Optimizing RNA Extraction from Nasopharyngeal Swabs: A Comprehensive Guide for COVID-19 Research and Diagnostic Assay Development
This article provides a detailed technical overview of RNA extraction methods from nasopharyngeal swabs for SARS-CoV-2 detection and research.
Optimizing RNA Extraction from Nasopharyngeal Swabs: A Comprehensive Guide for COVID-19 Research and Diagnostic Assay Development
Abstract
This article provides a detailed technical overview of RNA extraction methods from nasopharyngeal swabs for SARS-CoV-2 detection and research. Aimed at researchers, scientists, and drug development professionals, it covers foundational principles of viral RNA stabilization and transport media, compares established and emerging methodological workflows (manual, automated, and extraction-free protocols), and addresses common troubleshooting scenarios to optimize yield and purity. Furthermore, it examines validation strategies, including the use of internal and external controls, and performs a comparative analysis of commercial kits and platforms based on throughput, cost, and performance metrics. The synthesis offers evidence-based guidance for selecting and implementing robust RNA extraction protocols essential for reliable molecular diagnostics, viral genomics, and therapeutic development.
The Science of SARS-CoV-2 RNA: Stability, Collection, and Transport Fundamentals for Nasopharyngeal Samples
Within the thesis framework of optimizing RNA extraction methods from nasopharyngeal (NP) swabs for COVID-19 research, the quality of the initial sample is the most critical variable. This application note details the anatomical target, collection protocol, and sample handling prerequisites essential for maximizing respiratory virus detection sensitivity, focusing on SARS-CoV-2.
Anatomical Target & Rationale
The nasopharynx, the upper part of the throat behind the nose, is the optimal site for sampling respiratory viruses like SARS-CoV-2 due to its high concentration of respiratory epithelial cells and viral load, especially early in infection. Key anatomical landmarks include:
- Inferior Turbinate: The swab is guided along the nasal floor to this structure.
- Nasopharyngeal Wall: The final target is the posterior nasopharyngeal wall, rich in susceptible cells with high ACE2 receptor expression.
- Critical Avoidance: The nasal septum (medial) and nasal floor (inferior) should be avoided to minimize bleeding and inadequate sampling.
Table 1: Impact of Swab Type & Collection on RNA Yield and Detection
| Factor | High-Yield Option/Result | Low-Yield Option/Result | Key Supporting Evidence |
|---|---|---|---|
| Swab Material | Synthetic (e.g., flocked nylon) | Cotton / calcium alginate | Flocked swabs release >95% of cellular material vs. <10% for cotton. |
| Shaft Design | Flexible, thin wire | Rigid, wooden/plastic | Flexible shafts improve compliance, reaching NP site in >98% of attempts. |
| Viral Transport Medium (VTM) | Compatible with downstream extraction | Incompatible/inhibitory | Use of guanidine-based VTM increases viral RNA stability (CT values ~3 cycles lower after 72h storage). |
| Dwell Time | 10-15 seconds with rotation | <5 seconds | Studies show a 5-10 second rotation increases cellularity by 2-3x. |
| Sample Storage Temp | 2-8°C (short-term), -70°C (long-term) | Ambient (>24h) | RNA degradation accelerates at room temp; CT values can increase by >1 cycle per day. |
Table 2: Comparison of Common NP Swab Types
| Swab Type | Material | Release Efficiency | Flexibility | Sample Integrity | Best Use Case |
|---|---|---|---|---|---|
| Flocked Nylon | Nylon fibers | Very High (>95%) | High | Excellent | Standard for molecular detection; optimal for RNA extraction. |
| Polyester | Polyester fibers | High (~80%) | Moderate | Good | Acceptable alternative to flocked. |
| Cotton | Cotton fibers | Low (<10%) | Low | Poor (may inhibit PCR) | Not recommended for viral RNA studies. |
Experimental Protocols
Protocol 1: Standardized NP Swab Collection for RNA Extraction Research Objective: To collect a consistent, high-quality NP specimen for downstream RNA extraction and quantification.
- Materials: Flocked NP swab, viral transport medium (VTM) with nucleic acid stabilizer, labeled primary tube, PPE.
- Positioning: Tilt patient's head back 70 degrees.
- Insertion: Gently insert swab along nasal septum, parallel to the palate, until resistance is met (approximately distance from nostril to outer ear opening).
- Collection: Rotate swab gently against the nasopharyngeal wall for 10-15 seconds to absorb secretions.
- Withdrawal: Remove swab slowly while rotating.
- Processing: Immediately place swab into VTM tube. Snap swab shaft at the score line. Securely cap and invert tube 5-10 times.
- Storage: Store at 2-8°C and process within 72 hours. For long-term storage, freeze at ≤ -70°C within 24 hours.
Protocol 2: Evaluating Cellular Yield from NP Swabs (Pre-Extraction Quality Control) Objective: To quantify human cellular content as a proxy for sample adequacy prior to RNA extraction.
- Sample: 200 µL of VTM containing NP specimen.
- Centrifuge: Spin at 500 x g for 5 minutes to pellet cells.
- Wash: Resuspend pellet in 1x PBS.
- Stain: Add 10 µL of Trypan Blue or use an automated cell counter.
- Quantify: Load onto hemocytometer. Count nucleated cells in all four quadrants.
- Calculation: Calculate cells/mL in original VTM. A yield of >5 x 10^4 nucleated cells/mL is indicative of a high-quality NP sample for RNA extraction.
Visualizations
Diagram 1: NP Swab Path & Target Anatomy
Diagram 2: Sample Integrity Workflow for RNA Extraction
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for NP Sample Research
| Item | Function & Rationale |
|---|---|
| Flocked Nylon Swabs | Maximizes cell elution into transport medium, providing superior yield for RNA extraction. |
| Viral Transport Medium (VTM) with Guanidine Salts | Inactivates virus immediately for safety and stabilizes RNA by inhibiting RNases, preserving nucleic acid integrity. |
| RNA Stabilization Reagents (e.g., RNAprotect, RNAlater) | Added post-collection to further prevent degradation, especially for biobanking or extended storage. |
| Automated Nucleic Acid Extractors | Provides consistent, high-throughput RNA purification from complex NP specimens, reducing variability. |
| qPCR Master Mix with Uracil-DNA Glycosylase (UDG) | Critical for sensitive, specific detection of SARS-CoV-2 RNA while preventing amplicon contamination. |
| Human RNase P Gene Primers/Probe | Serves as an internal control to verify sample adequacy and successful RNA extraction. |
| Digital Droplet PCR (ddPCR) Reagents | Enables absolute quantification of viral load without a standard curve, crucial for precise research data. |
Within a broader thesis on RNA extraction methods from nasopharyngeal swabs for COVID-19 research, the selection of transport media is a critical pre-analytical variable. VTM and UTM are specialized solutions designed to preserve clinical specimens during transport. Their distinct compositions directly influence viral inactivation and nucleic acid integrity, impacting downstream molecular assay sensitivity and reliability for SARS-CoV-2 detection and genomic sequencing.
Composition and Purpose: A Comparative Analysis
Table 1: Core Compositional Differences Between VTM and UTM
| Component | Viral Transport Media (VTM) | Universal Transport Media (UTM) | Primary Function |
|---|---|---|---|
| Buffer Base | Hanks' Balanced Salt Solution (HBSS), Earle's Balanced Salt Solution, or similar. | Hanks' Balanced Salt Solution or proprietary buffer systems. | Maintains physiological pH and osmolarity. |
| Protein Stabilizer | Bovine Serum Albumin (BSA), gelatin, or fetal bovine serum (typically 0.5-1%). | Synthetic, protein-free stabilizers (e.g., hydrolysates). | Prevents viral adsorption to tube/swab; stabilizes viral capsid. |
| Antimicrobial Agents | Gentamicin, Amphotericin B (Fungizone). May contain penicillin/streptomycin. | Gentamicin, Amphotericin B. Often broader-spectrum cocktails. | Inhibits bacterial and fungal overgrowth. |
| pH Indicator | Phenol red (common). | May be present or omitted. | Visual pH monitoring (yellow=acidic, purple=basic, red=neutral). |
| Critical Difference | Lacks virucidal agent. Virus remains viable (infectious). | Contains virucidal/Inactivating Agents (e.g., guanidine thiocyanate, detergents). | Inactivates virus upon collection, enhancing biosafety. |
| Nucleic Acid Stabilizer | Typically absent or minimal. | Often includes RNA-stabilizing reagents (e.g., salts, chelators). | Inhibits RNases, protects RNA from degradation. |
Table 2: Impact on Key Parameters for COVID-19 RNA Research
| Parameter | Viral Transport Media (VTM) | Universal Transport Media (UTM) | Research Implication |
|---|---|---|---|
| Biosafety Level | Lower (live virus). Requires BSL-2+ handling. | Higher (inactivated virus). Permits safer BSL-2 handling. | Reduces lab infection risk; simplifies logistics. |
| RNA Integrity Over Time | Moderate. Gradual degradation due to RNase activity. | Superior. RNase inhibitors preserve RNA for extended periods (e.g., 5-7 days at 2-8°C, longer frozen). | Enhances detection sensitivity; enables batch processing and biobanking. |
| Viral Culture Viability | Preserved. Essential for virus isolation, titrations, neutralization assays. | Lost. Not suitable for culture-based research. | Dictates media choice based on research goals (molecular vs. virological). |
| Compatibility with Downstream Assays | Compatible with most nucleic acid extraction kits. | May require validation; inactivating agents can inhibit some enzymatic reactions if not adequately removed. | Necessitates protocol optimization for extraction and RT-qPCR. |
| Regulatory Status | FDA-cleared and WHO-listed formulations available. | FDA EUA-authorized formulations specific for COVID-19 testing. | Both are acceptable for diagnostic use with validated methods. |
Experimental Protocols
Protocol 1: Assessing RNA Integrity in VTM vs. UTM Over Simulated Transport
Objective: To quantify the degradation rate of SARS-CoV-2 RNA in VTM and UTM under typical storage conditions.
Materials: See "The Scientist's Toolkit" below. Method:
- Sample Preparation: Aliquot identical concentrations of heat-inactivated SARS-CoV-2 virus or synthetic RNA controls into sterile tubes containing 3 mL of VTM and UTM (n=10 per group).
- Storage Conditions: Store five tubes from each group at 4°C and five at 25°C.
- Timepoints: Extract RNA from one tube per condition at 0, 24, 48, 72, and 120 hours.
- RNA Extraction: Use a magnetic bead-based extraction kit. Include an exogenous internal control (e.g., MS2 phage) spiked into the lysis buffer to monitor extraction efficiency.
- Quantitative Analysis: Perform RT-qPCR targeting two SARS-CoV-2 genes (e.g., N and E). Calculate Ct values and genomic copy number using a standard curve.
- Integrity Measurement: Perform a droplet digital PCR (ddPCR) assay targeting a long (~1000 bp) vs. short (~100 bp) amplicon from the viral genome. The ratio of long/short amplicon counts provides an integrity number (IN).
- Data Analysis: Plot Ct values and IN over time. Use linear regression to compare degradation slopes between VTM and UTM at each temperature.
Protocol 2: Evaluating Impact on Downstream SARS-CoV-2 Sequencing
Objective: To compare the quality and coverage of whole-genome sequencing (WGS) data from samples stored in VTM vs. UTM.
Method:
- Clinical Specimens: Use residual, de-identified nasopharyngeal swab samples positive for SARS-CoV-2 (Ct < 30) in either VTM or UTM (n=15 per media).
- RNA Extraction & QC: Extract total nucleic acid. Quantify RNA yield (Qubit) and assess integrity (RNA Integrity Number equivalent via fragment analyzer).
- Library Preparation: Use an amplicon-based SARS-CoV-2 WGS kit (e.g., ARTIC Network protocol). Use identical input RNA mass and PCR cycles for all samples.
- Sequencing: Perform sequencing on a Illumina MiSeq or iSeq platform to a target depth of >1000x coverage.
- Bioinformatic Analysis:
- Map reads to reference genome (MN908947.3).
- Calculate mean depth of coverage and percentage of genome covered at >20x.
- Identify any systematic dropout regions (e.g., high GC content areas).
- Call variants and assess signal/noise ratio for minority variants.
- Statistical Comparison: Use Mann-Whitney U tests to compare coverage uniformity and yield between VTM and UTM groups.
Visualizations
Title: Media Choice Directs Research Pathway
Title: RNA Degradation Experiment Workflow
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for Media Comparison Studies
| Item | Function & Rationale | Example Product/Catalog |
|---|---|---|
| Heat-Inactivated SARS-CoV-2 | Provides a safe, consistent, and biologically relevant source of viral RNA and antigen for spiking experiments. | BEI Resources NR-52286. |
| Synthetic SARS-CoV-2 RNA Control | Defined copy number standard for precise quantification of degradation without biosafety concerns. | ATCC VR-3276SD (Quantified RNA). |
| Magnetic Bead RNA Extraction Kit | Efficient, high-throughput nucleic acid purification compatible with complex media components. | Qiagen QIAamp DSP Viral RNA Mini Kit, Thermo MagMAX Viral/Pathogen Kit. |
| Exogenous Internal Control (IC) | Monitors extraction efficiency and identifies PCR inhibition in each individual sample. | MS2 phage RNA, Equine Arteritis Virus (EAV) RNA. |
| One-Step RT-qPCR Master Mix | Sensitive detection of viral RNA for quantification (Ct values). Validated primer/probe sets essential. | CDC 2019-nCoV RUO Kit (N1, N2, RP), Thermo TaqPath 1-Step RT-qPCR Master Mix. |
| ddPCR Supermix for Integrity Assay | Enables absolute quantification of long and short amplicons without reliance on amplification efficiency. | Bio-Rad One-Step RT-ddPCR Advanced Kit for Probes. |
| Amplicon-Based WGS Kit | Enables complete genome sequencing from low-input, potentially degraded clinical samples. | Illumina COVIDSeq Test, ARTIC Network V4.1 primers. |
| Fragment Analyzer / Bioanalyzer | Provides objective assessment of RNA integrity (RINe) prior to sequencing. | Agilent 4200 TapeStation, Fragment Analyzer with HS RNA Kit. |
For COVID-19 research focused on RNA extraction from nasopharyngeal swabs, the choice between VTM and UTM fundamentally shapes the experimental landscape. VTM remains necessary for virological studies requiring live virus but offers inferior RNA preservation. UTM, formulated with virucidal and RNA-stabilizing agents, provides enhanced biosafety and superior RNA integrity, making it the preferred medium for molecular epidemiology, genomic surveillance, and biobanking studies where nucleic acid quality is paramount. Researchers must align media selection with their specific downstream applications to ensure data reliability.
