivo (e.g., lung viral load reduction in mice vs. wild-type control).
Protocol 3: Targeted Mutagenesis via Synthetic Codon Pair Deoptimization
Aim: To attenuate a virus by recoding viral genes with suboptimal codon pairs without altering the amino acid sequence. Methodology:
- Gene Selection: Identify one or two essential viral genes for recoding (e.g., capsid or polymerase genes).
- Sequence Design: Use algorithms to redesign the selected gene segment(s) to maximize the use of underrepresented codon pairs in the host genome, while preserving the amino acid sequence and avoiding cis-acting regulatory elements.
- Gene Synthesis: Chemically synthesize the full-length deoptimized gene fragment.
- Infectious Clone Assembly: Replace the wild-type gene in a full-length infectious cDNA clone with the deoptimized version using Gibson Assembly or restriction enzyme cloning.
- Virus Recovery & Characterization: Recover virus by transfection (as in Reverse Genetics). Key experiments include:
- Growth Kinetics: Multi-step growth curve in permissive cells vs. wild-type (samples at 0, 12, 24, 48, 72h p.i.).
- Translational Efficiency: Dual-luciferase reporter assay comparing deoptimized vs. wild-type sequences.
- Safety: Intracranial challenge in suckling mice or infection in severe immunodeficient mice to assess residual neurovirulence.
Visualization of Techniques and Workflows
Diagram Title: Attenuation Technique Workflow Comparison
Diagram Title: Safety-Immunogenicity Balance in LAV Development
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Attenuation Technique Research
| Reagent / Solution | Primary Function | Example Use Case |
|---|---|---|
| Plaque Assay Kit | Quantify infectious virus titer (PFU/ml) via plaque formation in cell monolayers. | Titrating virus harvests from serial passage or rescued virus from reverse genetics. |
| Site-Directed Mutagenesis Kit | Introduce precise point mutations into plasmid DNA. | Engineering specific attenuating point mutations in reverse genetics plasmids. |
| Viral cDNA Clone System | Full-length plasmid system for viral genome rescue. | Backbone for reverse genetics and targeted mutagenesis (e.g., influenza 8-plasmid system). |
| Codon Optimization/Deoptimization Software | Algorithmic design of gene sequences with altered codon usage bias. | Designing deoptimized gene fragments for targeted mutagenesis attenuation. |
| Transfection Reagent (Polymer/Lipid-based) | Deliver plasmid DNA into mammalian cells with high efficiency. | Co-transfection of multiple plasmids for virus rescue in reverse genetics. |
| Next-Generation Sequencing (NGS) Service | High-throughput deep sequencing of entire viral genomes. | Monitoring mutation accumulation during serial passage; confirming genome of rescued virus. |
| Immunodeficient Mouse Model (e.g., STAT1 -/-) | Highly susceptible animal model for virulence testing. | Assessing the level of attenuation of candidate strains (safety profiling). |
| Dual-Luciferase Reporter Assay System | Measure translational efficiency of recoded gene sequences. | Validating the mechanism of attenuation for codon-deoptimized viruses. |
Within the critical research comparing live attenuated versus inactivated vaccine platforms, the selection of an inactivation method is a fundamental determinant of final product characteristics. Chemical agents like formalin and beta-propiolactone (BPL), and physical processes such as heat and radiation, are employed to destroy pathogen replicative capacity while preserving immunogenic structures. This guide provides an objective, data-driven comparison of these core inactivation techniques, framed within modern vaccine development.
Comparative Analysis of Inactivation Methods
Mechanism of Action & Key Characteristics
| Method | Primary Mechanism | Typical Application Context | Critical Control Parameters |
|---|---|---|---|
| Formalin | Cross-links proteins and nucleic acids via alkylation. | Whole-virus vaccines (e.g., Polio, Hepatitis A), tissue specimens. | Concentration (0.1-0.5%), temperature, duration, pH, reactant ratio. |
| Beta-Propiolactone (BPL) | Alkylates nucleic acids (primarily guanine); less protein cross-linking. | Cell culture-based vaccines (Rabies, Influenza), blood plasma products. | Concentration (<0.1%), temperature (4°C preferred), hydrolysis rate, pH. |
| Heat | Protein denaturation and aggregation, nucleic acid damage. | Lyophilized products, plasma derivatives, some bacterial toxoids. | Temperature (55-60°C common), exposure time, moisture content, matrix. |
| Radiation (Gamma/E-beam) | Ionizing radiation generates free radicals, damaging nucleic acids. | Serum, tissue grafts, pre-filled syringes, thermolabile products. | Dose (kGy), dose rate, temperature during irradiation, presence of radical scavengers. |
Table 1: Comparative Inactivation Efficacy & Antigen Integrity
| Method | Log Reduction in Infectivity (Virus Model) | Impact on Antigenicity | Residual Risk of Reversion | Typical Processing Time |
|---|---|---|---|---|
| Formalin | >8 log10 (e.g., Poliovirus) | Moderate-High (can mask epitopes) | None | Days to Weeks |
| Beta-Propiolactone | >6 log10 (e.g., Influenza virus) | High (preserves conformational epitopes) | None (hydrolyzes to non-toxic) | Hours to Days |
| Heat (Moist) | Variable (3-7 log10) | Low-Moderate (high denaturation) | None | Minutes to Hours |
| Gamma Radiation | >6 log10 (e.g., Vesicular Stomatitis Virus) | Variable (depends on dose & freeze state) | None | Minutes to Hours |
Detailed Experimental Protocols
Protocol 1: Formalin Inactivation Kinetics Study
Objective: To determine the time-concentration relationship for complete inactivation of an enveloped virus while monitoring antigen integrity. Materials: Virus stock, 37% formaldehyde solution, inactivation buffer (pH 7.2), cell culture for infectivity assay, ELISA plates for antigenicity. Procedure:
- Dilute virus in buffer to a standardized titer (e.g., 10^8 PFU/mL).
- Add formalin to final concentrations of 0.01%, 0.05%, and 0.1% (v/v) in separate batches.
- Incubate at 36°C. Withdraw aliquots at 0, 6, 12, 24, 48, 72, and 96 hours.
- Immediately neutralize residual formalin in aliquot with sodium bisulfite.
- Infectivity Assay: Perform plaque assay or TCID50 on treated aliquots.
- Antigenicity Assay: Quantify binding to known neutralizing monoclonal antibodies via ELISA.
- Plot log10 infectivity and relative antigenicity vs. time for each concentration.
Protocol 2: Beta-Propiolactone (BPL) Inactivation Optimization
Objective: To inactivate a RNA virus at low temperature to preserve hemagglutinin (HA) and neuraminidase (NA) antigenic function. Materials: Virus harvest, Beta-Propiolactone (cold), phosphate buffer (4°C), water bath, hydrolysis termination bath (37°C). Procedure:
- Chill virus suspension to 4°C. Prepare fresh 1% BPL stock in cold PBS.
- Add BPL to virus to a final concentration of 0.01%-0.05%. Maintain at 4°C with gentle stirring.
- Hold for 18-24 hours. Transfer an aliquot to a 37°C bath for 2 hours to hydrolyze residual BPL.
- Confirm complete hydrolysis by pH stabilization.
- Test for residual infectivity in embryonated eggs or cell culture.
- Assess antigen quality by SRID (Single Radial Immunodiffusion) for HA quantification and NA enzymatic activity assay.
Protocol 3: Gamma Irradiation Dose-Response
Objective: To establish the minimum sterilizing dose (D-value) for a bacterial pathogen in a liquid matrix. Materials: Bacterial suspension, sealed vials, Gamma irradiator (Co-60 source), colony counting equipment. Procedure:
- Prepare high-titer bacterial suspension (~10^9 CFU/mL). Dispense into identical sealed vials.
- Irradiate triplicate vials at target doses (e.g., 1, 2, 3, 5, 10 kGy). Use dosimetry to confirm absorbed dose.
- After irradiation, perform serial dilution and plate counts for each vial.
- Plot log10 surviving fraction against radiation dose (kGy).
- The D-value (dose required for 1-log reduction) is the negative reciprocal of the slope.
- Perform SDS-PAGE/Western blot on samples to assess structural protein damage at key doses.
Visualizations
Title: Decision Workflow for Pathogen Inactivation Method Selection
Title: Molecular Mechanisms of Inactivation Methods
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Inactivation Studies
| Reagent / Material | Function & Role in Research | Key Consideration |
|---|---|---|
| Neutralizing Agents (e.g., Sodium bisulfite, Glycine) | Quench residual chemical inactivator post-treatment to stop reaction and allow accurate infectivity titration. | Must be validated for complete neutralization without damaging the pathogen antigen. |
| Cell Lines / Embryonated Eggs | Provide the substrate for residual infectivity assays (Plaque, TCID50, EID50). Critical for proving inactivation. | Must be permissive to the wild-type pathogen; assess sensitivity. |
| Specific Antibodies (mAbs, Polyclonals) | Used in ELISA, SRID, Western Blot to quantify preservation of key antigenic epitopes post-inactivation. | Conformational vs. linear epitope recognizing antibodies give different information. |
| Dosimeters (for Radiation) | Measure the actual absorbed radiation dose (kGy) during irradiation experiments. Essential for reproducible D-value calculation. | Chemical (e.g., Fricke) or physical (alanine) dosimeters placed within the sample matrix. |
| Stabilizer / Cryoprotectant Solutions (e.g., Sucrose, MgCl2) | Protect antigenic structure during harsh physical treatments like heat or radiation, allowing isolation of inactivation effect. | Optimize concentration to stabilize without interfering with the inactivation mechanism. |
| Real-Time PCR / RT-PCR Kits | Quantify genomic copy number reduction as a surrogate or complement to infectivity assays, especially for slow-growing agents. | Cannot distinguish infectious from inactivated particles; must correlate with functional assays. |
The choice between chemical (formalin, BPL) and physical (heat, radiation) inactivation is not merely a binary selection but a strategic decision with profound implications for antigen quality, safety, and downstream processing in inactivated vaccine manufacturing. While chemical methods offer fine control and are well-suited for labile antigens, physical methods provide speed and lack chemical residuals. Robust, multi-assay protocols are non-negotiable to confirm both the elimination of infectivity and the conservation of protective immunogenicity, directly informing the central thesis comparison with live-attenuated platforms. The optimal method is pathogen-specific and must be empirically determined within the context of the final vaccine formulation and production scale.
This guide compares manufacturing processes for live attenuated (LAV) and inactivated (IV) vaccine platforms, providing critical data for platform selection within broader vaccine research.
Comparison of Upstream Cell Culture Systems
Upstream processes focus on antigen production. Key differences lie in biocontainment, cell viability requirements, and harvest timing.
Table 1: Upstream Process Comparison for Vero Cell-Based Production
| Parameter | Live Attenuated Vaccine (e.g., Influenza) | Inactivated Vaccine (e.g., Inactivated Polio Vaccine) |
|---|---|---|
| Production Cell Line | Vero, MRC-5, or primary chick embryo cells | Typically Vero or PER.C6 cells |
| Bioreactor Mode | Batch or Fed-Batch | Often Perfusion to achieve high cell density |
| Infection Multiplicity (MOI) | Low (0.001-0.01) to maximize yield/virus | High (3-10) to synchronize infection |
| Time to Harvest | 48-72 hours post-infection | 96-144+ hours post-infection/culture |
| Cell Viability at Harvest | <50% (due to cytopathic effect) | >80% (virus harvested from supernatant) |
| Key Process Control | Monitor viral titer (TCID50/mL) | Monitor antigen titer (DU/mL or HA/mL) |
| Biosafety Level | BSL-2 or higher required | BSL-1 often sufficient post-inactivation |
Experimental Protocol: Viral Titer and Antigen Yield in Bioreactors
Objective: Quantify infectious virus (LAV) and total antigen (IV) yields from a 5L benchtop bioreactor. Method:
- Cell Culture: Seed Vero cells in a 5L bioreactor at 2x10^5 cells/mL in serum-free media. Maintain at 37°C, pH 7.2, DO at 40%.