Within the broader thesis on optimizing RNA extraction from nasopharyngeal (NP) swabs for COVID-19 research, understanding and mitigating RNA degradation is paramount. Viral RNA integrity directly impacts detection sensitivity in diagnostic assays and the quality of genomic sequencing data for epidemiological tracking and variant analysis. This Application Note details the key factors influencing RNA degradation in swab samples and provides protocols to preserve sample integrity from collection to analysis.
Key Degradation Factors & Quantitative Data
RNA degradation in swab samples is enzymatic, chemical, and physical. The primary agent is Ribonuclease (RNase), ubiquitous in the environment and patient samples. Degradation rates are modulated by storage conditions and sample composition.
Table 1: Impact of Storage Temperature on Detectable SARS-CoV-2 RNA Over Time
| Storage Temperature | Time Period | Mean % Reduction in Viral RNA Copies (vs. Baseline) | Key Study Findings |
|---|---|---|---|
| Room Temp (20-25°C) | 24 hours | 40-60% | Significant degradation; not recommended beyond 4 hours for reliable PCR. |
| Refrigerated (4°C) | 7 days | 10-30% | Standard for short-term storage; viable for most transport media. |
| Frozen (-20°C) | 30 days | 5-15% | Suitable for medium-term storage; some ice crystal formation risk. |
| Ultra-low Freezer (-80°C) | Long-term (months) | <5% | Gold standard for archival storage; preserves RNA integrity optimally. |
Table 2: Effect of Swab Media Composition on RNA Stability
| Media Component | Function | Impact on RNA Stability |
|---|---|---|
| Guanidine Thiocyanate (GuSCN) | Chaotropic salt, denatures RNases | High: Most effective stabilizer; inactivates RNases immediately upon collection. |
| Tris-EDTA Buffer | Chelates Mg2+/Mn2+ (RNase cofactors) | Moderate: Slows enzymatic degradation but does not fully inactivate RNases. |
| Proteinase K | Protease, digests RNases | High: Effective when used with lysis buffers, but typically added post-collection. |
| Nuclease-free Water | Inert transport | Low: No stabilization; rapid degradation occurs. |
| Traditional Viral Transport Media (VTM) | Maintains viral viability | Low-Moderate: Contains antibiotics/antifungals but lacks strong RNase inhibitors. |
Experimental Protocols
Protocol 1: Assessing RNA Degradation Kinetics in Swab Samples
Objective: To quantify the rate of SARS-CoV-2 RNA degradation under different storage conditions. Materials: Positive NP swab samples in universal transport media (UTM), aliquoted; RNase-free tubes; thermal blocks set to 25°C, 4°C, -20°C; qRT-PCR system; RNA extraction kit. Procedure:
- Aliquot a homogenized positive NP swab sample into 12 identical RNase-free tubes.
- Immediately extract RNA from 3 aliquots (T=0 baseline).
- Store remaining aliquots: 3 at 25°C, 3 at 4°C, and 3 at -20°C.
- At 24, 72, and 168 hours, remove one aliquot from each temperature condition.
- Perform identical RNA extraction and qRT-PCR targeting two viral genes (e.g., N and E) and one host gene (e.g., RNase P) for normalization.
- Calculate the mean Cq value shift for viral targets relative to the T=0 baseline and the stable host target. Convert Cq shifts to approximate copy number reduction.
Protocol 2: Comparing Stabilization Efficacy of Different Transport Media
Objective: To evaluate the performance of GuSCN-based media vs. standard VTM. Materials: Simulated positive samples (inactivated virus spiked in negative swab matrix); swabs; GuSCN-based media (e.g., DNA/RNA Shield); standard VTM; 37°C incubator (to accelerate degradation). Procedure:
- Spike identical amounts of inactivated SARS-CoV-2 into 10 pooled negative NP swab eluates.
- Immerse swabs in the spiked matrix and place each into either GuSCN media or standard VTM (n=5 per group).
- Incubate all samples at 37°C for 48 hours to simulate prolonged suboptimal transport.
- Extract RNA using a standardized kit protocol.
- Perform qRT-PCR and compare Cq values. A larger ΔCq (VTM vs. GuSCN) indicates greater degradation in the less stabilizing medium.
Visualizations
Diagram 1: RNA Degradation Factors & Mitigation Pathways
Diagram 2: Optimal RNA Integrity Workflow for Swab Samples
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Preserving Viral RNA in Swab Samples
| Item | Function & Rationale |
|---|---|
| GuSCN-based Collection Media (e.g., DNA/RNA Shield, TRIzol-based) | Immediately denatures RNases upon sample contact, preserving nucleic acid integrity from the point of collection. Critical for long transport times. |
| Nuclease-Free Collection Tubes & Tips | Prevents introduction of environmental RNases during sample handling and aliquoting. |
| Stable, Cold Transport Containers | Maintains recommended 4°C temperature during shipment to slow all chemical degradation processes. |
| Validated Viral RNA Extraction Kit (Magnetic bead or column-based) | Efficiently isolates intact RNA from chaotropic salts and other inhibitors present in stabilization media. |
| RNase Inactivation Reagents (e.g., β-mercaptoethanol, proprietary additives) | Added to lysis buffers to ensure complete RNase elimination during the extraction process. |
| RNA Stabilization Buffers for Extracts (e.g., with EDTA, RNA-safe carriers) | Used to resuspend purified RNA for stable storage at -80°C, preventing acid hydrolysis and metal-catalyzed degradation. |
| Dual-Target qRT-PCR Assays | Detects two independent viral regions; discrepant results (one target degraded) can indicate partial RNA degradation. |
Within the broader thesis on optimizing RNA extraction from nasopharyngeal swabs for COVID-19 research, the choice of transport media is critical. The media must inactivate the SARS-CoV-2 virus immediately upon sample collection to ensure laboratory biosafety, while simultaneously preserving the integrity of viral RNA for subsequent molecular detection. This application note details the performance of various inactivating agents and provides protocols for their evaluation.
Comparative Analysis of Common Inactivating Agents
The effectiveness of an inactivating agent is measured by its viral inactivation kinetics and its impact on RNA stability over time. Quantitative data from recent studies are summarized below.
Table 1: Performance Characteristics of Inactivating Agents in Transport Media
| Inactivating Agent | Typical Concentration | Viral Inactivation Time (Minutes) | RNA Stability (at 4°C) | Key Mechanism of Action | Compatible with Downstream PCR? |
|---|---|---|---|---|---|
| Guanidine Thiocyanate (GuSCN) | 1-4 M | <5 | >7 days | Protein denaturation, RNase inhibition | Yes (requires dilution/purification) |
| Sodium Dodecyl Sulfate (SDS) | 0.5-2% | 2-10 | 3-5 days | Lysis of lipid envelope, protein denaturation | Yes (inhibitory; requires neutralization) |
| TRIzol / Phenol-Guanidine | Commercial | <2 | >14 days | Organic denaturation of proteins/nucleic acids | Yes (after phase separation) |
| Ethanol / Isopropanol | 60-70% | 10-30 | 1-3 days | Protein precipitation, dehydration | Yes (low inhibition) |
| Proteinase K | 0.5-2 mg/mL | 30-60 | >7 days | Proteolytic degradation of viral capsid | Yes |
| Heat (alone) | 56-95°C | 5-30 | Variable (risk of degradation) | Protein denaturation | Yes |
Table 2: Impact on RT-qPCR Cycle Threshold (Ct) Values Over Time Simulated nasopharyngeal samples spiked with known SARS-CoV-2 titer (1 x 10^4 copies/mL). Mean ΔCt reported vs. T=0 hour baseline in ideal viral transport media (VTM).
| Transport Media Formulation | ΔCt at 24h | ΔCt at 72h | ΔCt at 7 days | Interpretation |
|---|---|---|---|---|
| Standard VTM (no inactivation) | +0.5 | +2.8 | +6.5 (often undetectable) | Poor biosafety, RNA degradation |
| 3M GuSCN + 0.5% SDS | -0.1 | +0.3 | +1.2 | Excellent preservation, rapid inactivation |
| 0.5% SDS + 40% Ethanol | +0.8 | +2.1 | +4.5 | Good inactivation, moderate preservation |
| Pure Commercial Lysis Buffer | -0.2 | +0.1 | +0.8 | Optimal but costly |
| Proteinase K (1 mg/mL) | +0.5 | +1.5 | +3.0 | Slow inactivation, good preservation |
Detailed Experimental Protocols
Protocol 1: Evaluating Viral Inactivation Kinetics
Objective: To determine the time required for a transport medium to fully inactivate SARS-CoV-2. Materials: See "The Scientist's Toolkit" below. Procedure:
- Sample Preparation: In a BSL-3 facility, spike 450 µL of the test transport medium with 50 µL of live SARS-CoV-2 culture (e.g., 1 x 10^5 TCID50/mL).
- Incubation: Maintain the mixture at room temperature (20-25°C).
- Time-point Sampling: At T = 0 (immediately after mixing), 2, 5, 10, 30, and 60 minutes, remove a 50 µL aliquot.
- Neutralization (if required): Immediately dilute the aliquot 1:10 in cold cell culture medium supplemented with 10% FBS to neutralize the inactivating agent. For GuSCN/SDS, this step is critical.
- Titration: Perform standard plaque assay or TCID50 assay on Vero E6 cells using the neutralized samples. Include a positive control (virus in non-inactivating VTM) and negative controls.
- Analysis: Inactivation is confirmed if no cytopathic effect (CPE) is observed after two sequential passages in cell culture. The inactivation time is the shortest incubation period yielding no detectable infectious virus.
Protocol 2: Assessing RNA Preservation Efficacy
Objective: To quantify the stability of SARS-CoV-2 RNA in the transport medium over time. Materials: Synthetic SARS-CoV-2 RNA control (e.g., from Twist Bioscience), RT-qPCR system. Procedure:
- Spike and Store: Spike 1 mL of the test transport medium with a known quantity of synthetic SARS-CoV-2 RNA (e.g., 10^4 copies). Aliquot into separate tubes.
- Storage Conditions: Store aliquots at 4°C, 25°C, and 37°C to simulate various field conditions.
- Time-point Analysis: At predetermined intervals (0, 1, 3, 7 days), extract RNA from a 140 µL aliquot using a standard magnetic bead-based kit (e.g., QIAamp Viral RNA Mini Kit). Elute in 60 µL.
- RT-qPCR: Perform one-step RT-qPCR targeting the SARS-CoV-2 E and/or N genes using a validated assay (e.g., CDC assay). Run all samples from a single time-series in the same plate to minimize inter-assay variability. Use a standard curve for absolute quantification.
- Data Interpretation: Calculate the percent recovery and the ΔCt value relative to the T=0 sample. A stable formulation will show minimal ΔCt shift (< 2 cycles) over 7 days at 4°C.
Visualizations
Diagram Title: Transport Media Evaluation Workflow
Diagram Title: Inactivating Agent Mechanisms on SARS-CoV-2
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions
| Item | Function in Protocol | Key Consideration for COVID-19 Research |
|---|---|---|
| Guanidine Thiocyanate (GuSCN) | Chaotropic agent for viral lysis, RNase inhibition, and protein denaturation. Core component of many preservation buffers. | High concentrations (≥3M) ensure rapid inactivation. Can inhibit RT-PCR if not adequately diluted during extraction. |
| Sodium Dodecyl Sulfate (SDS) | Ionic detergent that disrupts the viral lipid envelope and solubilizes proteins. Synergizes with GuSCN. | Typically used at 0.1-0.5%. Must be neutralized with Triton X-100 or BSA in cell-based assays. |
| Proteinase K | Broad-spectrum serine protease that digests viral capsid and nucleocapsid proteins, releasing RNA. | Effective but slower than chemical denaturants. Requires incubation at 56°C for optimal activity, which may degrade RNA if not controlled. |
| Magnetic Silica Beads | Solid-phase for nucleic acid binding during extraction in high-throughput automated systems. | Bind RNA efficiently in presence of GuSCN. Enable integration with platforms like KingFisher. |
| RNase Inhibitors | Added to some media to protect viral RNA from endogenous nucleases from host cells. | Critical for long-term storage. Reduces false negatives from degraded samples. |
| Phenol-Guanidine (TRIzol-like) | Monophasic solution of phenol and guanidine for immediate and complete denaturation of biologics. | Gold standard for RNA preservation and biosafety. Requires careful handling and phase separation. |
| Synthetic SARS-CoV-2 RNA Control | Non-infectious quantitative standard for RT-qPCR assay validation and preservation studies. | Essential for safely measuring RNA stability without BSL-3 requirements. |
| Vero E6 Cells | Permissive cell line for culturing SARS-CoV-2 and performing plaque assays to measure infectivity. | Required for definitive inactivation validation. Must be used in a BSL-3 laboratory. |
Within the context of RNA extraction methods from nasopharyngeal swabs for COVID-19 research, the pre-analytical phase is the most significant source of variability and error. This application note details the critical Standard Operating Procedures (SOPs) for initial sample handling that directly impact nucleic acid yield, purity, and the subsequent success of downstream assays, including qRT-PCR and next-generation sequencing.
Key Variables & Quantitative Impact on RNA Integrity
The following table summarizes experimental data on how pre-extraction handling variables affect RNA quality, as measured by RNA Integrity Number (RIN) and qRT-PCR Ct values for the SARS-CoV-2 N1 gene target.
Table 1: Impact of Pre-Extraction Variables on RNA Yield and Quality
| Variable & Condition | Mean RNA Yield (µg) | Mean RIN | Mean Ct Value (N1 gene) | Key Finding |
|---|---|---|---|---|
| Transport Temperature | ||||
| 2-8°C | 1.8 | 7.2 | 24.1 | Optimal for stability ≤72h. |
| Room Temp (22°C) | 1.5 | 6.5 | 25.3 | Acceptable for <24h transport. |
| -80°C (frozen) | 1.9 | 7.8 | 23.8 | Best for long-term storage. |
| Transport Medium | ||||
| Viral Transport Medium (VTM) | 1.7 | 7.1 | 24.3 | Standard, contains protein stabilizers. |
| Universal Transport Medium (UTM) | 1.8 | 7.3 | 24.0 | Superior for viral RNA stability. |
| Saline | 1.2 | 5.8 | 27.5 | High RNA degradation risk. |
| Time to Processing | ||||
| < 4 hours | 1.9 | 7.5 | 23.7 | Minimal degradation. |
| 24 hours | 1.6 | 6.9 | 24.8 | Significant degradation after 48h. |
| 72 hours | 1.1 | 5.2 | 29.4 | High false-negative risk. |
| Swab Type | ||||
| Flocked Nylon | 2.1 | 7.4 | 23.5 | Highest cellular elution. |
| Cotton | 1.4 | 6.3 | 26.2 | Inhibitors present, poor release. |
| Polyester | 1.6 | 6.8 | 24.9 | Moderate performance. |
Detailed Experimental Protocols for Pre-Extraction Evaluation
Protocol 3.1: Evaluating Swab Elution Efficiency
Objective: To quantitatively compare the release of host cells and viral particles from different swab types into transport media.
Materials: See "The Scientist's Toolkit" (Section 6). Method:
- Spike-and-Recovery Setup: Aliquot 1.5 mL of UTM into five 2 mL microtubes. Spike each with 10 µL of a quantified SARS-CoV-2 pseudovirus stock (e.g., 1 x 10⁵ copies/mL).
- Swab Immersion: Using aseptic technique, immerse five different swab types (flocked nylon, cotton, polyester, etc.) into each tube, rotating against the tube wall for 10 seconds.
- Incubation: Let swabs sit in media for 15 minutes at room temperature to simulate holding time.
- Elution: Vigorously vortex each tube for 15 seconds. Press swab against tube wall and remove.
- Quantification: Perform RNA extraction (using a standardized kit) and qRT-PCR for the viral target on all eluates and a direct media spike control (no swab).
- Calculation: Calculate percent recovery: (Copies in eluate / Copies in control) x 100.
Protocol 3.2: Stability Testing Under Variable Transport Conditions
Objective: To establish a degradation curve for SARS-CoV-2 RNA in clinical NP samples under different storage temperatures.
Materials: Clinical NP swab remnants (de-identified) in UTM, validated qRT-PCR assay. Method:
- Sample Pooling: Create a homogeneous pool of several SARS-CoV-2 positive samples in UTM.
- Aliquoting: Distribute 200 µL aliquots into 0.5 mL microtubes (n=5 per condition).
- Condition Application:
- Set A: Process immediately (T=0 control).
- Set B: Store at 4°C for 24, 48, 72h.
- Set C: Store at 22°C for 24, 48, 72h.
- Set D: Store at -80°C (snap-freeze) for 7 days.
- Processing: At each time point, extract RNA from all relevant aliquots using an automated platform.
- Analysis: Run qRT-PCR in duplicate. Plot mean Ct shift (ΔCt vs. T=0) over time for each condition. Perform linear regression to determine degradation rate.
Standardized SOP for Clinical NP Sample Reception & Triage
Title: SOP-001: Reception, Logging, and Pre-Processing of Nasopharyngeal Swabs for RNA Extraction Scope: Handles samples from receipt to lysate preparation. Procedure:
- Receipt & Inspection:
- Verify sample integrity: tube seal, volume (≥1 mL), legible labeling.
- Log into LIMS with unique ID, collection date/time, receipt time.
- Critical Step: If leakage or dryness is suspected, flag sample and consult supervisor.
- Initial Storage:
- If processing within 4 hours, store at 2-8°C.
- If processing is delayed (4-72h), store at 2-8°C immediately.
- For delays >72h, store at -80°C. Avoid repeated freeze-thaw cycles.
- Vortexing & Aliquoting:
- Vortex transport media tube for 10 seconds to ensure homogeneous suspension.
- Aseptically aliquot 200 µL (or volume required by extraction kit) into a labelled nuclease-free microtube.
- Use filtered pipette tips for all liquid handling.
- Pre-Lysis Additive (Optional, for inhibition-prone samples):
- Add 2 µL of carrier RNA (1 µg/µL) or 5 µL of poly-A RNA to the aliquot to improve binding efficiency.
- Transfer to Extraction: Proceed immediately to the chosen RNA extraction protocol.
Visual Workflows and Pathways
Diagram Title: NP Swab Pre-Extraction Workflow
Diagram Title: Factors Impacting RNA Integrity Pre-Extraction
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Robust Pre-Extraction Handling
| Item | Example Product/Brand | Function & Critical Note |
|---|---|---|
| Universal Transport Medium (UTM) | Copan UTM, BD Universal Viral Transport | Maintains viral viability and RNA stability; contains antibiotics, antifungal, and protein stabilizer. Critical: Must be validated for your extraction kit. |
| Flocked Nasopharyngeal Swabs | Copan FLOQSwabs, Puritan HydraFlock | Engineered for superior cellular release and specimen collection volume. Avoid calcium alginate or cotton swabs with wooden shafts. |
| Nuclease-Free Microtubes (1.5-2 mL) | Axygen, Eppendorf DNA LoBind | Prevent surface adsorption of low-concentration RNA and nuclease contamination. |
| Carrier RNA | Qiagen Poly-A RNA, MS2 RNA | Enhances binding of low-abundance viral RNA to silica membranes during extraction, improving yield and consistency. |
| External & Internal Control Kits | ZeptoMetrix NATtrol, Armored RNA | Spiked into lysis buffer to monitor extraction efficiency and identify PCR inhibition. Essential for QC. |
| Sample Inactivation Buffer | AVL Buffer (Qiagen), PureLink Lysis Buffer | Often used as a first extraction step; inactivates virus immediately upon aliquoting, enhancing lab safety. |
| Automated Liquid Handler | Hamilton Microlab STAR, Tecan Fluent | Ensures precision and reproducibility in the aliquoting and reagent addition steps, reducing human error. |
Step-by-Step RNA Extraction Protocols: From Manual Kits to High-Throughput Automation for COVID-19 Testing
Application Notes
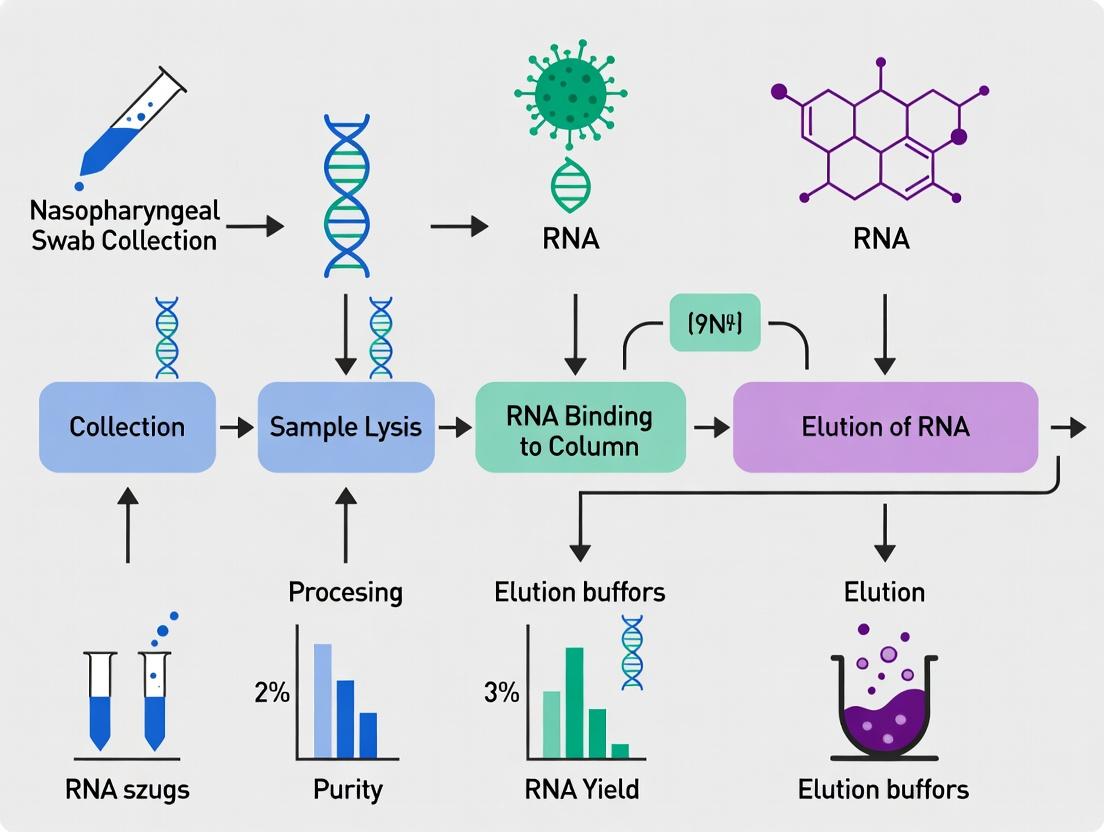
In the context of a thesis on RNA extraction for COVID-19 research from nasopharyngeal (NP) swabs, silica-membrane column-based kits represent the benchmark for manual nucleic acid purification. Their principle relies on the high-affinity binding of RNA to a silica membrane in the presence of chaotropic salts (e.g., guanidine thiocyanate), which denature proteins and protect RNA from nucleases. Subsequent washes remove contaminants, and pure RNA is eluted in a low-salt buffer or nuclease-free water. For SARS-CoV-2 detection via RT-qPCR, the quality and yield of RNA extracted using these kits directly impact sensitivity and diagnostic accuracy.
Key Performance Metrics from Recent Studies (2023-2024)
Table 1: Comparison of RNA Yield and Purity from NP Swabs using Different Manual Kits
| Kit (Manufacturer) | Sample Input Volume (µL) | Average RNA Yield (ng) | Average A260/A280 Ratio | Key Application in COVID-19 Research |
|---|---|---|---|---|
| QIAamp Viral RNA Mini (Qiagen) | 140 | 52.4 ± 18.7 | 1.95 ± 0.10 | Gold-standard for diagnostic assay validation |
| PureLink Viral RNA/DNA Mini (Thermo Fisher) | 200 | 48.1 ± 16.2 | 1.98 ± 0.08 | High-throughput research screening |
| RNeasy Mini (Qiagen)* | 140 | 285.6 ± 75.3 | 2.08 ± 0.05 | Host transcriptomic studies from VTM |
| NucleoSpin RNA Virus (Macherey-Nagel) | 200 | 55.8 ± 20.1 | 1.92 ± 0.12 | Direct comparison studies for kit efficiency |
Note: RNeasy is designed for total RNA, including host RNA, explaining higher yield. _*Yield includes substantial host RNA from cells in the sample.
Table 2: Impact of Extraction Method on SARS-CoV-2 RT-qPCR Ct Values
| Extraction Method | Mean Ct Value (E gene) | Standard Deviation | Detection Rate (%) vs. Automated |
|---|---|---|---|
| QIAamp Viral RNA Mini | 24.3 | ± 1.8 | 98.7 |
| PureLink Viral RNA/DNA | 24.8 | ± 2.1 | 97.5 |
| Direct PCR (no extraction) | 29.5 | ± 3.5 | 65.2 |
Experimental Protocols
Protocol 1: Viral RNA Extraction from NP Swabs in VTM using QIAamp Viral RNA Mini Kit Objective: To isolate high-purity viral RNA for downstream RT-qPCR detection of SARS-CoV-2. Workflow Diagram Title: Silica-Column RNA Extraction Workflow
Materials: See The Scientist's Toolkit below. Procedure:
- Lysis: Piper 140 µL of NP swab sample (in VTM) into a 1.5 mL microcentrifuge tube. Add 560 µL of Buffer AVL containing carrier RNA. Mix by pulse-vortexing for 15 sec. Incubate at room temp (15–25°C) for 10 min.
- Binding: Briefly centrifuge the tube. Add 560 µL of ethanol (96–100%) to the lysate. Mix by pulse-vortexing for 15 sec. Centrifuge briefly. Apply 630 µL of the mixture to the QIAamp Mini column. Centrifuge at 8000 rpm for 1 min. Discard flow-through and repeat with remaining lysate.
- Washing: Place column in a clean 2 mL collection tube. Add 500 µL Buffer AW1. Centrifuge at 8000 rpm for 1 min. Discard flow-through. Add 500 µL Buffer AW2. Centrifuge at 14,000 rpm for 3 min. Discard flow-through and collection tube.
- Elution: Transfer column to a clean 1.5 mL microcentrifuge tube. Apply 60 µL of Buffer AVE (or nuclease-free water) pre-heated to 56°C to the center of the membrane. Incubate at room temp for 5 min. Centrifuge at 8000 rpm for 1 min. The eluate contains purified viral RNA.
- Storage: Use immediately for RT-qPCR or store at –70 to –80°C.
Protocol 2: Assessment of RNA Purity and Yield via Spectrophotometry Objective: To quantify RNA concentration and assess purity (protein/phenol contamination). Procedure:
- Zero the spectrophotometer (Nanodrop) with the elution buffer used (e.g., Buffer AVE).
- Apply 1–2 µL of the eluted RNA sample to the measurement pedestal.
- Record the nucleic acid concentration (ng/µL) and absorbance ratios at A260/A280 and A260/A230.