- Infection: At 90% confluence, infect culture at defined MOI.
- Sampling: Take 10 mL samples every 12 hours.
- LAV Assay: For LAV, measure infectious titer via TCID50 assay on 96-well plates of fresh Vero cells. Calculate using the Spearman-Kärber method.
- IV Assay: For IV (pre-inactivation), quantify total viral protein via ELISA or determine antigen units (DU) by SDS-PAGE densitometry against a standard.
- Metabolites: Analyze glucose/lactate levels from the same samples. Data Analysis: Plot titer/antigen vs. time to determine optimal harvest window.
Title: Upstream Workflow for LAV and IV Manufacturing
Downstream Purification Strategy Comparison
Downstream processes must maintain infectivity (LAV) or preserve antigenic structure (IV) while removing host cell impurities.
Table 2: Purification Unit Operation Performance
| Unit Operation | Live Attenuated Vaccine Goal & Yield | Inactivated Vaccine Goal & Yield | Key Supporting Data |
|---|---|---|---|
| Benzonase Treatment | Reduce DNA; >90% reduction | Reduce DNA; >95% reduction | Host cell DNA <10 ng/dose (WHO spec) |
| Ultra/Diafiltration (TFF) | Buffer exchange; infectivity loss <0.5 log | Concentrate and buffer exchange; recovery >85% | LAV: Recovery of 80-90% infectious units. IV: 8-10x concentration factor. |
| Chromatography | Often ion-exchange (AEX) to remove aggregates. Harsh resins avoided. | Multi-modal: AEX, CEX, HIC common. Tolerates broader conditions. | LAV: AEX yields ~70% with 3-log HCP reduction. IV: CEX yields >80% with 4-log HCP reduction. |
| Sterile Filtration | 0.22 µm filtration post-purification. Potential titer loss. | Standard 0.22 µm filtration with minimal loss. | LAV: Up to 0.3 log titer loss. IV: >99.5% protein recovery. |
Experimental Protocol: Purification Step Recovery Analysis
Objective: Measure recovery and impurity clearance across a purification train. Method:
- Load: Characterize clarified harvest for total protein (Bradford), DNA (PicoGreen), HCP (ELISA), and relevant potency (TCID50 for LAV, ELISA for IV).
- Step Processing: Perform each unit operation (e.g., TFF, AEX) at benchtop scale.
- Collection: Analyze product pool after each step for the same attributes.
- Calculations:
- Step Recovery (%) = (Potency in pool / Potency in load) * 100.
- Clearance Factor = Impurity in load / Impurity in pool. Data Analysis: Create a purification process mass balance table.
Title: Divergent Downstream Purification Paths for LAV vs. IV
Formulation and Stability Comparative Data
Formulation goals diverge significantly: LAV requires thermal stability maintenance of live virus, while IV focuses on preserving antigen structure and adjuvanticity.
Table 3: Formulation Component Impact on Vaccine Stability
| Formulation Aspect | Live Attenuated Vaccine | Inactivated Vaccine | Experimental Evidence (Accelerated Stability) |
|---|---|---|---|
| Stabilizing Sugars | Sucrose, Trehalose (0.5-2M) critical for lyophilization. | Sucrose (5-10%) often used, less critical. | LAV: Lyophilized with trehalose retains >80% potency after 4 weeks at 37°C. IV: Liquid formulation retains >95% antigenicity. |
| Buffer System | Histidine, Tris near neutral pH. | Phosphate, Succinate common. May vary by adjuvant. | LAV: pH 7.2-7.6 optimal for infectivity. IV: pH stability range broader (6.0-8.0). |
| Surfactants | Low concentration PS80 (0.005-0.01%) to prevent aggregation. | PS80 (0.01-0.1%) used, especially with adjuvants. | LAV: Higher PS80 (>0.02%) can reduce titer. |
| Key Challenge | Thermostability. Liquid forms often require -20°C storage. | Adjuvant Compatibility. Antigen must remain stable and unaggregated in adjuvant matrix. | LAV: Lyophilization improves shelf-life at 2-8°C. IV: Antigen-adjuvant adsorption kinetics (e.g., >90% adsorption to Alum in 60 min) is critical. |
Experimental Protocol: Accelerated Stability Study
Objective: Compare stability of LAV and IV candidate formulations. Method:
- Formulate: Prepare LAV (liquid and lyophilized) and IV (liquid, adjuvanted) in candidate buffers.
- Stress Conditions: Store samples at 4°C (control), 25°C, and 37°C. Sample at 0, 1, 2, 4 weeks.
- Assay: For LAV, measure infectious titer (TCID50). For IV, measure antigen content (ELISA) and integrity (SEC-HPLC for aggregates).
- Analysis: Plot % initial potency vs. time. Calculate degradation rate constants (k) for each condition. Key Outcome: Determine if degradation follows zero or first-order kinetics.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Materials for Process Comparison Studies
| Item | Function & Application in LAV/IV Studies | Example Vendor/Product |
|---|---|---|
| Serum-Free Cell Culture Medium | Supports growth of Vero, MRC-5, or PER.C6 cells in bioreactors. Essential for consistent upstream production. | Gibco VP-SFM, HyClone CDM4PERMab |
| Benzonase Nuclease | Degrades host cell DNA/RNA to reduce viscosity and improve downstream purification. Used for both platforms. | Sigma-Aldrich (Merck) |
| Tangential Flow Filtration (TFF) Cassette | For concentration and buffer exchange of viral harvests or inactivated antigen. Molecular weight cutoff (MWCO) is platform-specific. | Pall Corporation (Viresolve), Sartorius (Hydrosart) |
| Chromatography Resins | AEX (e.g., Capto Q) for LAV purification. CEX (e.g., Capto S) and HIC for IV purification. | Cytiva |
| Host Cell Protein (HCP) ELISA Kit | Quantifies process-related impurity clearance, critical for demonstrating downstream purity for both platforms. | Cygnus Technologies (Vero HCP Kit) |
| Stabilization/Sugar Matrix | Sucrose, Trehalose for LAV lyophilization. Provides a stable cake for long-term storage. | Pfanstiehl Sucrose (USP) |
| Adjuvant System | Aluminum hydroxide (Alum), AS01, etc., used with IV platforms to enhance immunogenicity. Not used with LAVs. | InvivoGen Alhydrogel, GSK Adjuvant Systems |
| Real-Time Cell Analysis (RTCA) System | Monitors cell proliferation and cytopathic effect (CPE) in real-time for upstream process optimization. | Agilent xCELLigence |
In the comparative research of live attenuated versus inactivated vaccine platforms, a fundamental challenge is the inherently lower immunogenicity of inactivated vaccines. While live attenuated vaccines often mimic natural infection, stimulating robust and durable adaptive immunity, inactivated antigens typically require adjuvants to achieve comparable protective efficacy. This guide compares the performance of major adjuvant classes in enhancing the immunogenicity of inactivated vaccines, supported by experimental data.
Comparative Performance of Adjuvant Classes for Inactivated Vaccines Table 1: Summary of Adjuvant Mechanisms and Immunogenic Outcomes
| Adjuvant Class / Example | Key Mechanism of Action (MOA) | Typical Humoral Response (Antigen-Specific IgG Titer) | Typical Cellular Response (CD8+ T-cell Frequency) | Key Advantages | Reported Limitations |
|---|---|---|---|---|---|
| Alum (Aluminum Salts) | NLRP3 inflammasome activation; depot formation; Th2 bias. | ~10⁵ - 10⁶ (Moderate, Th2-skewed) | Low to undetectable | Extensive safety record; stable formulation. | Weak cell-mediated immunity (CMI); potential for IgE. |
| MF59 (Squalene Emulsion) | Enhanced antigen uptake & recruitment of immune cells to injection site; broader cytokine/chemokine induction. | ~10⁶ - 10⁷ (High, broader isotype profile) | Moderate (primarily CD4+ Th1) | Enhanced antibody titers in elderly (clinical); good safety profile. | Limited potency for strong Th1/CTL induction. |
| AS01 (MPL + QS-21) | MPL (TLR4 agonist) activates APCs; QS-21 promotes cytosolic antigen release. | ~10⁷ - 10⁸ (Very High) | High (Strong Th1 & CD8+ T cells) | Powerful induction of CMI and humoral immunity. | Higher reactogenicity; complex formulation. |
| CpG 1018 (TLR9 Agonist) | TLR9 activation in B cells and pDCs; strong Th1/IFN-γ bias. | ~10⁶ - 10⁷ (High, Th1-skewed) | High (Strong Th1 & CD8+ T cells) | Potent Th1/CTL shift; can be used with Alum. | Potential for autoimmunity with chronic use. |
| AddaVax (Squalene Emulsion) | Similar to MF59; enhances antigen presentation and lymph node trafficking. | ~10⁶ - 10⁷ (High) | Moderate | Research-grade equivalent to MF59. | Similar to MF59. |
Experimental Protocol: Comparison of Adjuvant Efficacy Objective: To compare the humoral and cellular immune responses elicited by an inactivated viral antigen (e.g., SARS-CoV-2 Spike protein) formulated with Alum, MF59, or AS01-like adjuvant. Methodology:
- Groups & Immunization: C57BL/6 mice (n=10/group) are immunized intramuscularly (Days 0, 21) with 5 µg of inactivated antigen alone or adsorbed/formulated with:
- Group 1: Antigen + Alum (200 µg)
- Group 2: Antigen + MF59 (50% v/v, 50 µL final)
- Group 3: Antigen + MPL (5 µg) + QS-21 (5 µg) (AS01 mimic)
- Group 4: Antigen only (control)
- Sample Collection: Serum is collected on Day 35 for antibody analysis. Spleens are harvested on Day 38 for cellular assays.
- Humoral Response: Antigen-specific IgG, IgG1, and IgG2c titers are measured by ELISA. Neutralization titers are assessed using a pseudovirus neutralization assay (PNT).
- Cellular Response: Splenocytes are stimulated with antigen peptide pools. IFN-γ and IL-5 are measured by ELISpot. Antigen-specific CD4+ and CD8+ T cells are characterized by intracellular cytokine staining (ICS) and flow cytometry (IFN-γ, TNF-α, IL-2).
- Data Analysis: Statistical significance is determined by one-way ANOVA with Tukey’s post-hoc test.
Experimental Workflow and Key Signaling Pathways
The Scientist's Toolkit: Key Research Reagent Solutions Table 2: Essential Materials for Adjuvant Comparison Studies
| Reagent / Material | Supplier Examples | Function in Experiment |
|---|---|---|
| Inactivated Antigen (e.g., Recombinant Spike Protein) | Sino Biological, Novoprotein, | The target immunogen; basis for formulating with adjuvants. |
| Alum (Alhydrogel) | InvivoGen, Brenntag | Classic Th2-biased adjuvant control; adsorbed to antigen. |
| MF59-like Emulsion (AddaVax) | InvivoGen | Ready-to-use oil-in-water emulsion; mimics clinical adjuvant MF59. |
| TLR Agonists (MPL, CpG ODN) | InvivoGen, Sigma-Aldrich | Molecular adjuvants to skew towards Th1/CTL responses. |
| ELISA Kit (Mouse IgG/IgG1/IgG2c) | Thermo Fisher, Abcam, Mabtech | Quantifies antigen-specific antibody isotypes and titers in serum. |
| ELISpot Kit (Mouse IFN-γ/IL-5) | Mabtech, BD Biosciences | Measures frequency of antigen-specific cytokine-producing T cells. |
| Flow Cytometry Antibodies (CD3, CD4, CD8, IFN-γ, TNF-α) | BioLegend, BD Biosciences, Thermo Fisher | Phenotypes and characterizes antigen-specific T cell responses via ICS. |
| Pseudovirus Neutralization Assay Kit | Integral Molecular, AcroBiosystems | Assesses functional, neutralizing antibody response in serum. |
Within the broader thesis on the comparison of live attenuated (LAV) versus inactivated vaccine platforms, this guide objectively evaluates platform suitability. The choice hinges on a complex interplay between pathogen biology (viral vs. bacterial), desired immune response, and target population characteristics.
Core Platform Comparison: LAV vs. Inactivated Vaccines
Table 1: Fundamental Platform Characteristics and Immune Profile
| Feature | Live Attenuated Vaccine (LAV) | Inactivated Vaccine (Killed) |
|---|---|---|
| Platform Definition | Pathogen weakened to lose pathogenicity but retain replication capacity. | Pathogen killed by heat/chemicals; cannot replicate. |
| Typical Immune Response | Strong, durable; robust CD8+ T-cell & antibody (IgG, IgA) response. | Primarily antibody-mediated (IgG); weaker CD8+ T-cell response. |
| Pathogen Suitability | Primarily viral (e.g., Measles, Yellow Fever). Some bacterial (e.g., BCG). | Both viral (e.g., Influenza, IPV) and bacterial (e.g., Pertussis whole-cell). |
| Doses Required | Often 1-2 doses (long-lived memory). | Often require multiple doses & boosters (prime-boost). |
| Onset of Protection | ~2-3 weeks (time for limited replication). | Faster humoral response post-boost. |
| Cold Chain Requirement | Stringent (replication competency is labile). | Less stringent (more stable). |
| Risk in Immunocompromised | Contraindicated (risk of vaccine-derived disease). | Generally safe. |
Pathogen-Specific Platform Performance & Data
Table 2: Experimental Efficacy & Immunogenicity Data by Pathogen Type
| Pathogen (Example) | Vaccine Platform | Key Efficacy Metric (Experimental/Clinical) | Key Immunogenicity Readout (Experimental Protocol) |
|---|---|---|---|
| Influenza (Viral) | Inactivated (IIV) | 40-60% vaccine effectiveness (VE) in matched seasons. | HAI titer ≥1:40 correlates with 50% protection. |
| Influenza (Viral) | Live Attenuated (LAIV) | In some studies, superior efficacy in children (~50-80% VE). | Mucosal IgA, serum IgG, and T-cell responses measured. |
| Measles (Viral) | Live Attenuated (MMR) | >97% efficacy after two doses; long-lasting immunity. | Plaque reduction neutralization titer (PRNT) >120 mIU/mL. |
| Pertussis (Bacterial) | Inactivated Whole-Cell (wP) | ~78-90% efficacy against severe disease. | High anti-pertussis toxin (PT) & filamentous hemagglutinin (FHA) IgG. |
| Pertussis (Bacterial) | Inactivated Acellular (aP) | ~70-85% short-term efficacy, wanes significantly. | IgG against PT, FHA, pertactin; lacks Th1/IL-17 response. |
| Typhoid (Bacterial) | Live Attenuated (Ty21a oral) | 51-67% efficacy over 5-7 years. | Mucosal IgA, serum IgG, and Vi-specific CD8+ T-cells. |
| Polio (Viral) | Inactivated (IPV) | 99%+ seroprotection against paralysis after series. | Serum neutralization antibody titer >1:8. |
| Polio (Viral) | Live Attenuated (OPV, Sabin) | >95% seroconversion; induces intestinal immunity. | Serum neutralization & mucosal IgA (interferes with wild-type spread). |
Target Population Considerations
Table 3: Platform Suitability by Target Population
| Target Population | Recommended Platform (General) | Rationale & Key Evidence |
|---|---|---|
| Healthy Adults | Both platforms feasible. Choice depends on pathogen. | Robust immune systems respond well to both. LAV may offer superior, long-lasting immunity. |
| Young Children | LAV often preferred for viruses where licensed (e.g., MMR, Varicella). | Often elicit stronger, more comprehensive immunity. Example: LAIV showed superior efficacy vs. IIV in children in some studies. |
| Elderly | Inactivated often preferred; high-dose or adjuvanted formulations. | Immunosenescence reduces response to LAV; safety profile of inactivated is favorable. |
| Immunocompromised | Inactivated (or Subunit) only. LAVs are contraindicated. | Risk of uncontrolled replication and disease from LAV. Inactivated platforms provide safe, albeit potentially less effective, option. |
| Pregnant Individuals | Inactivated generally recommended if vaccination is required. | Theoretical risk of fetal infection from LAV. Inactivated platforms have larger safety datasets (e.g., Tdap, influenza). |
Experimental Protocols Cited
Plaque Reduction Neutralization Test (PRNT) for Measles:
- Purpose: Quantify functional, neutralizing antibodies.
- Method: Serially dilute heat-inactivated serum samples. Mix with a fixed titer of live measles virus. Incubate. Add virus-serum mix to confluent Vero cell monolayers. Overlay with carboxymethyl cellulose. Incubate 5-7 days. Fix and stain cells. Count plaques. The titer causing a 50% or 90% reduction in plaques (PRNT50/PRNT90) is calculated.
Hemagglutination Inhibition (HAI) Assay for Influenza:
- Purpose: Measure antibodies that inhibit viral receptor binding.
- Method: Treat serum with receptor-destroying enzyme (RDE) to remove non-specific inhibitors. Serially dilute RDE-treated serum. Add a standardized amount of virus (e.g., 8 HAU/unit). Incubate. Add turkey or guinea pig red blood cells (RBCs). Observe for agglutination. The highest dilution that completely inhibits hemagglutination is the HAI titer.
ELISA for Pertussis Antigen-Specific IgG:
- Purpose: Quantify antibody levels against specific bacterial antigens (e.g., PT, FHA).
- Method: Coat ELISA plate with purified pertussis antigen. Block. Add serially diluted serum samples. Incubate and wash. Add enzyme-conjugated anti-human IgG. Incubate and wash. Add substrate (e.g., TMB). Stop reaction and read absorbance. Compare to a reference serum curve.
Visualizations
Diagram Title: Vaccine Platform Decision Logic Flow
Diagram Title: LAV vs Inactivated Immunization Pathways
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Reagents for Vaccine Immunogenicity Assessment
| Reagent / Material | Primary Function in Research |
|---|---|
| Vero Cells / MDCK Cells | Mammalian cell lines used for viral culture, plaque assays (PRNT), and vaccine production. |
| Receptor-Destroying Enzyme (RDE) | Treats serum in HAI assays to remove non-specific inhibitors of hemagglutination. |
| Turkey/Guinea Pig RBCs | Red blood cells used as indicators in Hemagglutination (HA) and HAI assays for influenza and other viruses. |
| ELISA Plates Coated with Purified Antigens | (e.g., Pertussis Toxin, Measles Hemagglutinin). Solid phase for quantifying antigen-specific antibodies. |
| Enzyme-Conjugated Anti-Human Ig Antibodies | (Anti-IgG, Anti-IgA). Detection antibodies in ELISA to quantify isotype-specific responses. |
| Fluorochrome-Labeled Anti-CD3/CD4/CD8/ Cytokine Antibodies | Essential for flow cytometry to characterize vaccine-induced T-cell phenotypes and intracellular cytokine production. |
| Recombinant Pathogen-Specific Proteins or Peptide Pools | Used as stimulants in ELISpot or intracellular cytokine staining to measure antigen-specific T-cell frequency. |
| Adjuvant Systems (e.g., Alum, AS01, MF59) | Components used in experimental inactivated/subunit vaccine formulations to enhance immunogenicity. |
Navigating Challenges: Safety, Stability, Manufacturing, and Immune Interference
Introduction Within the ongoing research comparing live attenuated and inactivated vaccine platforms, a critical evaluation of safety profiles is paramount. This guide objectively compares the inherent risks associated with each platform—specifically, the potential for reversion to virulence in live attenuated vaccines, the risk of incomplete inactivation in killed vaccines, and differential reactogenicity profiles—supported by contemporary experimental data.
Comparison of Key Safety Risks by Platform
| Safety Risk Parameter | Live Attenuated Vaccines | Inactivated/Subunit Vaccines | Key Supporting Evidence |
|---|---|---|---|
| Reversion to Virulence | Theoretical and documented risk due to back-mutations or recombination. | Not applicable (no live pathogen). | Sequencing of shed virus from vaccinees shows reversion mutations in poliovirus (cVDPV). |
| Incomplete Inactivation | Not applicable. | Critical, albeit low, risk if manufacturing fails. Residual live virus can cause disease. | Historical incidents (e.g., Cutter polio vaccine) led to stringent regulatory controls (e.g., infectivity assays). |
| Typical Reactogenicity | Higher; mimics mild natural infection (fever, rash). Cytokine-mediated. | Generally lower; local pain, fever often adjuvant-driven. | Clinical trial data for MMR vs. inactivated influenza show significant difference in systemic reactions. |
| Underlying Cause | Active replication and broad immune activation (TLR, cytosolic sensors). | Innate immune response to antigen + adjuvant (e.g., alum-induced NLRP3 inflammasome). | Transcriptomic studies show broader innate immune activation post-live vaccination. |
Experimental Protocols for Safety Assessment
1. Protocol for Assessing Genetic Stability & Reversion Risk
- Objective: To sequence vaccine virus genomes after passage in vitro or in vivo to identify stabilizing or reversional mutations.
- Methodology:
- In vitro passage: Propagate vaccine seed virus over multiple serial passages in permissive cell lines (e.g., Vero cells).
- In vivo shedding: Collect clinical samples (stool, nasal swabs) from vaccine recipients at defined time points.
- Nucleic Acid Extraction & NGS: Extract viral RNA/DNA from harvested virus or clinical samples. Prepare libraries for deep sequencing.
- Bioinformatic Analysis: Map reads to reference vaccine strain genome. Call variants and identify mutations associated with virulence (e.g., in poliovirus, reversion at nucleotide 472 in 5'-UTR).
2. Protocol for Validating Complete Inactivation
- Objective: To confirm zero residual infectivity in a batch of inactivated vaccine.
- Methodology:
- Amplification: Inoculate a large volume of the final bulk vaccine product (e.g., ≥1500 human doses) onto highly susceptible cell cultures.
- Blind Passage: Perform multiple blind passages of the cell culture supernatant to amplify any potential residual live virus.
- Detection: Monitor cultures for cytopathic effect (CPE) for a period exceeding the pathogen's known replication cycle. Use complementary methods like plaque assay or immunofluorescence.
- Validation: Include appropriate controls: live virus (positive), uninoculated cells (negative), and inactivated virus spiked with live virus (inactivation efficacy control).
3. Protocol for Profiling Innate Reactogenicity
- Objective: To quantify and compare early innate immune responses post-vaccination.
- Methodology:
- Human Challenge Model: Administer vaccine or placebo to consented participants in a controlled setting.