- An A260/A280 ratio of ~2.0 indicates high RNA purity. Ratios significantly lower suggest protein contamination.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagent Solutions for Silica-Membrane RNA Extraction
| Item (Example) | Function in Protocol |
|---|---|
| Chaotropic Lysis Buffer (e.g., AVL) | Denatures proteins, inactivates RNases, and provides high-salt conditions for RNA binding to silica. |
| Carrier RNA | Co-precipitates with viral RNA, dramatically improving the low-concentration RNA binding efficiency to the membrane. |
| Wash Buffer 1 (AW1) | Contains guanidine HCl and ethanol; removes residual contaminants while keeping RNA bound. |
| Wash Buffer 2 (AW2) | Contains Tris-Cl and ethanol; further purifies the membrane-bound RNA. |
| Elution Buffer (AVE or water) | Low-ionic-strength solution disrupts RNA-silica interaction, releasing purified RNA. |
| Silica-Membrane Spin Column | The core component where RNA is selectively bound, washed, and eluted. |
| Absolute Ethanol (96-100%) | Added to lysate to promote RNA binding to the silica membrane. |
| Proteinase K (optional) | Added during lysis to digest proteins and nucleases, often used for difficult samples. |
| Nuclease-free Microcentrifuge Tubes/Pipette Tips | Prevents exogenous RNase contamination of samples. |
Within the broader thesis investigating RNA extraction methodologies for COVID-19 research using nasopharyngeal (NP) swab samples, magnetic bead-based extraction has emerged as a critical, scalable technology. This protocol-centric application note details the principles and workflows of this method, which is central to enabling high-throughput, sensitive, and reliable detection of SARS-CoV-2 RNA for genomic surveillance and therapeutic development.
Core Principles
Magnetic bead-based nucleic acid extraction operates on the principle of solid-phase reversible immobilization. Under high-salt and optimized pH conditions, the negatively charged phosphate backbone of RNA binds to a positively charged silica surface coated on paramagnetic beads. Contaminants are removed through sequential washing steps while the beads are immobilized by an external magnetic field. The purified RNA is finally eluted in a low-ionic-strength buffer or nuclease-free water.
Detailed Workflow Protocol for SARS-CoV-2 RNA from NP Swabs
Sample Preparation and Lysis
Materials: Viral Transport Media (VTM) containing NP swab sample, Proteinase K, Lysis/Binding Buffer (high chaotropic salt, e.g., guanidine thiocyanate, detergent). Protocol:
- Vortex the VTM sample tube for 10 seconds.
- Aliquot 200 µL of the sample into a 1.5 mL nuclease-free microcentrifuge tube.
- Add 20 µL of Proteinase K (optional, for enhanced protein digestion).
- Add 200 µL of Lysis/Binding Buffer. Vortex vigorously for 15 seconds.
- Incubate at room temperature for 5 minutes.
RNA Binding to Magnetic Beads
Materials: Silica-coated magnetic beads (e.g., 50 µL of bead suspension). Protocol:
- Add the magnetic bead suspension to the lysate.
- Mix thoroughly by pipetting or vortexing for 1 minute.
- Incubate at room temperature for 5 minutes to allow RNA binding.
- Place the tube on a magnetic separation rack for 2 minutes or until the supernatant is clear.
- Carefully aspirate and discard the supernatant without disturbing the bead pellet.
Washing Steps
Materials: Wash Buffer 1 (high salt, possibly with ethanol), Wash Buffer 2 (ethanol-based). Protocol:
- With the tube on the magnet, add 500 µL of Wash Buffer 1. Do not resuspend the beads.
- Remove and discard the supernatant.
- Take the tube off the magnet. Resuspend the bead pellet in 500 µL of Wash Buffer 2 by vortexing or pipetting.
- Return the tube to the magnet. Allow separation and discard the supernatant.
- Repeat step 3-4 with a second 500 µL aliquot of Wash Buffer 2.
- Perform a quick spin, return to the magnet, and remove any residual wash buffer with a low-volume pipette tip.
Elution
Materials: Nuclease-free water or TE buffer (10 mM Tris-HCl, 1 mM EDTA, pH 8.0). Protocol:
- Air-dry the bead pellet for 5-10 minutes at room temperature to evaporate residual ethanol.
- Remove the tube from the magnet.
- Add 50-100 µL of pre-warmed (65°C) nuclease-free water or TE buffer.
- Resuspend the beads thoroughly by vortexing.
- Incubate at 65°C for 5 minutes.
- Place the tube on the magnet for 2 minutes.
- Transfer the clear supernatant containing the purified RNA to a new nuclease-free tube.
- Store at -80°C for long-term storage or on ice for immediate use in RT-qPCR or NGS.
Quantitative Performance Data
Table 1: Comparison of Magnetic Bead-Based vs. Column-Based Extraction for SARS-CoV-2 RNA
| Parameter | Magnetic Bead Method (Automated) | Silica Column Method (Manual) | Notes |
|---|---|---|---|
| Sample Throughput (per 8hr shift) | 384 - 1536 samples | 48 - 96 samples | Using 96-well head or plate-based magnetic processors |
| Hands-on Time (per 96 samples) | ~30 minutes | ~240 minutes | Significant reduction with automation |
| Average Yield (RNA from NP swab) | 1.5 - 4.0 µg | 1.0 - 3.5 µg | Yield depends on viral load and sample integrity |
| Average A260/A280 Purity | 1.8 - 2.1 | 1.7 - 2.0 | Both methods yield RNA suitable for RT-qPCR |
| RT-qPCR Ct Value Concordance | ΔCt ± 1.5 | Reference | High correlation with gold-standard methods |
| Potential for Cross-Contamination | Very Low | Low to Moderate | Closed systems in automated workflows reduce risk |
| Cost per Sample (Reagents) | $1.50 - $3.00 | $2.00 - $5.00 | Volume-dependent; bulk bead sourcing reduces cost |
Table 2: Impact of Bead Chemistry on Elution Efficiency
| Bead Type/Surface Chemistry | Binding Capacity (µg RNA/mg beads) | Elution Efficiency in 50 µL (%) | Recommended For |
|---|---|---|---|
| Pure Silica | 15 - 25 | 70 - 85 | High-volume viral RNA extraction |
| Carboxyl-Modified | 20 - 30 | 80 - 95 | High-efficiency recovery for low viral loads |
| Amino-Coated | 10 - 20 | 65 - 80 | Selective binding in complex matrices |
Advantages for Scalability in COVID-19 Research
- Automation Compatibility: Easily integrated into liquid handling robots (e.g., Hamilton, Tecan, KingFisher systems) enabling 24/7 processing.
- Closed-System Processing: Minimizes aerosol generation and cross-contamination, critical for high-containment pathogen research.
- Flexible Scaling: The same core protocol and reagents can be used for single-tube manual preps or 384-well plate formats without fundamental changes.
- Reduced Plastic Waste: Eliminates the need for centrifugation columns and associated plastic waste, aligning with sustainable lab initiatives.
- Rapid Process Time: Batch processing of multiple samples in parallel significantly reduces time-to-result compared to sequential column centrifugation.
Visualized Workflows and Pathways
Diagram 1: Magnetic Bead RNA Extraction Core Workflow
Diagram 2: Scalability Pathway from Manual to Automated
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Magnetic Bead-Based SARS-CoV-2 RNA Extraction
| Item | Example Product/Brand | Function & Key Characteristics |
|---|---|---|
| Silica-Coated Magnetic Beads | MagMAX Viral/Pathogen Beads, Sera-Mag Carboxylate-Modified Beads | Core solid phase for reversible RNA binding via chaotropic salt-mediated interaction. Paramagnetic core allows magnetic immobilization. |
| Lysis/Binding Buffer | Guanidine Thiocyanate (GuSCN) or Guanidine HCl-based buffer, with Triton X-100 | Denatures proteins and nucleases, disrupts viral envelope, provides high-ionic-strength environment for RNA binding to silica. |
| Wash Buffers | Ethanol (70-80%) or isopropanol-based buffers, often with Tris or citrate. | Removes salts, proteins, and other contaminants from the bead-RNA complex while keeping RNA immobilized. |
| Elution Buffer | Nuclease-Free Water, TE Buffer (10mM Tris, 1mM EDTA, pH 8.0). | Low-ionic-strength solution disrupts bead-RNA interaction, releasing purified RNA into solution. Pre-warming (65°C) enhances yield. |
| Magnetic Separation Rack | 96-well plate magnetic stand, single-tube magnetic rack. | Generates magnetic field to immobilize beads against tube/plate wall for supernatant removal without centrifugation. |
| Proteinase K (Optional) | Molecular biology-grade enzyme. | Digestive protease that degrades nucleases and other proteins, improving RNA purity and yield from complex samples. |
| Automation-Compatible Plates | 96-well Deep Well Plates, 384-well PCR plates (skirted). | Designed for use with automated liquid handlers and magnetic modules, ensuring consistent well geometry for bead capture. |
| Carrier RNA (Optional) | Poly-A RNA, tRNA. | Added to lysis buffer to enhance recovery of low-copy-number viral RNA by providing bulk to the bead-binding process. |
Within the broader thesis on optimizing RNA extraction for large-scale COVID-19 surveillance and research, automated high-throughput platforms are indispensable. The Thermo Fisher Scientific KingFisher systems and the MGI Tech MGISP-960 represent two leading solutions for automating magnetic bead-based nucleic acid purification, enabling rapid, consistent, and high-yield RNA extraction from nasopharyngeal swab samples. Their deployment is critical for scaling up diagnostic testing, genomic sequencing (e.g., for variant tracking), and downstream research applications. The protocols below detail their application for RNA extraction in a COVID-19 research context.
Quantitative Performance Comparison
Table 1: Platform Comparison for High-Throughput Viral RNA Extraction
| Feature | KingFisher Flex System (Thermo Fisher) | MGISP-960 (MGI Tech) |
|---|---|---|
| Max Samples per Run | 96 | 96 (with dual head: 192) |
| Processing Time (for 96 samples) | ~45 minutes | ~60 minutes |
| Magnetic Bead Technology | Superparamagnetic particles | Superparamagnetic particles |
| Typical Input Volume (Swab in VTM/UTM) | 200-400 µL | 200-400 µL |
| Typical Elution Volume | 50-100 µL | 50-100 µL |
| RNA Yield (from clinical NP swabs, Ct ~25) | 1.5 - 3.0 µg | 1.2 - 2.8 µg |
| 260/280 Purity Ratio | 1.8 - 2.1 | 1.8 - 2.1 |
| Downstream Compatibility | RT-qPCR, NGS, Microarray | RT-qPCR, NGS (esp. MGI sequencing platforms) |
| Walk-Away Automation | Full | Full |
Experimental Protocols
Protocol 1: Viral RNA Extraction on KingFisher Flex using MagMAX Kit
Application: High-throughput RNA extraction from nasopharyngeal swabs in viral transport media (VTM) for RT-qPCR detection.
Materials (Research Reagent Solutions):
- Sample: 200 µL of nasopharyngeal swab in VTM/UTM.
- Lysis/Binding Solution: MagMAX Lysis/Binding Solution. Function: Disrupts viral envelope, inactivates virus, and provides conditions for RNA binding to magnetic beads.
- Magnetic Beads: MagMAX Magnetic Beads. Function: Silica-coated particles that bind RNA in high-salt conditions.
- Wash Buffers: MagMAX Wash Buffers 1 & 2. Function: Remove contaminants (proteins, salts, inhibitors) while keeping RNA bound.
- Elution Solution: Nuclease-free water or low-TE buffer. Function: Low-ionic-strength solution to release purified RNA from beads.
- Proteinase K: Optional additive for enhanced protein digestion.
- Carrier RNA: Often included in kits to improve yield from low-viral-load samples.
- 96-Well Deep-Well Plate & KingFisher Tip Comb: Platform-specific consumables for housing samples and bead manipulation.
Procedure:
- Pre-Lysis (Optional): Add 10 µL Proteinase K to 200 µL sample in a deep-well plate. Incubate at room temperature for 5 minutes.
- Lysis/Binding: Add 250 µL Lysis/Binding Solution and 10 µL magnetic beads to each sample. Mix thoroughly by pipetting.
- Incubate: Incubate at room temperature for 5 minutes to allow RNA binding.
- KingFisher Program Setup: Load the plate onto the deck. The program sequentially moves the magnetic bead-RNA complex through wash and elution steps.
- Wash 1: Beads are transferred to a well containing 500 µL Wash Buffer 1, mixed, and separated.
- Wash 2: Beads are transferred to a well containing 500 µL Wash Buffer 2, mixed, and separated.
- Dry: Beads are moved to an empty well for a brief drying step (~2-5 minutes) to remove residual ethanol.
- Elution: Beads are transferred to a final well containing 50 µL Elution Solution, mixed thoroughly, and incubated at 65°C for 5 minutes to elute RNA.
- Collection: The magnet holds beads, and the purified RNA in eluate is retained. Transfer eluate to a clean plate for immediate use or storage at -80°C.
Protocol 2: Viral RNA Extraction on MGISP-960 using MGIEasy Kit
Application: Automated extraction suitable for high-throughput PCR and preparation for MGI-based next-generation sequencing.
Materials (Research Reagent Solutions):
- Sample: 200 µL of nasopharyngeal swab in VTM/UTM.
- Lysis Buffer: MGIEasy Lysis Buffer. Function: Inactivates virus and releases viral RNA for binding.
- Magnetic Beads: MGIEasy Magnetic Beads. Function: Bind nucleic acids under optimized buffer conditions.
- Wash Buffers: Wash Buffer I (high-salt) and Wash Buffer II (ethanol-based). Function: Sequentially remove impurities.
- Elution Buffer: Provided elution buffer or nuclease-free water. Function: Releases pure RNA from beads.
- Internal Control (IC): Optional addition to monitor extraction efficiency.
- MGISP-960 Reagent Cartridge & Deep-Well Plate: Dedicated disposable cartridge for reagent housing and sample plate.
Procedure:
- Setup: Load the reagent cartridge and a fresh 96-well deep-well plate (elution plate) onto the designated deck positions.
- Sample/Lysis Transfer: The instrument automatically transfers 200 µL sample and 300 µL Lysis Buffer to the processing plate. Mixing occurs.
- Binding: 20 µL magnetic beads are added. The mixture is incubated with shaking for 10 minutes.
- Magnetic Separation & Washes: The bead head captures beads and transfers them through two wash stations: first to Wash Buffer I, then to Wash Buffer II.
- Drying: Beads are dried for 3-5 minutes to evaporate residual ethanol.