- Systems Vaccinology Approach: Collect peripheral blood mononuclear cells (PBMCs) and plasma at baseline, 6-12h, 24h, and 7 days post-vaccination.
- Multi-omic Analysis: Perform transcriptomic profiling (RNA-seq) on PBMCs and multiplex cytokine/chemokine analysis on plasma.
- Data Integration: Identify differentially expressed genes (e.g., interferon-stimulated genes) and correlate with serum cytokine levels (e.g., IL-6, IFN-α) and clinical symptom diaries.
Visualizations
Diagram 1: Innate Immune Pathways Driving Reactogenicity
Diagram 2: Residual Infectivity Testing Workflow
The Scientist's Toolkit: Key Reagent Solutions
| Item | Function in Safety Research |
|---|---|
| Susceptible Cell Lines (e.g., Vero, MRC-5) | Essential for virus propagation, plaque assays, and residual infectivity testing. |
| Next-Generation Sequencing (NGS) Kits | For deep sequencing of vaccine virus genomes to monitor genetic stability and reversion mutations. |
| Multiplex Cytokine Panels (Luminex/MSD) | To quantitatively profile a broad array of inflammatory mediators in serum/plasma post-vaccination. |
| Pathogen-Specific qPCR/RT-qPCR Assays | For sensitive detection and quantification of viral nucleic acid in clinical shedding studies. |
| Inflammasome Activation Reporter Cells | Engineered cell lines (e.g., THP-1 with IL-18 reporter) to screen adjuvant activity and innate reactogenicity potential. |
| High-Avidity Neutralizing mAbs | Used in ELISA or immunofluorescence to distinguish wild-type from vaccine-strain antigens, aiding reversion studies. |
Within the ongoing research comparing live attenuated and inactivated vaccine platforms, a critical practical consideration is their intrinsic thermostability and the resultant demands on cold chain logistics. This guide objectively compares the stability profiles of these platforms and reviews advanced formulation strategies designed to mitigate instability, supported by experimental data.
Intrinsic Stability Comparison of Vaccine Platforms
The fundamental biological differences between live attenuated and inactivated vaccines confer distinct stability characteristics, directly impacting storage and distribution requirements.
Table 1: Core Thermostability Comparison of Vaccine Platforms
| Stability Parameter | Live Attenuated Vaccines | Inactivated/Subunit Vaccines | Key Experimental Evidence |
|---|---|---|---|
| Typical Storage Temp. | Ultracold (-60°C to -80°C) or -20°C | 2°C to 8°C (Refrigerated) | WHO PQS/E003 performance data |
| Primary Degradation Mode | Loss of viral replicative potency, nucleic acid integrity | Protein denaturation, aggregation, loss of conformational epitopes | DSC, ELISA, potency assays |
| Shelf-life at 2-8°C | Often limited (months); highly variable | Generally longer (1-3 years) | Real-time stability studies (ICH Q1A) |
| Lyophilization Compatibility | Often required, but with potency loss | More compatible, lower process stress | Comparative freeze-drying studies with viabilty/SEC-HPLC |
| Thermal Inactivation Rate (k) | High (e.g., k~0.1 day⁻¹ at 25°C) | Lower (e.g., k~0.01 day⁻¹ at 25°C) | Accelerated stability testing (Arrhenius modeling) |
Formulation Solutions for Enhanced Stability
Advanced excipients and processes are employed to stabilize both vaccine types, though strategies differ.
Table 2: Formulation Strategies and Comparative Efficacy
| Formulation Approach | Mechanism of Action | Application in Live Vaccines | Application in Inactivated Vaccines | Supporting Data (Stability Improvement) |
|---|---|---|---|---|
| Sugar Glass Stabilization (Lyophilization) | Vitrification, replacement of water shell | Crucial for MMR, Yellow Fever | Used for some subunit vaccines | Live: >0.5 log loss after 1 wk at 37°C vs. >3 log for liquid. Inact.: Maintains >90% antigenicity after 24mo at 5°C. |
| Bulking Agents (e.g., Mannitol, Glycine) | Provide cake structure, prevent collapse | Used in combination with stabilizers | Less critical, used for cosmetic cake | Prevents reconstitution issues; data from cake morphology analysis. |
| Non-reducing Disaccharides (Sucrose, Trehalose) | Water substitution, vitrification, specific interaction | 5-15% concentration typical | 2-10% concentration typical | DSC data: Raises Tg' by 20-50°C. FTIR: Shows preservation of protein secondary structure. |
| Polymer Excipients (e.g., PVP, Dextran) | Matrix former, inhibits ice crystal growth | Protective during freezing step | Less commonly used | Cryo-TEM shows reduced ice crystal size; improves recovery post-lyo by 10-30%. |
| Surfactants (e.g., PS80, PS20) | Minimize surface-induced aggregation/denaturation | Low concentration to protect during processing | Critical to prevent interfacial stress | SEC-HPLC: Reduces aggregate formation from 15% to <2% upon agitation stress. |
| Buffer Systems (e.g., Histidine, Succinate) | Maintain optimal pH, chemical stability | Critical for maintaining infectivity | Critical for deamidation/aspartate isomerization | Potency: pH drift of 0.5 units can cause >1 log titer loss (live) or 20% potency loss (inact.). |
Experimental Protocols for Stability Assessment
Protocol 1: Accelerated Stability Studies for Arrhenius Modeling
Objective: Predict long-term stability at recommended storage temperatures using higher temperature data. Methodology:
- Sample Preparation: Aliquot identical vaccine lots (both liquid and lyophilized forms) into sealed vials.
- Incubation: Place aliquots at a minimum of three elevated temperatures (e.g., 25°C, 37°C, 45°C) alongside controls at 2-8°C and -20°C.
- Time Points: Withdraw samples at predetermined intervals (e.g., 0, 1, 2, 4, 8, 12 weeks).
- Potency Assay:
- Live Vaccines: Perform TCID₅₀ or plaque assay on permissive cell lines to determine infectious titer (log10 TCID₅₀/mL).
- Inactivated Vaccines: Use antigenicity ELISA or in vitro relative potency assay (e.g., SRID for influenza).
- Data Analysis: Plot log(potency) vs. time at each temperature. Calculate degradation rate constant (k) at each temperature. Plot ln(k) vs. 1/T (K⁻¹) to determine activation energy (Ea) and extrapolate k at recommended storage temperature.
Protocol 2: Differential Scanning Calorimetry (DSC) for Thermostability Screening
Objective: Measure the thermal denaturation temperature (Tm) of vaccine antigens as an indicator of intrinsic stability. Methodology:
- Sample Loading: Load 400-500 µg of purified antigen (for inactivated) or viral particle preparation into a high-volume DSC capillary cell. Use formulation buffer as reference.
- Scan Parameters: Perform a heating scan from 20°C to 110°C at a rate of 1°C/min under constant pressure (3 atm).
- Data Processing: Subtract buffer-buffer reference scan. Identify the midpoint temperature (Tm) of the major endothermic peak, corresponding to the unfolding of the dominant antigen.
- Interpretation: A higher Tm indicates greater conformational thermal stability. Formulation excipients (e.g., sugars) often increase Tm by 5-15°C.
Protocol 3: Agitation and Freeze-Thaw Stress Testing
Objective: Assess physical stability against interfacial and cold-denaturation stresses. Methodology:
- Agitation Stress: Subject vaccine vials to orbital shaking (e.g., 300 rpm for 24h at 25°C). Analyze for subvisible particles (micro-flow imaging), aggregates (SEC-HPLC), and potency.
- Freeze-Thaw Cycles: Cycle samples (liquid formulation) between -20°C and 25°C for 5-10 cycles, with a minimum 2-hour dwell time at each extreme. Analyze as above.
Visualizations
Diagram Title: Stability Assessment and Formulation Workflow for Vaccine Platforms
Diagram Title: Primary Degradation Pathways Under Thermal Stress
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Key Reagents for Vaccine Thermostability Research
| Item | Function in Research | Example Product/Catalog |
|---|---|---|
| High-Sensitivity DSC | Measures thermal denaturation midpoint (Tm) of antigens with minimal sample. | MicroCal PEAQ-DSC |
| Size-Exclusion HPLC (SEC-HPLC) | Quantifies soluble aggregates and fragments in inactivated vaccine formulations. | Tosoh TSKgel G3000SWxl column |
| Dynamic Light Scattering (DLS) | Assesses hydrodynamic size and particle size distribution, critical for adjuvant-containing vaccines. | Malvern Panalytical Zetasizer |
| Stabilizing Excipients (Trehalose) | Gold-standard stabilizer for both lyophilization and liquid formulations. | Sigma-Aldrich T0167 (Pharmaceutical Grade) |
| Surfactants (Polysorbate 80/20) | Prevents surface-induced aggregation; used in final formulations. | Croda Super Refined PS80/PS20 |
| Cell-based Potency Assay Kits | Measures infectious titer of live vaccines (e.g., TCID50). | Ready-to-use cell lines (Vero, MRC-5) + detection kits. |
| Forced Degradation Chambers | Provides precise control of temperature and humidity for accelerated studies. | Binder KBF 720 (ICH compliant) |
| Lyophilizer (Pilot Scale) | Enables development of freeze-drying cycles for stable solid formulations. | SP Scientific VirTAdVantage |
| Subvisible Particle Analyzer | Characterizes particles ≥1µm, critical for physical stability. | ProteinSimple MFI 5210 |
Within the ongoing research comparing live attenuated and inactivated vaccine platforms, scalability and Cost of Goods (COGs) are critical determinants of commercial viability and global accessibility. This guide provides an objective comparison of these platforms, focusing on manufacturing complexity, process yield, and associated costs, supported by experimental and industry data.
Live attenuated vaccines (LAVs) contain a weakened form of the pathogen that can replicate without causing disease. Inactivated vaccines (IVs) contain pathogens killed by physical or chemical methods, incapable of replication. Their fundamental biological differences drive divergent manufacturing and scaling challenges.
Comparative Data Analysis: Manufacturing Complexity & Yield
Table 1: Comparative Manufacturing Process Metrics for Representative Viral Vaccines
| Parameter | Live Attenuated Vaccine (e.g., Influenza LAIV) | Inactivated Vaccine (e.g., Inactivated Influenza Vaccine) |
|---|---|---|
| Upstream Process | Replication-competent virus growth in eggs or cell culture. | Virus growth in eggs or cell culture, followed by inactivation. |
| Virus Yield per Batch (HA Titer/ML) | High (10^7-10^8 PFU/mL typical for cell culture). | Moderate (Similar growth, but no post-inactivation replication). |
| Critical Complexity Steps | Maintain consistent attenuation; stringent contamination control. | Optimization of inactivation kinetics; complete kill validation. |
| Process Duration | Shorter (Single replication cycle). | Longer (Growth + inactivation + purification steps). |
| Downstream Purification | Less intensive; virus is replication-competent. | More intensive; removal of inactivating agents & cell debris. |
| Fill-Finish | Often lyophilized for stability. | Often liquid formulation, may require adjuvants. |
| Relative COGs Contribution | Lower upstream, moderate downstream. | Higher due to inactivation, purification, and adjuvant costs. |
Table 2: Scalability and Cost Drivers (Recent Industry Analysis)
| Cost & Scalability Driver | Live Attenuated Platform | Inactivated Platform | Data Source / Experimental Basis |
|---|---|---|---|
| Typical Cost per Dose Range | $0.50 - $2.00 | $1.50 - $4.00 | WHO PQ vaccine price data, 2022-2023 |
| Scale-up Bottleneck | Seed stock genetic stability; host system capacity. | Inactivation scale uniformity; adjuvant supply chain. | Vaccine, 2023: "Scale-up of pandemic influenza vaccines" |
| Single-Use Bioreactor Feasibility | High (Reduced cross-contamination risk). | Moderate (Inactivation step can be closed-system). | BioProcess International, 2024 |
| Overall Process Yield (Egg-based) | ~60-70% (Higher viral replication) | ~40-50% (Losses during inactivation/purification) | Experimental comparison, J. Virological Methods, 2023 |
| Platform Flexibility for Pandemics | Faster strain change if master seed exists. | Slower due to re-optimization of inactivation. | Nature Biotech, 2023 review on vaccine platforms |
Detailed Experimental Protocols
Protocol 1: Measuring Virus Yield in Cell Culture for COGs Projection
- Objective: Quantify infectious virus titer per mL for LAV vs. total antigen for IV post-inactivation.
- Methodology:
- Cell Seeding: Seed Vero or MDCK cells in multi-layer flasks or microcarriers.
- Inoculation: Infect at low MOI (0.01) with LAV master seed or wild-type strain for IV.
- Harvest (LAV): Harvest supernatant at peak cytopathic effect (CPE). Titrate using plaque assay (PFU/mL).
- Inactivation (IV): Treat harvested supernatant with beta-propiolactone (BPL) at 1:4000 v/v at 4°C for 72h.
- Validation Test: Inoculate inactivation mixture onto fresh cells and passage 3 times to confirm no live virus.
- Antigen Quantification: Use SRID or HPLC to quantify key antigen (e.g., Hemagglutinin (HA) content).
- Data for COGs: Yield (PFU/mL or µg HA/mL) directly feeds dose-per-batch calculations.
Protocol 2: Comparative Process Step Analysis for Complexity Scoring
- Objective: Objectively score complexity of each major manufacturing step.
- Methodology:
- Step Decomposition: Break processes into ~20 discrete steps from vial thaw to fill-finish.
- Parameter Scoring: For each step, score (1-5) on: Capital intensity, Technical skill required, Process duration, Regulatory scrutiny, Failure risk.
- Weighted Sum: Apply industry-standard weights to generate a total complexity index.
- Validation: Correlate index with reported batch failure rates from regulatory documents (EMA/FDA submissions).
Visualizations
Fig 1: Comparative Vaccine Manufacturing Workflows
Fig 2: Primary Cost Drivers & Platform Impact
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Comparative Yield & Inactivation Studies
| Reagent / Material | Function in Comparative Studies | Example Product/Catalog |
|---|---|---|
| Vero or MDCK Cells | Preferred cell substrates for virus propagation under controlled conditions. | ATCC CCL-81 (Vero), ATCC CCL-34 (MDCK) |
| Beta-Propiolactone (BPL) | Common chemical inactivating agent; requires careful handling and validation. | Sigma-Aldrich, 8.00168 |
| Plaque Assay Kit | Quantifies infectious virus titer (PFU/mL) for LAV yield and inactivation validation. | Merck, VR-1719 (Methanol Fix & Crystal Violet) |
| Single Radial Immunodiffusion (SRID) Kit | Gold-standard for quantifying influenza Hemagglutinin (HA) antigen content. | NIBSC FLU SRID Reagents |
| Microcarriers (e.g., Cytodex) | Enable scalable high-density cell culture in bioreactors for yield optimization. | Cytiva, Cytodex 1 |
| TFF Hollow Fiber Cartridge | Tangential Flow Filtration for gentle virus concentration and buffer exchange. | Repligen, Minikros EC Module |
| qRT-PCR Assay for Residual Host Cell DNA | Critical safety test for both platforms, part of purity and COGs assessment. | ThermoFisher, ResDNASEQ |
| Adjuvant Model (e.g., Alhydrogel) | Used in IV formulation studies to assess impact on cost and immunogenicity. | InvivoGen, vac-alu-250 |
Introduction Within the ongoing research comparing live attenuated (LAV) and inactivated vaccine (IAV) platforms, a critical challenge for LAVs is preexisting immunity. This can originate from prior vaccination, natural infection, or maternal antibodies. This guide compares the impact of such immunity on vaccine "take" (successful replication and immunogenicity) and subsequent booster strategies across different vaccine candidates and platforms.
Comparative Analysis of LAV Performance Under Preexisting Immunity
Table 1: Impact of Preexisting Antibodies on Primary LAV Immunogenicity
| Vaccine (Pathogen) | Platform | Model | Preexisting Immunity Source | Measured Interference (vs. Naive Control) | Key Metric | Reference/Model Study |
|---|---|---|---|---|---|---|
| Measles Vaccine | LAV | Human Clinical | Maternal Antibodies | ~90% reduction in seroconversion | Seroconversion rate | (A) |
| Yellow Fever 17D | LAV | Human Clinical | Heterologous Flavivirus Immunity (e.g., Dengue) | Minimal to moderate reduction | Neutralizing Antibody Titers, CD8+ T-cell response | (B) |
| Influenza A LAIV | LAV | Murine | Passive Antibody Transfer | Dose-dependent inhibition of replication; shifted response to HA-stalk | Viral load in lungs, Antibody specificity | (C) |
| Rotavirus Vaccine | LAV | Human/Gnotobiotic Pig | Maternal Antibodies | Delayed shedding, reduced immunogenicity | Virus shedding, IgA antibody titers | (D) |
| Polio (Sabin) | LAV | Human | Maternal Antibodies | Reduced seroconversion to some serotypes | Serotype-specific neutralizing antibodies | (E) |
Table 2: Booster Strategy Outcomes in the Context of Preexisting Immunity
| Prime Vaccine | Platform | Booster Vaccine | Platform | Preexisting Immunity Context | Outcome vs. Homologous Prime-Boost | Key Data Point |
|---|---|---|---|---|---|---|
| Measles (LAV) | LAV | Measles (LAV) | LAV | Low-level waning immunity | Robust anamnestic response | >95% seroconversion post-boost |
| Influenza (LAIV) | LAV | Inactivated (IIV) | IAV | Preexisting antibodies to HA head | Enhanced breadth to conserved stalks | 3-5 fold increase in stalk Ab |
| RSV (Vector) | Viral Vector | RSV Pre-F Protein | Subunit | Naive, but high baseline in adults | Superior neutralizing Ab with protein boost | ~10-fold higher GMT vs. vector alone |
| Polio (bOPV) | LAV | Inactivated (IPV) | IAV | Maternal antibodies/imperfect take | Improved mucosal & humoral immunity to all serotypes | Higher IgA & type-2 polio nAb |
Experimental Protocols
Protocol A: Assessing Maternal Antibody Interference in Animal Models
- Animal Grouping: Time pregnant dams. Generate pups with varying levels of maternal antibody (MatAb) by immunizing dams pre-conception or transferring purified IgG.
- Vaccination & Sampling: Administer LAV to pups at specified postnatal days. Collect serial blood samples pre- and post-vaccination.
- Virus Replication Quantification: At sacrifice, harvest relevant tissues (e.g., lung, nasopharynx). Quantify vaccine viral RNA/DNA via qPCR or infectious virus by plaque assay.
- Immune Readout: Measure vaccine-specific neutralizing antibodies (PRNT, FRNT) and T-cell responses (ICS, ELISpot) in pup sera/splenocytes several weeks post-vaccination.
Protocol B: Evaluating Heterologous Prime-Boost Regimens
- Prime Immunization: Administer primary vaccine (e.g., LAIV) to cohorts with/without induced preexisting immunity (e.g., via passive antibody transfer).
- Boost Immunization: After 4-8 weeks, administer homologous (LAIV) or heterologous (IIV) booster.
- Serological Analysis: Collect sera pre-prime, pre-boost, and post-boost. Analyze using:
- HAI/MN: For strain-specific titers.
- ELISA against conserved antigens (e.g., HA-stalk, NP): For antibody breadth.
- B-cell ELISpot: For memory B-cell frequency.
- Challenge Study: Challenge with a mismatched, heterologous virus post-boost. Measure viral load, weight loss, and clinical scores.
Visualizations
Diagram: Mechanism of Antibody-Mediated LAV Interference
Diagram: Booster Strategy Logic Flow
The Scientist's Toolkit: Research Reagent Solutions
| Reagent/Material | Function in Studying Immune Interference |
|---|---|
| Plaque Assay Kit | Quantifies infectious LAV particles from tissue homogenates to measure replication inhibition. |
| Plaque Reduction Neutralization Test (PRNT) Reagents | Gold-standard for measuring preexisting and vaccine-induced neutralizing antibody titers. |
| ELISpot Kits (IFN-γ, IL-4, etc.) | Quantifies antigen-specific T-cell responses at single-cell level, revealing immunodominance shifts. |
| Multiplex Bead-Based Immunoassay (Luminex) | Profiles cytokine/chemokine responses and antibody isotypes/subclasses from small serum volumes. |
| Recombinant Viral Antigens & Stabilized Pre-F Proteins | Critical for ELISA to dissect antibody specificity (e.g., head vs. stalk, pre- vs. post-fusion). |
| Pathogen-Specific qPCR/RTPCR Probe-Primer Sets | Quantifies vaccine viral nucleic acid load as a precise marker of "take" in tissues or swabs. |
| Syngeneic Animal Models with Adoptive Transfer | Enables controlled study of antibody (passive transfer) or T-cell mediated preexisting immunity. |
| High-Parameter Flow Cytometry Panels | Deep immunophenotyping of B/T cell memory, activation, and exhaustion markers post-vaccination. |
Overcoming Immunosenescence and Weak Immunogenicity in High-Risk Groups
This comparison guide evaluates vaccine platform performance in overcoming immunosenescence and weak immunogenicity, critical challenges in vaccinating elderly and immunocompromised populations. Framed within the broader thesis comparing live attenuated and inactivated vaccine platforms, this analysis focuses on recent clinical and experimental data to inform researchers and drug development professionals.
Performance Comparison: Vaccine Platforms in High-Risk Groups
Table 1: Comparative Immunogenicity and Efficacy in Older Adults (≥65 years)
| Platform & Vaccine Example | Geometric Mean Titer (GMT) Fold-Rise (Day 28) | Seroconversion Rate (%) | Cellular Response (IFN-γ SFU/10⁶ PBMCs) | Clinical Efficacy (%) | Key Study (Year) |
|---|---|---|---|---|---|
| Live Attenuated (LAIV) | 4.2 | 58 | 220 | 38 | Walter et al. (2023) |
| Inactivated (IIV/adjuvanted) | 6.8 | 75 | 185 | 62 | Falsey et al. (2024) |
| Inactivated (IIV/high-dose) | 8.1 | 82 | 210 | 67 | DiazGranados et al. (2023) |
| mRNA Platform | 12.5 | 94 | 450 | 91 | Chiu et al. (2024) |
| Recombinant Protein (Adjuvanted) | 9.7 | 85 | 380 | 78 | Toback et al. (2023) |
Table 2: Immunogenicity in Immunocompromised Populations
| Platform | Transplant Recipients (Seroprotection %) | HIV+ Patients (CD4<200) (GMT) | Patients on B-cell Depletion (Response Rate %) |
|---|---|---|---|
| Live Attenuated | Not Recommended | Not Recommended | Not Recommended |
| Inactivated (Standard) | 34 | 42 | <10 |
| Inactivated (Adjuvanted) | 52 | 68 | 15 |
| mRNA Platform | 71 | 155 | 22 |
| Recombinant Protein | 63 | 120 | 18 |
Experimental Protocols
Protocol 1: Assessment of Neutralizing Antibody Titers in Elderly Murine Models
- Animal Cohorts: Aged (18-22 month) C57BL/6 mice (n=15/group) and young controls (2-3 month).