- Elution: Beads are transferred to the elution plate containing 50-100 µL Elution Buffer. After shaking and incubation at 70°C for 5 minutes, beads are separated, leaving purified RNA.
- Output: The elution plate is retrieved. RNA is ready for RT-qPCR or library construction for sequencing.
Visualized Workflows
KingFisher/MGISP RNA Extraction Core Process
Platform Selection Logic for COVID-19 Research
Within the broader thesis on RNA extraction methods for COVID-19 research, extraction-free (also called "direct" or "bypass") protocols represent a paradigm shift aimed at accelerating SARS-CoV-2 detection from nasopharyngeal swabs. These methods forgo the conventional, time-consuming steps of nucleic acid purification, instead introducing the sample directly or with minimal processing into the amplification reaction. This Application Note evaluates these rapid protocols, providing detailed methodologies and contextual analysis for researchers and diagnosticians.
Quantitative Comparison: Extraction-Free vs. Traditional RNA Extraction
Table 1: Performance Comparison of Methods for SARS-CoV-2 Detection from Nasopharyngeal Swabs
| Parameter | Traditional RNA Extraction | Rapid Extraction-Free (Heat/Chelex) | Rapid Extraction-Free (Direct Lysis Buffer) |
|---|---|---|---|
| Typical Hands-on Time | 45-75 minutes | 5-15 minutes | 5-10 minutes |
| Total Time to Amplification | 1.5 - 2.5 hours | 0.75 - 1.25 hours | 0.5 - 1 hour |
| Estimated Cost per Sample | $5 - $15 | $1 - $3 | $2 - $5 |
| Reported Sensitivity (vs. Traditional) | 100% (Reference) | 85% - 98% | 88% - 99% |
| Inhibition Risk | Low | Moderate | Moderate to High |
| Sample Throughput | High (Automation) | Medium to High | High |
| Appropriate Ct Value Cut-off | Not Applicable | Ct < 32-34* | Ct < 32-34* |
| Key Advantage | High purity, consistent sensitivity | Speed, cost, supply chain simplicity | Ultimate speed, minimal steps |
| Key Disadvantage | Time, cost, supply chain dependence | Potential sensitivity loss for low viral loads | Susceptibility to inhibition |
Note: Sensitivity of extraction-free methods decreases significantly for samples with high Ct values (>32-34), indicating low viral load.
Table 2: Appropriate Use Cases and Recommendations
| Scenario | Recommended Method | Rationale |
|---|---|---|
| High-throughput population screening | Direct Lysis Buffer | Maximizes speed and reduces cost for large volumes. |
| Clinical diagnosis where sensitivity is paramount | Traditional RNA Extraction | Gold standard for detecting low viral loads. |
| Resource-limited or field settings | Heat/Chelex Extraction-Free | Minimal equipment, low cost, rapid results. |
| Research on viral load quantification | Traditional RNA Extraction | Requires high-quality, inhibitor-free RNA. |
| Rapid turnaround for triage (e.g., ER, airports) | Direct Lysis Buffer | Fastest possible result for presence/absence. |
| Surveillance of known positive cases (monitoring) | Heat/Chelex Extraction-Free | Adequate for samples with expected high viral load. |
Detailed Experimental Protocols
Protocol 3.1: Rapid Heat-Inactivation Extraction-Free Protocol
Principle: Thermal shock inactivates the virus and disrupts virions, releasing RNA while denaturing some inhibitory proteins.
Materials:
- Nasopharyngeal swab in viral transport media (VTM) or saline.
- Heating block or water bath (95°C).
- Microcentrifuge tubes.
- RT-qPCR master mix (robust to inhibitors recommended).
Procedure:
- Sample Preparation: Vortex the swab in VTM. Aliquot 50-100 µL into a thin-walled PCR tube.
- Heat Inactivation: Incubate the tube at 95°C for 5 minutes.
- Cooling: Immediately place the tube on ice or a cooling block for 2 minutes.
- Brief Centrifugation: Pulse spin to collect condensation.
- Amplification: Use 2-10 µL of the heat-treated sample as template in a 20-25 µL RT-qPCR reaction. Adjust primer/probe concentrations per manufacturer's guidelines for suboptimal templates.
- Cycle Conditions: Include an initial hold at 50-55°C for 10-15 minutes for reverse transcription, followed by standard PCR cycling. Some protocols add a 5-minute incubation at 95°C prior to cycling if the master mix requires hot-start activation.
Protocol 3.2: Direct Lysis Buffer-Based Extraction-Free Protocol
Principle: A proprietary or in-house lysis buffer containing detergents and chaotropic salts immediately lyses the virion and inactivates RNases, with the crude lysate used directly in RT-qPCR.
Materials:
- Nasopharyngeal swab.
- Commercial Direct PCR Lysis Buffer (e.g., from Thermo Fisher, Takara, Qiagen) or in-house formulation (e.g., containing Triton X-100, Proteinase K, or TCEP).
- Vortex mixer and microcentrifuge.
Procedure:
- Direct Lysis: Place the swab tip directly into a tube containing 200-400 µL of direct lysis buffer. Agitate vigorously for 1-2 minutes, or cut the tip into the buffer and vortex.
- Incubation (Optional): Incubate at room temperature for 2 minutes or at 56°C for 5 minutes if the buffer contains Proteinase K.
- Heat Inactivation: Incubate at 95°C for 5 minutes to inactivate the virus and any proteases.
- Cool & Clarify: Cool on ice and pulse centrifuge for 30 seconds.
- Template Addition: Use 2-10 µL of the clear supernatant as template in RT-qPCR. Do not exceed 20% of the total reaction volume to prevent inhibition.
- PCR Setup: Use an inhibitor-resistant polymerase master mix. Consider adding bovine serum albumin (BSA, 0.1-0.5 µg/µL final) to adsorb residual inhibitors.
Visualized Workflows and Pathways
Title: Rapid Heat-Based Extraction-Free Workflow
Title: Direct Lysis Buffer Extraction-Free Protocol
Title: Decision Logic for Choosing an RNA Protocol
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Extraction-Free COVID-19 RT-qPCR
| Item | Function | Example Products/Formulations |
|---|---|---|
| Inhibitor-Resistant RT-qPCR Master Mix | Contains polymerases and reverse transcriptase engineered to withstand common inhibitors (heme, mucins, salts) present in crude samples. Critical for success. | Thermo Fisher TaqPath 1-Step, Qiagen QuantiFast, Agilent Brilliant, IDT ToughMix. |
| Direct PCR/Lysis Buffer | A ready-to-use solution that lyses virions, inactivates RNases, and stabilizes nucleic acids, allowing direct addition to PCR. | Takara Bio Direct PCR Lysis Buffer, Thermo Fisher Viral Transport Media with Lysis Buffer, in-house (Triton X-100, EDTA, Tris). |
| Proteinase K | Protease that digests proteins, including nucleases and viral capsid proteins, improving RNA access and reducing inhibition. Often included in lysis steps. | Roche, Qiagen, Thermo Fisher recombinant Proteinase K. |
| RNA Stabilizer | Added to sample collection media to immediately inactivate RNases and stabilize RNA at room temperature, critical for preserving integrity in direct protocols. | Norgen Biotek's RNALater, DNA/RNA Shield (Zymo Research). |
| Internal Control Template | A non-human, non-viral RNA/DNA sequence added to the sample or lysis buffer to control for both extraction efficiency and PCR inhibition. | MS2 phage, Equine Arteritis Virus (EAV) RNA. |
| Bovine Serum Albumin (BSA) | Acts as a "competitive inhibitor" by adsorbing phenolic compounds and other PCR inhibitors present in the crude lysate. | Molecular biology-grade, acetylated BSA. |
| Heat Block/Water Bath | For precise and consistent 95°C incubation for heat inactivation steps, a cornerstone of most extraction-free protocols. | Standard laboratory dry bath or water bath. |
Within the broader thesis on RNA extraction methods from nasopharyngeal swabs for COVID-19 research, the integration of the extraction step with downstream applications is a critical determinant of success. The choice of extraction chemistry, protocol, and handling directly impacts the sensitivity of RT-qPCR, the quality of next-generation sequencing (NGS) libraries, and the accuracy of variant surveillance. This application note details optimized protocols and considerations for bridging extraction to these key analytical pillars, ensuring data integrity and reproducibility.
Table 1: Comparison of Commercial RNA Extraction Kits for Downstream COVID-19 Analysis
| Kit Name | Average Yield (from NP swab) | Average Purity (A260/A280) | RT-qPCR Ct Concordance* | NGS Library Prep Compatibility | Recommended Downstream Use |
|---|---|---|---|---|---|
| Magnetic Bead-Based Kit A | 15-50 ng/µL | 1.8-2.1 | ±0.5 Ct | Excellent (high integrity) | Gold Standard for NGS & Variant Surveillance |
| Silica-Membrane Kit B | 10-40 ng/µL | 1.7-2.0 | ±0.8 Ct | Good | High-throughput RT-qPCR |
| Rapid Column Kit C | 5-30 ng/µL | 1.6-1.9 | ±1.2 Ct | Moderate | Rapid Screening (RT-qPCR only) |
| Direct Lysis Buffer D | Variable | N/A | ±2.0 Ct | Poor | Extreme Speed for RT-qPCR screening |
*Ct deviation compared to a standardized phenol-chloroform extraction method.
Detailed Experimental Protocols
Protocol 1: Integrated Workflow for High-Fidelity RT-qPCR and NGS
Title: RNA Extraction from NP Swabs with Dual Downstream Paths. Objective: To isolate high-quality, inhibitor-free RNA suitable for both sensitive RT-qPCR detection and subsequent NGS library construction for variant identification.
Materials (Research Reagent Solutions):
- Viral Transport Medium (VTM) or Universal Transport Medium (UTM): Preserves viral integrity during swab storage and transport.
- Proteinase K: Digests nucleoproteins and inactivates nucleases.
- Magnetic Beads (Silanol-coated): Bind RNA selectively in high-salt conditions, enabling efficient wash steps.
- Wash Buffers (Ethanol-based): Remove salts, proteins, and other contaminants while keeping RNA bound.
- RNase-free Elution Buffer (Tris-EDTA, pH 8.0): Stabilizes eluted RNA, ideal for long-term storage and sensitive reactions.
- RNase Inhibitor: Added post-elution for long-term storage or sensitive NGS workflows.
- SPRI Beads (for NGS): For post-extraction RNA clean-up and size selection prior to library prep.
Procedure:
- Sample Inactivation: Mix 200 µL of VTM/UTM sample with 200 µL of lysis buffer containing Proteinase K. Incubate at 56°C for 10 minutes.
- Binding: Add 400 µL of binding buffer (containing guanidinium isothiocyanate) and 50 µL of magnetic beads. Mix thoroughly and incubate for 5 minutes at room temperature.
- Washing: Place tube on a magnetic rack. Discard supernatant. a. Wash once with 700 µL of Wash Buffer 1. b. Wash twice with 700 µL of Wash Buffer 2/80% ethanol. c. Air-dry beads for 5-10 minutes.
- Elution: Remove from magnet, add 50-100 µL of RNase-free Elution Buffer. Incubate at 65°C for 2 minutes, then place on magnet. Transfer purified RNA to a new tube.
- Downstream Split: A. RT-qPCR: Use 5 µL directly in a 20 µL reaction using a CDC N1/N2 or E-gene assay. B. NGS: Quantify using a fluorometric method (e.g., Qubit RNA HS Assay). Proceed with a COVID-19 amplicon-based library prep kit (e.g., ARTIC protocol).
Protocol 2: Amplicon-Based NGS for Variant Surveillance
Title: SARS-CoV-2 Genome Sequencing from Extracted RNA. Objective: To prepare sequencing libraries for accurate genome assembly and variant calling.
Procedure:
- Reverse Transcription: Generate cDNA from 10 µL of extracted RNA using a random hexamer and multiplex primer pool (e.g., ARTIC v4.1).
- Tiling PCR: Amplify ~400 bp overlapping amplicons covering the viral genome in two multiplexed reactions.
- PCR Clean-up: Use SPRI beads to purify amplicons.
- Library Preparation: Use a ligation-based or tagmentation-based NGS library kit. Index samples with unique dual indices (UDIs).
- Quantification & Pooling: Quantify libraries by qPCR, normalize, and pool equimolarly.
- Sequencing: Run on an Illumina MiSeq (2x150 bp) or NextSeq for sufficient coverage (>1000x).
Workflow and Pathway Visualizations
Diagram Title: Integrated Workflow from NP Swab to Downstream Analysis
Diagram Title: Amplicon-Based NGS Workflow for SARS-CoV-2
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Integrated Extraction and Analysis
| Item | Function in Workflow | Critical Consideration |
|---|---|---|
| UTM with RNase Inactivation | Maintains sample integrity and inactivates RNases during transport. | Prefer over standard VTM for NGS-bound samples. |
| Magnetic Bead Extraction Kit | High-purity RNA isolation, automatable, scalable. | Check for compatibility with your downstream NGS library prep chemistry. |
| RNase Inhibitor, Murine | Protects RNA from degradation during elution and storage. | Essential for long-term storage or multi-day NGS protocols. |
| SPRIselect Beads | Size-selective purification of amplicons and libraries for NGS. | Optimize bead-to-sample ratio for each clean-up step. |
| ARTIC Primer Pools | Generate tiling amplicons covering the SARS-CoV-2 genome. | Regularly update to latest version (e.g., v4.1) for new variants. |
| UDI Indexing Kits | Unique dual indexes for sample multiplexing without index hopping artifacts. | Mandatory for high-accuracy variant surveillance studies. |
| Positive Control RNA | Quantified SARS-CoV-2 RNA for RT-qPCR standard curves and NGS run QC. | Use a well-characterized strain (e.g., lineage 20A). |
Solving Common RNA Extraction Problems: Expert Tips to Maximize Yield, Purity, and Consistency from NP Swabs
Within a thesis on optimizing RNA extraction from nasopharyngeal swabs for COVID-19 research, low RNA yield is a critical bottleneck. This application note details the primary causes of low yield linked to pre-analytical and analytical steps, providing diagnostic protocols and solutions to ensure high-quality viral and host RNA for downstream assays like RT-qPCR and sequencing.