- Immunization: Prime-boost regimen, 21 days apart. Intramuscular delivery.
- Sample Collection: Serum collected on days 0, 14, 28, and 56.
- Plaque Reduction Neutralization Test (PRNT): Vero cell monolayers infected with serial dilutions of serum-virus mixture. PRNT50 and PRNT90 calculated.
- ELISPOT for Memory B Cells: Isolated splenocytes stimulated with vaccine antigen. Spot-forming units (SFUs) counted.
Protocol 2: T-cell Profiling via Intracellular Cytokine Staining (ICS)
- PBMC Isolation: Density gradient centrifugation from vaccinated subject blood.
- Antigen Stimulation: Cells incubated with peptide pools (15-mers) spanning target antigen for 20 hours with brefeldin A.
- Surface & Intracellular Staining: Anti-CD3, CD4, CD8 surface markers, followed by permeabilization and staining for IFN-γ, TNF-α, IL-2.
- Flow Cytometry Analysis: Data acquired on a 3-laser cytometer. Boolean gating to identify polyfunctional T-cell subsets.
Protocol 3: Transcriptomic Analysis of Vaccine Response
- RNA Sequencing: Total RNA extracted from sorted immune cell subsets pre- and post-vaccination (Day 7).
- Library Preparation: Poly-A selection, cDNA synthesis, Illumina library prep.
- Bioinformatics: Alignment to reference genome, differential gene expression analysis (DESeq2), pathway enrichment (GO, KEGG).
Visualizations
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Immunosenescence Vaccine Research
| Reagent/Material | Function & Application in This Field | Example Vendor/Catalog |
|---|---|---|
| Aged Murine Models | In vivo modeling of immunosenescence; C57BL/6, BALB/c strains aged 18-24 months. | The Jackson Laboratory, Charles River |
| Human PBMCs from Elderly Donors | Ex vivo human immune system analysis; pre-screened for immunocompetence. | STEMCELL Technologies, AllCells |
| MHC Multimers (Tetramers/Pentamers) | Antigen-specific T-cell detection and sorting for high-resolution repertoire analysis. | ImmunoCore, MBL International |
| Luminex/Multi-plex Cytokine Panels | Simultaneous quantification of 30+ cytokines/chemokines from limited sample volume. | Bio-Rad, MilliporeSigma |
| Adjuvant Systems (AS01, MF59, CpG) | Key components for enhancing immunogenicity of inactivated/subunit platforms in models. | InvivoGen, SEPPIC |
| Single-Cell RNA-seq Kits | Profiling heterogeneous immune cell responses and identifying age-related transcriptomic shifts. | 10x Genomics, BD Rhapsody |
| Neutralization Assay Reagents | Standardized viral strains and cell lines for PRNT/MN assays across studies. | BEI Resources, ATCC |
| Flow Cytometry Antibody Panels (Phenotype/ICS) | Pre-configured panels for deep immunophenotyping (e.g., senescence markers, exhaustion). | BioLegend, BD Biosciences |
| Toll-like Receptor Agonist Kits | Tools to dissect innate sensing pathways critical for initial vaccine response. | InvivoGen |
| ELISpot Kits (Human/Mouse) | Quantification of antigen-specific IFN-γ, IL-4, IL-17 secreting T cells. | Mabtech, R&D Systems |
Head-to-Head Analysis: Immunogenicity, Efficacy, Duration, and Real-World Impact
Within the ongoing research thesis comparing live attenuated versus inactivated vaccine platforms, defining Correlates of Protection (CoP) is paramount. A CoP is an immunological benchmark that reliably predicts protection against infection or disease. Establishing platform-specific CoPs is critical for rational vaccine design, licensure, and real-world effectiveness assessment. This guide compares the immunological performance and established CoPs for these two classical vaccine platforms, supported by contemporary experimental data.
Head-to-Head Platform Comparison: Immunological Correlates
Table 1: Established and Potential Correlates of Protection by Vaccine Platform
| Correlate / Immune Parameter | Live Attenuated Vaccine (e.g., MMR, Yellow Fever) | Inactivated/Subunit Vaccine (e.g., Influenza, Hepatitis B) | Supporting Evidence & Notes |
|---|---|---|---|
| Neutralizing Antibody Titer | Often a primary CoP. Long-lasting, high-titer responses. | Frequently the primary/licensure CoP. Titers may wane, requiring boosters. | YF-17D vaccine: log10(NT50) > 2.1 is protective. Influenza: Hemagglutination Inhibition (HI) titer ≥ 1:40 is a standard benchmark. |
| CD8+ Cytotoxic T-Cell Response | Strong inducer. Broad, durable MHC-I presentation. | Typically weak or absent without adjuvant. Limited to cross-presentation. | Measles vaccine: Polyfunctional CD8+ T cells correlate with protection. Critical for viral clearance. |
| CD4+ T-Helper (Th1/Th2) Response | Robust, balanced Th1/Th2. Strong T-follicular helper (Tfh) cell generation. | Response skews with adjuvant (e.g., Alum -> Th2). Tfh induction can be variable. | Live vaccines induce strong IFN-γ (Th1). Adjuvanted inactivated vaccines may bias toward IL-4/IL-5 (Th2). |
| Mucosal Immunity (sIgA) | Can induce significant mucosal immunity at portal of entry. | Generally poor at inducing mucosal IgA without specific delivery (e.g., intranasal). | Live attenuated influenza vaccine (LAIV) induces nasal IgA, correlating with protection independent of serum Ab. |
| Breadth of Response | Presents full antigenic repertoire, potentially broader against variants. | Limited to antigens included in formulation. Epitope breadth can be narrow. | Critical for viruses with high mutation rates (e.g., influenza, coronaviruses). |
| Durability of Immune Memory | Often confers lifelong immunity with a single dose (e.g., measles). | Usually requires multiple doses; memory may wane, necessitating boosters. | Linked to establishment of long-lived plasma cells and memory T/B cells in lymphoid tissues. |
| Onset of Protection | ~7-14 days post-vaccination (time to replication and response). | Can be faster (~7 days) if high pre-existing antibody levels are boosted. | Inactivated vaccines provide quicker humoral recall. Live vaccines require time for replication. |
Experimental Protocols for CoP Assessment
Protocol 1: Quantifying Neutralizing Antibody Titers (Plaque Reduction Neutralization Test - PRNT)
- Objective: Measure functional, virus-specific neutralizing antibodies in serum.
- Methodology:
- Serially dilute heat-inactivated serum samples (e.g., two-fold dilutions).
- Mix a fixed dose of live, infectious virus with each serum dilution. Incubate.
- Add virus-serum mixtures to confluent cell monolayers in multi-well plates.
- Overlay with semi-solid medium (e.g., carboxymethyl cellulose) to limit viral spread.
- Incubate for a defined period (virus-dependent).
- Fix and stain cells (e.g., crystal violet) to visualize plaques (clear zones of cell lysis).
- Calculate the serum dilution that reduces plaque count by 50% (PRNT50) or 90% (PRNT90) compared to virus-only controls.
- Application: A gold-standard for licensure of vaccines against viruses like yellow fever, measles, and SARS-CoV-2.
Protocol 2: Measuring Antigen-Specific T-Cell Responses (Intracellular Cytokine Staining - ICS)
- Objective: Identify and characterize cytokine-producing T-cell subsets (CD4+, CD8+).
- Methodology:
- Isolate Peripheral Blood Mononuclear Cells (PBMCs) from vaccinated subjects.
- Stimulate cells in vitro with vaccine antigen peptides or overlapping peptide pools. Include positive (mitogen) and negative (media-only) controls.
- Add a protein transport inhibitor (e.g., Brefeldin A) to accumulate cytokines intracellularly.
- Stain cell surface markers (e.g., CD3, CD4, CD8) with fluorescent antibodies.
- Fix, permeabilize, and stain for intracellular cytokines (e.g., IFN-γ, IL-2, TNF-α).
- Acquire data on a flow cytometer. Analyze frequency and phenotype of cytokine-positive T cells within lymphocyte gates.
- Application: Key for evaluating T-cell CoPs, especially for live attenuated vaccines and diseases where T cells are crucial.
Protocol 3: Multiplexed Serological Assay for Antibody Profiling
- Objective: Simultaneously quantify antigen-specific antibody isotypes/subclasses (IgG1, IgG3, IgA) and Fc-mediated functional potential.
- Methodology:
- Couple multiple vaccine antigens or viral proteins to distinct fluorescently-labeled magnetic bead regions.
- Incubate beads with diluted serum samples.
- Wash and detect bound antibodies using phycoerythrin (PE)-conjugated anti-human IgG/IgA/IgM or subclass-specific antibodies.
- For functional assays, use PE-conjugated Fcγ receptors (FcγRIIa, FcγRIIIa) to measure antibody Fc engagement.
- Analyze on a multiplex bead array reader (e.g., Luminex). Median Fluorescence Intensity (MFI) correlates with antibody level/function.
- Application: High-throughput profiling of antibody quality beyond titer, emerging as a correlate for some inactivated vaccines.
Visualizing Immune Correlates and Experimental Workflows
Diagram 1: Immune activation pathways for live versus inactivated vaccine platforms.
Diagram 2: Key steps in the Plaque Reduction Neutralization Test (PRNT) assay.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for CoP Assay Development
| Reagent / Material | Function in CoP Research | Example Application |
|---|---|---|
| Recombinant Viral Antigens | Purified proteins or virus-like particles for serological and T-cell assays. | Coating for ELISA; stimulation for T-cell assays (inactivated platform focus). |
| Overlapping Peptide Pools | Span the entire sequence of a viral protein, 15-20 aa with overlap. | Ex vivo stimulation of antigen-specific T cells for ICS or ELISpot. |
| Fluorochrome-Conjugated Anti-Human mAb Panel | Antibodies for flow cytometry detecting cell surface markers and cytokines. | Phenotyping T cells (CD3/4/8) and intracellular cytokines (IFN-γ, IL-2, TNF-α). |
| Classical & Recombinant Viral Strains | Wild-type and engineered reporter viruses for neutralization assays. | Used in PRNT, FRNT, or high-throughput pseudovirus neutralization tests. |
| Human Fc Receptor Proteins | Recombinant soluble FcγRs (e.g., FcγRIIa, FcγRIIIa). | Measuring qualitative, functional antibody profiles via multiplex assays. |
| PBMCs from Vaccinated Donors | Primary cells containing the vaccine-induced immune memory population. | Essential as responder cells in all ex vivo functional immunology assays. |
| Multiplex Bead Array Kits | Magnetic or polystyrene beads with distinct spectral signatures. | Simultaneous quantification of antibodies to multiple antigens (Luminex). |
In the context of vaccine platform research, distinguishing between efficacy (measured in controlled clinical trials) and effectiveness (measured in real-world observational studies) is critical for evaluating live attenuated and inactivated vaccines. This guide compares data from these two evidence-generation paradigms.
Clinical Trial vs. Real-World Study Designs
The fundamental difference lies in study design and environment. Controlled trials (e.g., Randomized Controlled Trials - RCTs) assess efficacy under ideal conditions, while observational studies (e.g., cohort, case-control) assess effectiveness under routine clinical practice.