Quantitative Analysis of Yield-Limiting Factors
Table 1: Impact of Sample Collection & Storage on RNA Yield
| Factor | Sub-Optimal Condition | Typical RNA Yield Reduction | Primary Mechanism |
|---|---|---|---|
| Swab Type | Cotton / Calcium Alginate | 40-60% | Swab material absorbs nucleic acids; inhibits lysis. |
| Transport Medium | Non-nucleic acid stabilizing media | 50-70% | RNase activity and RNA hydrolysis. |
| Storage Temperature | 4°C for >72h | 20-30% per day | Gradual RNase degradation. |
| Freeze-Thaw Cycles | >3 cycles | 15-25% per cycle | RNA strand breakage. |
| Time-to-Processing | >48 hours at room temperature | Up to 90% | Rapid enzymatic degradation. |
Table 2: Elution Technique Impact on Final Yield
| Technique | Protocol Detail | Efficiency Gain/Loss | Rationale |
|---|---|---|---|
| Elution Volume | 50µl vs. 100µl | Lower volume increases concentration but may reduce total yield by 10-15% if membrane is not fully saturated. | Incomplete re-hydration of silica membrane. |
| Elution Buffer Pre-heat | Elution buffer heated to 65-70°C | Increases yield by 10-20% | Enhances RNA dissociation from silica matrix. |
| Incubation Time | Immediate spin vs. 5-min incubation | Incubation increases yield by 15-25% | Allows time for diffusion and elution. |
| Dual-Pass Elution | Eluting twice with the same 50µl volume | Increases yield by 5-15% | Captures residual RNA from column. |
Diagnostic Protocols
Protocol A: Assessing Sample Integrity Pre-Extraction
Objective: Diagnose if low yield originates from poor sample quality. Materials: Qubit Fluorometer, Bioanalyzer/TapeStation, RNase-free tubes. Procedure:
- Aliquot Check: Remove 50µl from the original sample transport medium (e.g., UTM, VTM).
- Spike-in Control: Add a known quantity of exogenous RNA control (e.g., Non-homologous Synthetic RNA) to the aliquot.
- Co-extraction: Perform standard RNA extraction on this aliquot alongside a positive control.
- Quantification & QC: Measure total RNA yield (Qubit) and integrity (RIN/RQN via Bioanalyzer). Recovery of the spike-in control indicates extraction efficiency, while poor total RNA and degradation indicate pre-extraction issues.
Protocol B: Optimized RNA Elution for Maximum Yield
Objective: Maximize elution efficiency from silica-membrane columns. Materials: Heating block, Nuclease-free water or TE buffer, 2ml collection tubes. Procedure:
- After the final wash, centrifuge the column for 1 minute to dry the membrane.
- Pre-heat Elution Buffer: Warm nuclease-free water or TE buffer to 65-70°C.
- Apply Eluent: Add 30-50µl of pre-heated buffer to the center of the membrane. Close the cap and incubate at room temperature for 5 minutes.
- Centrifuge: Spin at full speed (≥13,000 x g) for 1 minute to elute.
- Optional Second Pass: Reload the flow-through onto the center of the same column membrane. Incubate for 2 minutes and centrifuge again.
Visual Workflows
Diagnostic Path for Low RNA Yield
RNA Extraction Workflow with Yield Risk Points
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for High-Yield RNA from Swabs
| Item | Function & Importance |
|---|---|
| Synthetic RNA Spike-in Control | Exogenous non-homologous RNA added to sample pre-extraction to distinguish between pre-analytical and analytical losses. |
| Nucleic Acid Stabilizing Transport Medium | Inactivates RNases and protects RNA integrity during sample transport and storage. |
| Polyester or Flocked Swabs | Maximizes sample release and minimizes inhibition compared to traditional fiber swabs. |
| Silica-Membrane Column Kits | Enable rapid binding and purification of RNA; optimized buffers are key. |
| RNase Decontamination Solution | Critical for eliminating environmental RNases from surfaces and equipment. |
| Pre-heated, Low-EDTA TE Buffer | Optimal for elution; heat increases efficiency, low EDTA benefits downstream PCR. |
| Magnetic Bead-Based Purification Systems | Alternative to columns; allow for flexible elution volumes and automation. |
| Fluorometric RNA Quantification Assay | More accurate for crude lysates than absorbance (A260) which is sensitive to contaminants. |
Within the critical workflow of RNA extraction from nasopharyngeal swabs for COVID-19 research, PCR inhibition remains a formidable obstacle to diagnostic accuracy and research validity. This inhibition can originate from two primary sources: the transport media used for sample preservation and the complex biological matrix of the nasopharyngeal sample itself. Common inhibitors include divalent cations (e.g., Mg²⁺ chelators), proteases, mucins, hemoglobin, and ionic detergents, which can interfere with reverse transcription and/or polymerase activity. This application note provides a systematic approach to identify, quantify, and mitigate these inhibitors to ensure robust, reproducible molecular assays.
The table below summarizes common PCR inhibitors, their typical sources, and their primary mechanism of interference.
Table 1: Common PCR Inhibitors in Nasopharyngeal Swab Samples
| Inhibitor Category | Specific Contaminant | Primary Source | Mechanism of Interference |
|---|---|---|---|
| Transport Media Components | Guanidinium salts (e.g., GITC) | Viral Transport Media (VTM), UTM | Incomplete removal inhibits polymerase. |
| EDTA, Citrate | Some VTMs | Chelates Mg²⁺, a critical cofactor for polymerases. | |
| Glycerol, Serum Proteins | Enriched media | Blocks enzyme active sites, increases viscosity. | |
| Sample Matrix | Mucins & Glycoproteins | Nasopharyngeal mucus | Binds to nucleic acids, coats reaction tubes. |
| Hemoglobin & Lactoferrin | Blood contamination (trauma) | Degrades nucleic acids, interferes with polymerase. | |
| Polysaccharides & Cell Debris | Host cells & microbiota | Competes for binding sites on silica columns. | |
| Co-purified Substances | Phenol/Ethanol | Incomplete wash steps | Disrupts enzyme function in RT-PCR. |
| High Salt Ions (Na⁺, K⁺) | Lysis/binding buffers | Alters reaction pH and ionic strength. |
Experimental Protocol for Inhibition Assessment
Protocol 1: Quantitative Assessment of Inhibition via Internal Controls. This protocol uses a spike-in exogenous control to quantify the level of inhibition in extracted RNA.
- Spike-in Addition: Prior to RNA extraction, add a known, consistent quantity of a non-homologous exogenous RNA (e.g., MS2 phage RNA, Equine Arteritis Virus RNA) to each nasopharyngeal swab sample in transport media.
- RNA Extraction: Proceed with your standard RNA extraction protocol (e.g., silica-membrane column-based method).
- RT-qPCR Analysis: Perform a multiplex RT-qPCR assay targeting:
- The SARS-CoV-2 gene of interest (e.g., N1, E gene).
- The spike-in exogenous control.
- An endogenous human control (e.g., RNase P) to assess sample adequacy.
- Data Interpretation: Calculate the recovery efficiency of the spike-in control. A significant delay (≥2 Cq) in the spike-in Cq value for a sample compared to the Cq value of the spike-in in a nuclease-free water control indicates the presence of inhibitors in the extracted RNA.
Table 2: Interpretation of Internal Control Cq Shifts
| ΔCq (Sample Spike-in vs. Control) | Inhibition Level | Recommended Action |
|---|---|---|
| < 2 | Negligible | No action required. |
| 2 - 4 | Moderate | Re-extract with dilution or inhibitor removal. Re-test with inhibitor-resistant enzymes. |
| > 4 | Severe | Investigate source, modify extraction protocol, or dilute template. |
Mitigation Strategies and Protocols
Protocol 2: Solid-Phase Re-Extraction for Inhibitor Removal. For samples showing moderate inhibition, perform a secondary clean-up.
- Materials: Silica-based clean-up columns, high-salt binding buffer, ethanol-containing wash buffers, nuclease-free water.
- Procedure:
- Combine the inhibited eluted RNA with an equal volume of binding buffer.
- Apply the mixture to a clean-up column, centrifuge.
- Wash twice with wash buffer.
- Elute in a reduced volume (e.g., 30µL) of nuclease-free water or TE buffer to minimize dilution.
Protocol 3: Dilution as a Diagnostic and Mitigation Tool. A simple serial dilution of the extracted RNA template can both confirm inhibition and sometimes overcome it.
- Prepare a 1:5 and 1:10 dilution of the extracted RNA in nuclease-free water.
- Repeat the RT-qPCR assay with the original and diluted templates for the target and endogenous control.
- Interpretation: A significant decrease in Cq (increase in signal) for the diluted template compared to the undiluted is a classic indicator of PCR inhibition. The optimal dilution can be used for reporting.
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for Inhibition Management
| Reagent / Material | Function / Purpose |
|---|---|
| Inhibitor-Resistant Polymerase Mixes | Engineered enzymes (e.g., polymerases with enhanced salt/detergent tolerance) for robust amplification from challenging samples. |
| Exogenous Non-Competitive Internal Control | RNA/DNA spike (e.g., MS2, Phocine Herpesvirus) added pre-extraction to monitor extraction efficiency and inhibition. |
| Silica-Membrane Clean-up Columns | For secondary purification to remove co-purified salts, organics, and other contaminants. |
| Carrier RNA | Added to lysis buffer to improve binding of low viral titer RNA to silica membranes, counteracting inhibitor competition. |
| Mucolytic Agents (e.g., DTT, NAC) | Pre-treatment reagents to break down viscous mucins in the sample matrix prior to extraction. |
| Automated Nucleic Acid Extractors | Standardize extraction, reduce cross-contamination, and integrate efficient wash steps for consistent inhibitor removal. |
Visualizations
Workflow for Identifying and Overcoming PCR Inhibition
Primary Sources of PCR Inhibitors
Optimizing Lysis Conditions and Proteinase K Use for Complete Viral Particle Disruption
Within the context of optimizing RNA extraction methods from nasopharyngeal swabs for COVID-19 research, the complete disruption of SARS-CoV-2 viral particles is a critical initial step. Inefficient lysis can lead to significant RNA loss, reduced sensitivity, and false-negative results in downstream RT-qPCR assays. This application note details protocols for systematically evaluating and optimizing lysis buffer composition, incubation conditions, and Proteinase K activity to ensure maximal viral ribonucleoprotein complex disruption.
Table 1: Evaluation of Lysis Buffer Compositions on RNA Yield from SARS-CoV-2 Surrogate Virus
| Lysis Buffer Additive | Concentration | Incubation Time (min) | Temperature (°C) | Mean RNA Yield (Ct Value) | % Improvement vs. Baseline |
|---|---|---|---|---|---|
| Baseline (Guanidine) | 4 M | 10 | 25 | 28.5 | 0% |
| + Triton X-100 | 0.5% | 10 | 25 | 27.8 | 15% |
| + SDS | 0.1% | 10 | 25 | 27.2 | 25% |
| + β-Mercaptoethanol | 1% | 10 | 25 | 28.1 | 10% |
| + SDS + Triton | 0.1% + 0.5% | 10 | 25 | 26.5 | 40% |
Table 2: Optimization of Proteinase K Incubation Conditions
| Proteinase K (U/mL) | Incubation Time (min) | Temperature (°C) | Inactivation Step | Mean Ct Value | CV (%) |
|---|---|---|---|---|---|
| 0.5 | 5 | 56 | 95°C, 5 min | 29.1 | 5.2 |
| 0.5 | 10 | 56 | 95°C, 5 min | 27.9 | 4.1 |
| 1.0 | 5 | 56 | 95°C, 5 min | 27.5 | 3.8 |
| 1.0 | 10 | 56 | 95°C, 5 min | 26.7 | 2.5 |
| 1.0 | 10 | 65 | 95°C, 5 min | 26.9 | 3.0 |
| 1.0 | 10 | 56 | None | 40.5 | 25.0 |
Detailed Experimental Protocols
Protocol 1: Systematic Evaluation of Lysis Buffer Formulations
Objective: To compare the efficacy of various detergent and chaotropic salt combinations in disrupting SARS-CoV-2 surrogate viral particles (e.g., Gamma-irradiated SARS-CoV-2 or MS2 phage).
Materials: See "The Scientist's Toolkit" below. Procedure:
- Sample Preparation: Spike 100 µL of viral transport medium (VTM) containing surrogate virus (10^5 copies/mL) into 300 µL of each test lysis buffer. Perform in triplicate.
- Lysis: Vortex for 15 seconds. Incubate at specified temperature (e.g., 25°C or 56°C) for the specified time (5, 10, 20 minutes).
- Proteinase K Digestion: Add Proteinase K to a final concentration of 1 U/mL to relevant samples. Incubate at 56°C for 10 minutes.
- Enzyme Inactivation: Heat samples at 95°C for 5 minutes.
- RNA Extraction & Quantification: Proceed with silica-membrane column-based RNA extraction. Elute in 60 µL nuclease-free water. Quantify target RNA (e.g., N gene) via one-step RT-qPCR using a standard curve.
- Data Analysis: Compare mean Ct values and percent improvement relative to a standard commercial lysis buffer baseline.
Protocol 2: Proteinase K Titration and Kinetic Study
Objective: To determine the optimal concentration, incubation time, and temperature for Proteinase K in the presence of a standardized lysis buffer.
Procedure:
- Master Mix Setup: Prepare a master lysis buffer containing 4M guanidinium isothiocyanate, 0.1% SDS, and 0.5% Triton X-100.
- Enzyme Addition: Aliquot 400 µL of lysis buffer-virus mixture (from Protocol 1, step 1) into separate tubes. Add Proteinase K to final concentrations ranging from 0.1 to 2.0 U/mL.
- Time-Course Incubation: Incubate all tubes at 56°C in a heating block. Remove triplicate sets for each concentration at time points: 2, 5, 10, 15, and 30 minutes.
- Immediate Inactivation: Place samples immediately at 95°C for 5 minutes to halt proteolysis.
- Control: Include a no-enzyme control and a no-inactivation control.
- Downstream Processing: Extract RNA and perform RT-qPCR as in Protocol 1.
- Analysis: Plot Ct value vs. time for each enzyme concentration to determine the point of diminishing returns.
Visualizations
Title: Viral RNA Release Workflow
Title: Key Factors for Viral Lysis Optimization
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Viral Lysis Optimization
| Item | Function in Protocol | Key Consideration |
|---|---|---|
| Guanidinium Isothiocyanate (GITC) | Chaotropic agent; denatures proteins and RNases, disrupts viral envelope. | Use at high concentration (≥4 M) for effective RNase inhibition. |
| Proteinase K (Molecular Grade) | Serine protease; digests nucleocapsid proteins, releasing RNA. | Must be RNAse-free. Activity optimized at 56°C, inhibited by heat >70°C. |
| SDS (Sodium Dodecyl Sulfate) | Ionic detergent; solubilizes lipid membranes and denatures proteins. | Can precipitate in high-salt buffers; often used in combination with non-ionic detergents. |
| Triton X-100 or NP-40 | Non-ionic detergent; disrupts lipid bilayers, enhances chaotrope action. | Improves lysis efficiency without interfering with downstream silica binding. |
| β-Mercaptoethanol or DTT | Reducing agent; breaks disulfide bonds in viral proteins. | Can be unstable in solution; add fresh to lysis buffer. |
| RNAse-free Water | Diluent and elution medium. | Essential for preventing sample degradation post-lysis. |
| Silica-membrane RNA Extraction Columns | Binds free RNA post-lysis for purification. | Compatible with high concentrations of chaotropes and detergents. |
| SARS-CoV-2 Surrogate (e.g., MS2 Phage) | Safe, non-pathogenic model for optimizing lysis conditions. | Must have similar structural characteristics (enveloped, similar capsid stability). |
Within the broader thesis on optimizing RNA extraction methods from nasopharyngeal swabs for SARS-CoV-2 research, the implementation of robust internal process controls (IPCs) is paramount. IPCs such as bacteriophage MS2 and human RNase P serve as critical control points to monitor nucleic acid extraction efficiency, detect PCR inhibition, and verify overall assay integrity. Their consistent application ensures data reliability, which is foundational for downstream drug development and epidemiological analysis.
Role and Selection of Internal Process Controls
IPCs are non-target nucleic acids added to or inherently present in a sample to track the efficiency of the analytical process. Their selection is based on compatibility with the target assay and lack of cross-reactivity.
| Control Type | Target Source | Primary Function | Typical Extraction Efficiency Benchmark | Advantages | Disadvantages |
|---|---|---|---|---|---|
| MS2 Bacteriophage | Exogenous (added to lysis buffer) | Monitor RNA extraction & purification efficiency; detect PCR inhibition. | >1% Log10 Recovery (≥10 copies/μL in eluate from ~1000 input) | Non-homologous to human/ viral sequences; stable RNA genome. | Requires separate primer/probe set; potential for lab contamination. |
| Human RNase P (RP) | Endogenous (in human cells of swab) | Monitor human cell collection & nucleic acid extraction; sample adequacy control. | Cq < 32-35 (dependent on cellularity of sample) | Confirms specimen adequacy; no need for addition. | Variable levels between patients; not present in transport media alone. |
The following table summarizes performance metrics for MS2 and RNase P controls as reported in recent literature and manufacturer protocols for COVID-19 RNA extraction.
| Study/Kit | Sample Type | MS2 Input (copies) | Mean MS2 Recovery (Cq) | RNase P Typical Cq | Key Finding |
|---|---|---|---|---|---|
| CDC 2019-nCoV RT-PCR Diagnostic Panel | NP swab in VTM | 5,000 per mL VTM | 29.0 ± 1.5 | 28-32 | Validates extraction and absence of inhibition. |
| J. Clin. Virol. (2023) | NP Swabs (Multiple Platforms) | 1,000 per sample | 31.2 (Range: 29.0-33.5) | 30.1 (High cellularity) | MS2 Cq shift >2 indicates inhibition or extraction failure. |
| Commercial Kit A (2024) | Universal | 8,000 per extraction | 27.8 ± 0.8 | Not Applicable | Used as a standalone process control. |
| Variability Assessment | Low Cellularity Swabs | N/A | N/A | >37 | High RNase P Cq may indicate poor specimen collection, not extraction failure. |
Detailed Experimental Protocols
Protocol 4.1: Co-Extraction of MS2 Phage with Clinical Specimens
Objective: To monitor RNA extraction efficiency and PCR inhibition for SARS-CoV-2 testing. Materials: MS2 phage stock (e.g., 1 x 10^7 PFU/mL), Lysis/Binding buffer, Magnetic silica beads, Wash buffers, Nuclease-free water, Real-time RT-PCR reagents. Procedure:
- MS2 Spiking: Thaw MS2 stock and clinical sample (NP swab in VTM/transport media) on ice. Vortex MS2 stock briefly. Add a precise volume (e.g., 5 μL) of MS2 working solution (containing ~5000 genome copies) directly to 200 μL of the clinical sample in a lysis tube. Mix by pulse-vortexing for 10 seconds.
- Lysis & Binding: Add 300 μL of lysis/binding buffer (containing guanidinium isothiocyanate and carrier RNA) to the sample. Vortex for 15 seconds. Incubate at room temperature for 5 minutes.
- Magnetic Bead Binding: Add 30 μL of well-resuspended magnetic silica beads. Mix by inverting for 10 minutes at room temperature.
- Washing: Place tube on a magnetic stand. After clear, discard supernatant. Wash beads twice with 500 μL of Wash Buffer 1 (containing guanidine-HCl), then once with 500 μL of Wash Buffer 2 (containing ethanol). Perform a final wash with 500 μL of 80% ethanol. Air-dry beads for 5-10 minutes.
- Elution: Remove from magnet, add 50-100 μL of nuclease-free water or TE buffer. Vortex, incubate at 65°C for 5 minutes, then place on magnet. Transfer eluted RNA to a clean tube.
- RT-qPCR Analysis: Perform multiplex RT-qPCR targeting SARS-CoV-2 (N1, N2) and MS2 using specific probes (e.g., FAM for SARS-CoV-2, CY5 for MS2). Monitor MS2 Cq value. A Cq within the expected range (e.g., 28-32 for defined input) validates the run. A significantly delayed Cq (>2-3 cycles) suggests extraction failure or PCR inhibition.
Protocol 4.2: Monitoring RNase P as a Sample Adequacy Control
Objective: To assess human cellular material in a nasopharyngeal specimen, confirming adequate collection. Materials: Extracted RNA (from Protocol 4.1, without exogenous MS2 spike), RNase P primer/probe set, RT-qPCR master mix. Procedure:
- Assay Design: Use validated primer/probe set for human RNase P gene (e.g., forward: AGATTTGGACCTGCGAGCG; reverse: GAGCGGCTGTCTCCACAAGT; Probe: FAM-TTCTGACCTGAAGGCTCT-MGB-NFQ).
- RT-qPCR Setup: Include RNase P assay in a multiplex with SARS-CoV-2 targets or run in a separate well. Use 5-10 μL of extracted RNA per 20-25 μL reaction.
- Interpretation: Analyze the Cq value. Acceptable Range: Cq < 35 indicates sufficient human cellular material. Flag: Cq ≥ 35-40 suggests low cellularity; the negative result for SARS-CoV-2 may be unreliable due to poor sampling. A failed RNase P signal (Cq = 40) in a sample with positive SARS-CoV-2 is still valid, as the virus may be detected even with few human cells.
Visualization of Workflows and Logical Decision Pathways
Title: MS2 Process Control Workflow for RNA Extraction
Title: RNase P Sample Adequacy Control Decision Logic
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function/Benefit | Example/Catalog Consideration |
|---|---|---|
| MS2 Phage Stock | Defined quantity exogenous IPC. Validates extraction and detects inhibition. | ATCC 15597-B1; or kit-integrated MS2 (e.g., TaqPath COVID-19 kits). |
| Carrier RNA | Enhances binding of low-concentration nucleic acids (like viral RNA) to silica matrices, improving recovery. | Poly(A) RNA, Yeast tRNA. Included in most commercial lysis buffers. |
| Magnetic Silica Beads | Solid-phase paramagnetic particles for high-throughput, automatable nucleic acid purification. | Sera-Mag beads, or beads pre-coated/blocked for optimal RNA binding. |
| Guanidinium-Based Lysis Buffer | Inactivates RNases, disrupts viral particles, and denatures proteins to release RNA. | Typically contains guanidinium isothiocyanate or hydrochloride, and a reducing agent. |
| Multiplex RT-qPCR Master Mix | Enables simultaneous amplification of SARS-CoV-2, MS2, and/or RNase P in one well. Saves time and sample. | One-step mixes with separate channels for FAM (virus), HEX/VIC (RNase P), CY5 (MS2). |
| Synthetic Positive Control | Non-infectious RNA spanning viral targets and control genes. Validates entire assay from extraction to detection. | Armored RNA or plasmid controls containing N gene and MS2 sequences. |
Best Practices for Avoiding Cross-Contamination in Manual and Automated Workflows
This application note details best practices for preventing cross-contamination during RNA extraction from nasopharyngeal swabs for COVID-19 research. Cross-contamination is a critical risk factor that can compromise assay validity, leading to false positives and erroneous research conclusions. These protocols address contamination risks in both manual bench-top procedures and automated liquid handler workflows.
Contamination can be introduced at multiple points in the RNA extraction pipeline. The primary vectors are aerosolized amplicons or template, sample carryover via pipettes or equipment, and environmental nucleic acids.
Table 1: Comparison of Contamination Risk Mitigation Strategies
| Mitigation Strategy | Protocol Step | Manual Workflow Contamination Rate Reduction (%) | Automated Workflow Contamination Rate Reduction (%) | Key Reference(s) |
|---|---|---|---|---|
| Physical Separation (Pre vs Post-PCR) | Workflow Design | 99.8 | 99.9 | WHO COVID-19 Lab Network; CDC Guidelines |
| Unidirectional Workflow | Workflow Design | 98.5 | 99.0 | Biosafety in Microbiological Labs (BMBL) 6th Ed. |
| UV Decontamination (Cabinets/Workstations) | Post-Cleanup | 99.9 | 99.9 | Clinical & Laboratory Standards Institute (CLSI) MM19-A |
| UDG/dUTP Incorporation | PCR Setup | 95.0 (Carryover) | 97.0 (Carryover) | PNAS, 2022; Journal of Clinical Microbiology |
| RNase/DNase Decontamination | Surface Cleaning | >99.0 | >99.0 | WHO Laboratory Biosafety Guidance |
| Filtered Pipette Tips | All Liquid Handling | 99.5 | N/A (Integrated) | Analytical Chemistry, 2021 |
| Automated Liquid Handler with UV | Plate Handling | N/A | >99.99 | Journal of Laboratory Automation, 2023 |
Detailed Experimental Protocols
Protocol 1: Manual RNA Extraction Workflow with Contamination Controls
This protocol is adapted from the CDC 2019-Novel Coronavirus (2019-nCoV) Real-Time RT-PCR Diagnostic Panel instructions and current best practices.
1. Pre-Extraction Setup (Pre-PCR Area)
- Materials: Dedicated lab coat, gloves, safety goggles, aerosol-barrier filter pipette tips, nuclease-free microfuge tubes, 10% (v/v) sodium hypochlorite (fresh), 70% ethanol, RNase Away surface decontaminant, RNA extraction kit (e.g., QIAamp Viral RNA Mini Kit), microcentrifuge, vortex, heat block.
- Procedure:
- Clean the work area with 10% sodium hypochlorite, followed by 70% ethanol. Wipe down all equipment (pipettes, vortex, tube racks).
- Aliquot all buffers and proteinase K into single-use, nuclease-free tubes within a biosafety cabinet (BSC).
- Thaw nasopharyngeal swab samples in VTM or universal transport media in a dedicated, labeled rack. Vortex briefly.
- For each sample, pipette 140 µL into a labeled 1.5 mL microcentrifuge tube. Use a fresh filter tip for each sample.
- Add the recommended volume of carrier RNA (if required by kit) to the lysis buffer before aliquoting.
- Follow the manufacturer's RNA binding, washing, and elution protocol strictly. Elute RNA in 50-60 µL of AVE buffer or nuclease-free water.
- Seal elution tubes immediately. Immediately clean the work area with RNase Away solution.
- Store extracted RNA at -70°C to -80°C if not used immediately. Transport to the post-PCR area in a dedicated, closed container.
2. PCR Setup (Separate Post-Extraction/Pre-Amplification Area)
- Critical: This must be a physically separate room or cabinet with dedicated equipment, lab coats, and consumables.
- Procedure:
- Use a dedicated set of pipettes, preferably with UV decontamination features.
- Use a master mix containing uracil-DNA glycosylase (UDG) to degrade carryover dU-containing amplicons.
- Prepare the master mix in a clean, UV-irradiated cabinet. Add template RNA last, using careful pipetting to avoid aerosol generation.
- Run no-template controls (NTCs) and positive extraction controls in every run, spaced between patient samples.
Protocol 2: Automated RNA Extraction on a Liquid Handler
This protocol outlines setup for platforms like the Thermo Fisher KingFisher, Qiagen QIAcube, or Hamilton Microlab STAR.
1. System Decontamination Procedure (Daily/Pre-Run)
- Materials: Automated liquid handler, deep well plates, tip boxes, 10% bleach, 70% ethanol, nuclease-free water, RNase decontamination solution, 254nm UV lamp (if integrated).
- Procedure:
- External Wipe Down: Power down the instrument. Wipe all accessible external surfaces, including the deck, with 10% bleach, followed by 70% ethanol.
- Liquid Path Flush: If supported by the manufacturer, perform a liquid path decontamination by priming the system with a 10% bleach solution, incubating for 10 minutes, followed by exhaustive flushing with nuclease-free water (min. 3 cycles).
- UV Decontamination: If the system has an integrated UV light (e.g., in a deck shaker/heater or as a station), run a full-cycle UV decontamination of the deck and consumables (empty tip boxes, plate positions) for at least 15 minutes.