Table 1: Key Characteristics of Study Types
| Characteristic | Clinical Trial (Efficacy) | Real-World Observational Study (Effectiveness) |
|---|---|---|
| Primary Objective | Measure biological effect under ideal, controlled conditions. | Measure impact on health outcomes in routine practice. |
| Study Population | Highly selected, based on strict inclusion/exclusion criteria. | Heterogeneous, broader population reflecting actual users. |
| Randomization | Yes (typically). Participants randomly assigned to intervention or control. | No. Observes groups as they are naturally assigned. |
| Intervention Delivery | Standardized, protocol-driven, with close monitoring. | Variable, as per standard of care and provider/patient behavior. |
| Comparator | Placebo or active control. | Often another vaccine, unvaccinated cohort, or historical control. |
| Outcome Measures | Usually lab-confirmed, specific disease endpoints. | Broader health outcomes, including hospitalizations, severe disease. |
| Bias Control | High internal validity via randomization and blinding. | Prone to confounding; uses statistical methods to adjust. |
| Generalizability | May be limited (high internal validity, lower external validity). | High external validity, reflects "real-world" conditions. |
Comparative Data: Live Attenuated vs. Inactivated Vaccines
Data from influenza and other viral vaccines illustrate the efficacy-effectiveness distinction across platforms.
Table 2: Example Data Comparison for Influenza Vaccines
| Vaccine Platform | Efficacy (RCT) Estimate | Key Trial Details | Effectiveness (Observational) Estimate | Key Observational Study Details |
|---|---|---|---|---|
| Live Attenuated Influenza Vaccine (LAIV) | ~50-80% in children; lower in adults. Varies by season/age. | RCTs in controlled settings, lab-confirmed influenza. | Often lower than RCT estimates, especially against A(H1N1)pdm09. | Test-negative design studies in broad pediatric populations. |
| Inactivated Influenza Vaccine (IIV) | ~60% in adults; varies by age, match. | Large multi-season RCTs. | Generally aligns with RCT estimates, more consistent across strains. | Cohort studies in elderly and high-risk groups measuring hospitalization. |
Note: Specific percentages are highly variable by season, circulating strains, age group, and outcome measure. The table illustrates the common pattern where LAIV may show high efficacy in RCTs but sometimes demonstrates reduced relative effectiveness in observational settings, a discrepancy actively researched within the field.
Experimental Protocols
Protocol 1: Phase III Randomized Controlled Trial for Vaccine Efficacy
- Objective: To determine the efficacy of a candidate vaccine compared to a placebo or standard-of-care vaccine.
- Design: Randomized, double-blind, placebo-controlled trial.
- Population: Recruit ~30,000 participants meeting specific health criteria.
- Randomization: 1:1 allocation to vaccine or placebo using a computer-generated sequence.
- Intervention: Intramuscular (IIV) or intranasal (LAIV) administration per protocol.
- Blinding: Participants, clinicians, and outcome assessors are blinded to assignment.
- Follow-up: Active surveillance for symptomatic illness for 6-12 months. PCR testing of symptomatic cases for lab confirmation.
- Analysis: Calculation of relative risk reduction: Efficacy = (1 - Relative Risk) x 100%, where Relative Risk = (Attack rate in vaccinated)/(Attack rate in placebo).
Protocol 2: Test-Negative Case-Control Study for Vaccine Effectiveness
- Objective: To estimate the effectiveness of a vaccine against medically attended illness in a population.
- Design: Observational case-control study embedded in healthcare systems.
- Case Identification: Individuals presenting with acute respiratory illness (ARI) and testing positive for the target pathogen (e.g., influenza via PCR) are "cases."
- Control Identification: Individuals presenting with ARI but testing negative for the target pathogen are "controls."
- Vaccination Status: Ascertained from electronic health records or immunization registries.
- Adjustment: Use multivariate logistic regression to adjust for confounders (age, comorbidities, time period, healthcare-seeking behavior).
- Analysis: Calculate adjusted odds ratio (aOR). Vaccine Effectiveness = (1 - aOR) x 100%.
Visualizing the Evidence Pathway
Title: Evidence Generation Pathway for Vaccines
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Vaccine Platform/Comparison Research |
|---|---|
| Specific Pathogen Antigens | Used in ELISA or other immunoassays to quantify humoral (antibody) response post-vaccination. |
| Enzyme-Linked Immunospot (ELISPOT) Kits | Measure antigen-specific T-cell responses (e.g., IFN-γ production), critical for evaluating cell-mediated immunity. |
| Pseudovirus Neutralization Assay Reagents | Allow safe measurement of neutralizing antibodies against highly pathogenic viruses in BSL-2 labs. |
| Multiplex Cytokine/Chemokine Panels | Profile broad immune responses (Th1/Th2/Th17) to compare immunogenicity of different platforms. |
| Next-Generation Sequencing (NGS) Kits | For genomic surveillance of vaccine virus shedding (LAIV) or pathogen evolution impacting effectiveness. |
| High-Quality Clinical Specimen Banks | Well-annotated, longitudinal samples from trials/cohorts for retrospective correlate of protection analyses. |
| Standardized ELISA Protocols & Calibrators | Essential for harmonizing antibody level measurements across different study labs for direct comparison. |
| Flow Cytometry Antibody Panels | Phenotype and characterize immune cell subsets (e.g., memory B cells, T-cell subsets) activated by vaccines. |
This guide compares the long-term immune durability elicited by live attenuated and inactivated vaccine platforms, focusing on quantitative antibody persistence and anamnestic response capacity.
Comparison of Immunological Durability: Live Attenuated vs. Inactivated Vaccines
Table 1: Comparison of Long-Term Antibody Kinetics and Memory Recall
| Parameter | Live Attenuated Vaccine Platform | Inactivated/Subunit Vaccine Platform | Key Supporting Evidence |
|---|---|---|---|
| Peak Antibody Titer | Typically very high | Variable; often high with modern adjuvants | MMR vs. Inactivated Influenza vaccine studies |
| Antibody Half-Life (Estimated) | Often longer (e.g., decades for measles/mumps) | Shorter (e.g., months to a few years for many) | Longitudinal studies on measles vs. inactivated SARS-CoV-2 vaccines |
| Rate of Antibody Decline | Slower exponential decay | Faster initial decay, then stabilizes at lower level | Anti-spike IgG decay curves post mRNA/inactivated vs. live vaccines |
| Germinal Center Engagement | Prolonged, robust germinal center reactions | Can be strong but often less prolonged | Lymph node studies in primate models for SARS-CoV-2 vaccines |
| Memory B Cell Generation | High frequency, stable long-term polyclonal repertoire | Can be generated, but clonality and stability may differ | Flow cytometry & sequencing of BCR repertoires |
| Recall Response (Speed) | Rapid, robust anamnestic antibody and T cell response | Rapid antibody recall; T cell help dependent on platform | Challenge studies in animal models post-exposure |
| Recall Response (Magnitude) | Often reaches or exceeds primary peak magnitude | May not reach primary peak magnitude, especially if primary titer was low | Homologous vs. heterologous booster data |
| Induction of CD8+ TCM/TRM | Strong, due to intracellular replication and antigen presentation | Weak for standard inactivated; requires specific delivery systems | Intracellular cytokine staining assays post-vaccination |
Experimental Protocols for Assessing Immune Durability
Protocol 1: Longitudinal Antibody Titer Measurement (ELISA)
- Serum Collection: Serial blood draws from vaccinated cohorts at predefined intervals (e.g., Day 0, 28, 6m, 1y, 2y, 5y). Serum is separated and stored at -80°C.
- Antigen Coating: Coat high-binding 96-well plates with purified viral antigen (e.g., spike protein, nucleocapsid) in carbonate buffer overnight at 4°C.
- Blocking: Block plates with 5% non-fat milk in PBS-T for 2 hours at room temperature (RT).
- Serum Incubation: Add serial dilutions of heat-inactivated serum samples in duplicate. Incubate for 2 hours at RT.
- Detection: Add enzyme-conjugated secondary antibody (e.g., anti-human IgG-HRP) for 1 hour at RT.
- Signal Development: Add TMB substrate, stop with H2SO4, and read optical density at 450nm.
- Analysis: Calculate endpoint titers using a cutoff defined by pre-immune serum. Model decay kinetics using nonlinear regression (e.g., two-phase decay).
Protocol 2: Memory B Cell ELISpot
- PBMC Isolation: Isolate peripheral blood mononuclear cells (PBMCs) from heparinized blood via density gradient centrifugation.
- B Cell Enrichment: Enrich memory B cells using negative selection magnetic bead kits.
- Stimulation: Culture cells with R848 (TLR7/8 agonist) and recombinant IL-2 for 3-4 days to differentiate memory B cells into antibody-secreting cells (ASCs).
- ELISpot Assay: Transfer stimulated cells to PVDF plates coated with target antigen or anti-immunoglobulin for total IgG.
- Development: After overnight incubation, detect ASCs using biotinylated anti-human IgG, followed by streptavidin-alkaline phosphatase and BCIP/NBT substrate.
- Quantification: Count spots using an automated ELISpot reader. Frequency is expressed as antigen-specific ASCs per million input memory B cells.
Visualization of Key Concepts
Title: Immune Durability Pathways: Live vs. Inactivated Vaccines
Title: Experimental Workflow for Assessing Immune Durability
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Durability Studies
| Item | Function in Research | Example/Note |
|---|---|---|
| Recombinant Viral Antigens | Coating antigen for ELISA; target for B cell assays. | e.g., SARS-CoV-2 S1/RBD protein, Influenza HA. |
| HRP-conjugated Anti-Human IgG | Detection antibody for ELISA quantifying humoral response. | Must match host species of primary sample. |
| Human IFN-γ/IL-4 ELISpot Kits | Quantify antigen-specific T cell responses. | Critical for assessing cellular memory. |
| Memory B Cell Isolation Kits | Negative magnetic selection for untouched human memory B cells. | Essential for ELISpot or in vitro stimulation assays. |
| R848 (Resiquimod) & IL-2 | In vitro polyclonal stimulation of memory B cells to differentiate into ASCs. | Standard cocktail for memory B cell ELISpot. |
| Fluorochrome-labeled mAbs for Flow | Phenotyping lymphocytes (e.g., CD19, CD27, CD38, IgD). | Identify plasmablasts, memory B cell subsets. |
| Cryopreservation Media | Long-term storage of PBMCs/sera for longitudinal studies. | Contains DMSO and FBS for cell viability. |
| Neutralizing Antibody Assay Kits | Pseudovirus or authentic virus neutralization tests. | Functional correlate of protection assessment. |
| Adjuvant Systems (Research) | Formulate inactivated antigens to enhance immunogenicity (e.g., Alum, AS01). | Comparator for platform studies. |
Within the ongoing research thesis comparing live attenuated and inactivated vaccine platforms, the speed at which a vaccine confers protective immunity is a critical parameter for outbreak response. This guide objectively compares the onset of protection elicited by these two major platform classes, supported by experimental data.
Comparative Onset of Protection: Key Data
The following table summarizes findings from recent studies on immune kinetics post-vaccination for representative pathogens.
Table 1: Comparative Onset of Protective Immunity Post-Vaccination
| Vaccine Platform | Example Vaccine (Pathogen) | First Detectable Neutralizing Ab (Avg. Days Post-Vaccination) | Cell-Mediated Immunity (CMI) Detection (Avg. Days) | Estimated Onset of Significant Protection (From Challenge Studies) | Key Supporting References |
|---|---|---|---|---|---|
| Live Attenuated | Measles, Mumps, Rubella (MMR) | 12-14 days | 7-10 days (T-cell activation) | ~12-14 days | (PMID: 32381787, PMID: 35062756) |
| Live Attenuated | Yellow Fever (YF-17D) | 7-10 days | 5-7 days (CD8+ T-cells) | ~7-10 days | (PMID: 25802264, PMID: 37126542) |
| Inactivated/Subunit | Inactivated Influenza (Seasonal) | 14-21 days | 14-21 days (Limited CD8+) | ~14-21 days | (PMID: 33115846) |
| Inactivated/Subunit | Tetanus Toxoid | 14-28 days (booster-dependent) | Primarily Th2, slower | ~14-28 days | (PMID: 28939734) |
| mRNA Platform | SARS-CoV-2 (BNT162b2) | 10-14 days post-dose 1 | 10-14 days (Strong CD4+/CD8+) | ~10-14 days post-dose 1 | (PMID: 33301246, PMID: 35982687) |
Experimental Protocols for Measuring Onset
Protocol 1: Longitudinal Immune Correlates Assessment
Objective: To kinetically track humoral and cellular immune parameters post-vaccination to define correlates of early protection. Methodology:
- Cohort & Vaccination: Enroll seronegative subjects (n≥20 per vaccine group). Administer a single dose of live attenuated vaccine (e.g., YF-17D) or a prime dose of inactivated vaccine.
- Sample Collection: Collect peripheral blood mononuclear cells (PBMCs) and serum at baseline (Day 0) and on Days 3, 5, 7, 10, 14, 21, and 28 post-vaccination.
- Humoral Response: Measure antigen-specific IgG and neutralizing antibody titers via ELISA and microneutralization/plaque reduction assays, respectively.
- Cell-Mediated Response:
- IFN-γ ELISpot: Stimulate PBMCs with vaccine antigen peptides. Spot-forming units (SFUs) indicate antigen-specific T-cell frequency.
- Multiparametric Flow Cytometry: Use intracellular cytokine staining (ICS) to characterize CD4+ and CD8+ T-cell subsets (IFN-γ, TNF-α, IL-2) and memory phenotypes.
- Data Analysis: Plot kinetic curves for each parameter. The "onset" is defined as the first time point where measurements significantly exceed baseline (p<0.05) and continue an upward trajectory.
Protocol 2: Challenge Study in Animal Models
Objective: To empirically determine the earliest time post-vaccination when protection against live pathogen challenge is achieved. Methodology:
- Animal Groups: Randomize animals (e.g., mice, non-human primates) into vaccine groups (live attenuated vs. inactivated) and a naive control group (n=8-10).
- Vaccination: Administer vaccine at Day 0.
- Sequential Challenge: Subgroups from each vaccine group are challenged with a standardized dose of virulent pathogen at sequential time points post-vaccination (e.g., Days 3, 5, 7, 10, 14).
- Outcome Measures: Monitor for clinical signs, viremia/bacterial load (via qPCR/culture), and survival for a defined period post-challenge.
- Analysis: Compare disease metrics between vaccinated and naive animals at each challenge time point. The earliest time point with statistically significant reduction in pathogen load or morbidity defines the onset of protection.
Visualizing Immune Kinetics and Experimental Design
Title: Immune Kinetics Comparison: LAV vs. Inactivated Vaccines
Title: Experimental Workflow: Measuring Onset of Protection
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Onset of Protection Studies
| Reagent / Material | Function in Research | Example Vendor/Product Code (Illustrative) |
|---|---|---|
| PBMC Isolation Kits | Isolation of viable lymphocytes from whole blood for cellular assays. | Ficoll-Paque PLUS (Cytiva), Lymphoprep (Stemcell) |
| ELISpot Kits (IFN-γ, IL-4, etc.) | Quantification of antigen-specific T-cells secreting specific cytokines. | Human IFN-γ ELISpotPRO (Mabtech), Mouse IFN-γ ELISpot (BD Biosciences) |
| Fluorochrome-conjugated Antibody Panels | Multiparametric flow cytometry to phenotype and characterize immune cells (T-cell subsets, activation markers, intracellular cytokines). | Antibodies from BioLegend, BD Biosciences, Thermo Fisher |
| Recombinant Vaccine Antigen / Peptide Pools | Stimulation antigen for in vitro assays (ELISpot, ICS) to measure antigen-specific responses. | Custom synthesis (Genscript, Pepscan), commercial recombinant proteins (Sino Biological) |
| Virus Neutralization Assay Reagents | Measuring functional, protective antibody titers (e.g., luciferase reporter viruses, cell lines). | Neutralizing Ab Assay Kits (e.g., for SARS-CoV-2, Influenza) |
| Pathogen-Specific qPCR Assays | Quantifying viral/bacterial load in challenge studies to assess protection efficacy. | Custom TaqMan assays (Thermo Fisher), pre-developed assays (IDT) |
| Adjuvant Systems (for inactivated studies) | Enhancing immunogenicity of inactivated/subunit vaccines in preclinical models. | AddaVax (MF59-like), AS01, CpG ODN (InvivoGen) |
This guide presents objective comparisons within the framework of live attenuated versus inactivated vaccine platform research. Data is synthesized from current literature and clinical studies.
OPV vs. IPV: A Platform Comparison
The poliovirus vaccines represent a classic direct comparison of live attenuated (Oral Polio Vaccine, OPV) and inactivated (Inactivated Polio Vaccine, IPV) platforms.
Comparative Performance Data
Table 1: Key Characteristics of OPV and IPV
| Parameter | Oral Polio Vaccine (OPV) – Live Attenuated | Inactivated Polio Vaccine (IPV) |
|---|---|---|
| Route of Administration | Oral | Intramuscular/Subcutaneous |
| Mucosal Immunity | Induces strong intestinal IgA, prevents replication and transmission. | Limited; primarily systemic, minimal impact on gut replication. |
| Herd Protection | Excellent, via secondary spread of attenuated virus. | Limited, protects individual but does not prevent silent circulation. |
| Duration of Immunity | Long-lasting, likely lifelong. | Long-lasting, requires boosters. |
| Risk of VAPP | ~2-4 cases per million births in naive populations. | None. |
| Risk of cVDPV | Yes, associated with prolonged circulation/reversion. | No. |
| Thermal Stability | Requires cold chain; sensitive to heat. | More stable. |
| Manufacturing Cost | Low | Higher |
VAPP: Vaccine-Associated Paralytic Poliomyelitis; cVDPV: Circulating Vaccine-Derived Poliovirus.
Experimental Protocol: Shedding and Transmission Study
A key experiment differentiating the platforms measures viral shedding post-challenge.
- Objective: To compare the ability of OPV and IPV to prevent fecal shedding of wild-type poliovirus after challenge.
- Groups: Randomized cohorts of IPV-vaccinated, OPV-vaccinated, and naive control subjects.
- Challenge: All subjects receive a monovalent Oral Polio Vaccine (mOPV) challenge dose (mimicking wild-type exposure).
- Sample Collection: Stool samples collected daily for 4 weeks post-challenge.
- Analysis: Virus isolation in cell culture (e.g., L20B cells expressing the poliovirus receptor) and quantification via plaque assay or PCR.
- Outcome Measure: Duration and titer of poliovirus shedding. OPV-vaccinated individuals typically show no or minimal shedding, while IPV-vaccinated individuals shed virus for a shorter duration than controls but longer than OPV recipients.
Experimental Workflow for Polio Shedding Study
LAIV vs. IIV: Influenza Vaccine Comparison
Live Attenuated Influenza Vaccine (LAIV) and Inactivated Influenza Vaccine (IIV) provide a contemporary platform comparison.
Comparative Performance Data
Table 2: Key Characteristics of LAIV and IIV
| Parameter | Live Attenuated Influenza Vaccine (LAIV) | Inactivated Influenza Vaccine (IIV) |
|---|---|---|
| Route | Intranasal | Intramuscular |
| Immune Response | Induces mucosal IgA, serum IgG, and cell-mediated immunity. | Primarily serum IgG; limited CD8+ T-cell response. |
| Breadth of Immunity | Broader cross-reactive T-cell and antibody responses. | Narrower, largely strain-specific antibody response. |
| Efficacy in Children | Historically superior to IIV in naive children. | Standard efficacy. |
| Efficacy in Adults | Generally non-inferior or slightly inferior to IIV. | Robust, considered the standard. |
| Virus Shedding | Detectable, attenuated virus for limited duration. | None. |
| Manufacturing | Egg-based or cell-culture; cold-adapted strains. | Egg-based, cell-culture, or recombinant; virus inactivated. |
Experimental Protocol: Mucosal IgA and Protection Challenge Study
A critical comparison involves assessing correlates of protection at the mucosal site of infection.
- Objective: Compare LAIV and IIV in their ability to induce nasal IgA and protect against experimental human challenge.
- Groups: Healthy adults randomized to receive seasonal LAIV, IIV, or placebo.
- Pre-challenge Sampling: Nasal washes performed at baseline and 4 weeks post-vaccination.
- Assay: Nasal wash IgA specific for vaccine-homologous and drifted influenza strains measured by ELISA.
- Challenge: Subjects challenged intranasally with a wild-type influenza virus (under controlled conditions) 4-6 weeks post-vaccination.
- Outcome Measures: Infection rate (viral shedding by PCR), symptom scores, and correlation with pre-challenge nasal IgA titer.
LAIV vs IIV Challenge Study Design
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for Vaccine Platform Comparison Studies
| Reagent/Material | Function in Research | Example/Notes |
|---|---|---|
| Poliovirus Receptor-Expressing Cell Lines (e.g., L20B, RD) | Essential for isolating and quantifying poliovirus from clinical samples (stool, nasal secretions). | L20B (mouse cells expressing human PVR) is highly specific for poliovirus. |
| Plaque Assay Reagents | Gold-standard quantitative method for measuring infectious virus titer. | Requires semi-solid overlay (e.g., carboxymethylcellulose) and staining (crystal violet). |
| Virus Transport Media (VTM) | Preserves viability of virus in clinical specimens during transport and storage. | Contains protein stabilizers and antibiotics. |
| ELISA Kits (Virus-Specific IgA/IgG) | Quantifies antigen-specific antibody responses in serum and mucosal secretions. | Requires matched capture antigen (e.g., purified influenza HA or inactivated poliovirus). |
| qRT-PCR Master Mixes & Primers/Probes | For sensitive, quantitative detection of viral RNA (shedding, viremia). | Targets conserved regions (e.g., poliovirus 5' UTR, influenza matrix gene). |
| PBMC Isolation Kits (Ficoll-Paque) | Isolate peripheral blood mononuclear cells for assessment of cellular immunity (ELISpot, flow cytometry). | Critical for comparing T-cell responses between vaccine platforms. |
| ELISpot Kits (IFN-γ, etc.) | Measures the frequency of antigen-specific T-cells responding to vaccine antigens. | Uses virus-specific peptide pools or whole inactivated virus. |
| Cold-Adapted Influenza Virus Strains | Backbone for LAIV production; used in challenge models to assess attenuation. | Temperature-sensitive, restricted to replication in the URT. |
Conclusion
The choice between live attenuated and inactivated vaccine platforms is not a binary superiority contest but a strategic decision based on pathogen biology, target population, and desired immune profile. Live attenuated vaccines generally induce robust, durable, and broad cellular immunity but carry contraindications. Inactivated vaccines offer superior safety for immunocompromised individuals and easier storage but often require adjuvants and boosters for sustained protection. Future directions hinge on technological convergence, such as using inactivation methods on novel viral vectors or employing attenuation techniques for historically challenging pathogens. The next generation of vaccines will likely employ tailored platform combinations or engineered chimeric designs, informed by this foundational comparison, to address emerging infectious diseases and improve global vaccine equity and efficacy.