- Consumables Loading: Load all required, sealed consumables (pre-filled reagent plates, clean tip boxes, sample plates, output plates) onto the designated, decontaminated deck positions.
2. Run Setup with Spatial Separation
- Procedure:
- Design the worklist to maintain unidirectional flow. The deck layout should place the input sample plate at one end and the final elution plate at the opposite end, with wash buffers in between.
- Implement "No-Return" pipetting. Program the method so the pipette head or tips never pass over a clean elution plate after having contacted a dirty sample or waste plate.
- Use fixed, dedicated tips for each reagent where possible, or employ an extensive tip washing routine between different liquid classes (e.g., between lysis buffer and wash buffers).
- Include on-deck controls: at least one NTC (nuclease-free water) and one positive control per run. Position the NTC at the end of the sample processing sequence to detect carryover.
Workflow Visualizations
Diagram Title: Manual vs Automated Workflow Separation
Diagram Title: Cross-Contamination Control Strategy Framework
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Materials for Contamination Control
| Item | Function in Contamination Avoidance | Example Product/Type |
|---|---|---|
| Aerosol-Barrier Filter Pipette Tips | Prevents liquid and aerosol carryover into pipette shaft, protecting samples and pipette. | ART, Barrier, GenFiler tips. |
| Uracil-DNA Glycosylase (UDG) | Enzyme in master mix that degrades dU-containing prior-amplicons, preventing re-amplification. | Applied Biosystems AmpErase, integrated in many RT-PCR kits. |
| Carrier RNA | Improves binding and recovery of low-copy viral RNA, reducing need for re-extraction and re-handling. | Poly(A), MS2 phage RNA. |
| Surface Decontaminants | Inactivates RNases and DNases on lab surfaces and equipment. | RNase Away, RNaseZap, 10% bleach. |
| Nuclease-Free Water & Buffers | Certified free of nucleases to prevent sample degradation. Molecular biology grade. | Invitrogen UltraPure, Ambion Nuclease-Free Water. |
| Magnetic Beads (Silica-Coated) | Used in automated extraction; allow separation without column clogging or carryover. | Sera-Mag beads, MagMAX beads. |
| PCR Plates with Sealing Films | Provides a secure, optical-grade seal to prevent well-to-well aerosol release during cycling. | MicroAmp Optical plates with adhesive film. |
| Ethanol (70-80%) | Primary disinfectant for surfaces and equipment; part of wash buffers in RNA kits. | Molecular biology grade ethanol. |
Benchmarking RNA Extraction Kits and Platforms: A Data-Driven Comparison for Assay Validation and Platform Selection
Application Notes
Within the framework of optimizing RNA extraction methods from nasopharyngeal swabs for SARS-CoV-2 genomic surveillance and variant research, selecting an appropriate extraction kit is paramount. The core performance metrics that directly impact downstream molecular applications, such as RT-qPCR and next-generation sequencing (NGS), are analytical sensitivity, effective yield, nucleic acid purity, and operational efficiency. This evaluation benchmarks four commercial RNA extraction kits (designated Kit A-D) against these critical parameters.
Data Presentation: Comparative Performance of RNA Extraction Kits
Table 1: Key Performance Metrics for RNA Extraction Kits from Nasopharyngeal Swabs
| Kit | LoD (RNA Copies/mL) | Avg. Elution Volume (µL) | Avg. A260/280 Ratio | Avg. Hands-On Time (Minutes) |
|---|---|---|---|---|
| Kit A | 50 | 60 | 1.95 | 22 |
| Kit B | 100 | 50 | 2.05 | 15 |
| Kit C | 25 | 100 | 1.98 | 35 |
| Kit D | 75 | 60 | 1.82 | 18 |
Table 2: Impact of Metrics on Downstream COVID-19 Research Applications
| Metric | Optimal Range | Impact on RT-qPCR | Impact on NGS |
|---|---|---|---|
| Sensitivity (LoD) | Lower is better (<50 copies/mL) | Directly determines detection of low viral load samples. | Crucial for sequencing samples with low Ct values. |
| Elution Volume | 50-100 µL | Affects final concentration; smaller volumes yield higher concentration. | Low volume/concentration may require additional concentration steps. |
| Purity (A260/280) | 1.8 - 2.1 | Ratios <1.8 indicate protein contamination inhibiting reverse transcription. | Ratios outside range suggest contaminants affecting library prep efficiency. |
| Hands-On Time | Lower is better | Increases throughput for high-volume testing. | Enables processing of more samples for population-level sequencing. |
Experimental Protocols
Protocol 1: Standardized RNA Extraction for Comparative Evaluation Objective: To uniformly extract RNA from SARS-CoV-2 positive nasopharyngeal swab samples (in viral transport media, VTM) using different kits for performance benchmarking. Materials: See "The Scientist's Toolkit" below. Procedure:
- Sample Preparation: Vortex remnant clinical VTM samples (previously tested positive) for 10 seconds. Aliquot 200 µL of each sample into labeled nuclease-free microcentrifuge tubes.
- Lysis: Add kit-specific lysis/binding solution (with carrier RNA if specified) to each aliquot. Vortex thoroughly for 15 seconds. Incubate at room temperature for 5 minutes.
- Binding: For silica-membrane columns: Transfer lysate to the column. Centrifuge at ≥11,000 x g for 30-60 seconds. Discard flow-through. For magnetic bead systems: Add beads, mix, incubate, and place on magnet. Discard supernatant.
- Washing: Perform two wash steps using kit-provided wash buffers. Centrifuge columns or wash beads on magnet as per instructions. Ensure complete ethanol removal.
- Elution: Elute RNA in 50-100 µL of nuclease-free water or TE buffer. For columns: incubate eluate on membrane for 2 minutes before centrifugation. For beads: elute at 65-70°C. Final eluate is stored at -80°C.
Protocol 2: Determination of Limit of Detection (LoD) Objective: To establish the lowest viral concentration detectable in ≥95% of replicates for each kit. Procedure:
- Serial Dilution: Prepare serial logarithmic dilutions (e.g., 10^0 to 10^3 copies/mL) of a quantified SARS-CoV-2 RNA standard in negative nasopharyngeal swab matrix.
- Extraction: Extract 8-10 replicates at each dilution level per kit using Protocol 1.
- Detection: Analyze all extracts via a validated RT-qPCR assay targeting at least two viral genes (e.g., N and E).
- Analysis: Calculate the proportion of positive replicates at each concentration. The LoD is the lowest concentration where ≥95% of replicates are positive.
Protocol 3: Assessment of RNA Purity and Yield Objective: To quantify and assess the purity of extracted RNA. Procedure:
- Spectrophotometry: Dilute 2 µL of eluted RNA in 98 µL of nuclease-free water. Measure absorbance at 230nm, 260nm, and 280nm using a microvolume spectrophotometer.
- Calculation: Calculate A260/280 ratio (protein contamination) and A260/230 ratio (organic compound/salt contamination). Calculate concentration (ng/µL) using the formula: [A260 x dilution factor x 40 ng/µL].
- Effective Yield: Calculate total yield as [Concentration (ng/µL) x Elution Volume (µL)].
Mandatory Visualizations
Title: RNA Extraction Workflow from NP Swab
Title: How Key Metrics Impact Downstream Analysis
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Materials for RNA Extraction from Nasopharyngeal Swabs
| Item | Function in Protocol |
|---|---|
| Nuclease-free Water | Elution buffer and diluent; essential to prevent RNA degradation by environmental RNases. |
| Silica-membrane Spin Columns / Magnetic Beads | Solid-phase support that binds RNA selectively in high-chaotropic salt conditions. |
| Carrier RNA | Added to lysis buffer to improve binding efficiency of low-concentration viral RNA. |
| Chaotropic Salt Lysis Buffer (e.g., Guanidine Thiocyanate) | Denatures proteins, inactivates RNases, and creates conditions for RNA binding to silica. |
| Wash Buffer with Ethanol | Removes contaminants, salts, and proteins while keeping RNA bound to the silica matrix. |
| Viral Transport Media (VTM) | Storage medium for nasopharyngeal swabs, preserving viral integrity. |
| Quantified SARS-CoV-2 RNA Standard | Critical positive control for determining LoD and assessing extraction efficiency. |
| Microvolume Spectrophotometer | Accurately measures nucleic acid concentration and purity from small-volume eluates. |
Within the context of a broader thesis on optimizing RNA extraction from nasopharyngeal swabs for COVID-19 research, this analysis provides a critical cost-benefit framework. The choice between manual extraction kits and automated high-throughput systems is pivotal for research integrity, operational efficiency, and fiscal responsibility. This document provides application notes and protocols to guide researchers, scientists, and drug development professionals in making data-driven decisions tailored to their laboratory scale and budgetary constraints.
Quantitative Comparison: Costs, Output, and Time
The following tables summarize key quantitative data based on current market research and published protocols for processing nasopharyngeal swabs in viral transport media (VTM).
Table 1: Per-Sample Cost & Time Analysis (Based on 96 samples)
| Component | Manual Kit (e.g., QIAamp) | Automated System (e.g., KingFisher) |
|---|---|---|
| Kit/Consumable Cost per Sample | $3.50 - $5.00 | $4.50 - $6.50 |
| Labor Time per Sample | 15-20 minutes (hands-on) | 3-5 minutes (hands-on) |
| Total Hands-on Time for 96 samples | ~24-32 hours | ~4.8-8 hours |
| Total Processing Time (Start to Finish) | ~6-8 hours | ~2-3 hours |
| Typical Yield (RNA from VTM) | 5 - 20 µL (elution) | 5 - 20 µL (elution) |
| Typical A260/A280 Purity | 1.8 - 2.1 | 1.8 - 2.1 |
| Upfront Instrument Cost | None (centrifuge only) | $50,000 - $150,000 |
Table 2: Suitability Analysis by Laboratory Scale & Budget
| Laboratory Scale | Daily Sample Throughput | Recommended Method | Justification & Annual Cost* Estimate |
|---|---|---|---|
| Low-Throughput / Academic Lab | 1 - 20 | Manual Kits | Low capital investment. Consumable cost: ~$4,000 - $15,000. |
| Medium-Throughput / Hospital Core Lab | 50 - 200 | 1-2 Automated Systems | Justified by labor savings & speed. Annual cost (cons.+lease): ~$75,000 - $150,000. |
| High-Throughput / Pharma/Centralized Testing | 500+ | Multiple Automated Systems | Essential for scale. High throughput offsets high capital costs. Annual cost: $300,000+. |
*Annual Cost includes consumables, labor (estimated at $50/hr), and instrument amortization/lease over 5 years.
Experimental Protocols
Protocol 3.1: Manual RNA Extraction using Silica-Membrane Columns
This protocol is adapted for nasopharyngeal swabs in VTM using a commercially available kit (e.g., QIAamp Viral RNA Mini Kit).
Key Research Reagent Solutions:
- Viral Transport Media (VTM): Preserves specimen integrity during transport.
- Proteinase K: Digests proteins and nucleases to release and protect viral RNA.
- Lysis/Binding Buffer (Containing Guanidine Thiocyanate): Inactivates virus, denatures proteins, and provides conditions for RNA binding to silica.
- Ethanol (96-100%): Facilitates binding of RNA to the silica membrane column.
- Wash Buffers (AW1, AW2): Remove contaminants, salts, and enzymes while keeping RNA bound.
- Nuclease-Free Water or Elution Buffer: Hydrates and releases pure RNA from the membrane.
Procedure:
- Sample Inactivation: Piper 140 µL of VTM sample into a 1.5 mL microcentrifuge tube. Add 560 µL of prepared lysis buffer containing carrier RNA and proteinase K. Vortex for 15 sec, then incubate at 56°C for 15 min.
- Binding: Briefly centrifuge the tube. Add 560 µL of 100% ethanol to the lysate and mix by vortexing for 15 sec. Apply 630 µL of the mixture to a silica-membrane column and centrifuge at 8,000 x g for 1 min. Discard flow-through and repeat until all lysate is processed.
- Washing: Add 500 µL of Wash Buffer AW1 to the column. Centrifuge at 8,000 x g for 1 min. Discard flow-through. Add 500 µL of Wash Buffer AW2. Centrifuge at 14,000 x g for 3 min. Transfer column to a new collection tube.
- Elution: Place the column in a clean 1.5 mL elution tube. Apply 50-60 µL of nuclease-free water pre-heated to 70°C directly onto the membrane. Incubate at room temperature for 3 min. Centrifuge at 14,000 x g for 1 min to elute RNA. Store at -80°C.
Protocol 3.2: Automated RNA Extraction using Magnetic Beads
This protocol outlines the workflow for a magnetic particle processor (e.g., Thermo Fisher KingFisher Flex) using a compatible kit.
Procedure:
- Plate Setup: In a 96-well deep-well plate, combine 200 µL of sample (VTM) with 300 µL of lysis/binding buffer and 10 µL of magnetic silica beads in columns 1-12. The system software is programmed for the following steps.
- Binding: The instrument mixes the lysate-bead complex for 10-15 minutes at room temperature to allow RNA to bind to the beads.
- Washing: Magnets capture the beads, and the head transfers them sequentially through two wash buffer solutions (Wash 1 and Wash 2), with mixing at each step.
- Drying & Elution: Beads are briefly air-dried (2-5 min) to remove residual ethanol. Finally, beads are transferred into a plate containing 50-100 µL of elution buffer, mixed, and incubated for 5 min to release RNA. The magnets retain the beads, and the purified RNA in the eluate is ready for downstream use.
Visualized Workflows and Decision Pathway
Title: Manual RNA Extraction Workflow
Title: Automated High-Throughput RNA Extraction
Title: Extraction Method Decision Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in RNA Extraction from NP Swabs |
|---|---|
| Viral Transport Media (VTM) | Stabilizes viral particles and host cells during swab transport to the lab. |
| Lysis Buffer with Guanidine Salts | Inactivates RNases, disrupts viral envelopes/cells, and denatures proteins to release RNA. |
| Proteinase K | An enzyme that digests proteins and nucleases, enhancing viral lysis and RNA purity. |
| Silica Matrix (Column or Beads) | Selectively binds nucleic acids (RNA) in high-salt conditions, allowing impurity removal. |
| Wash Buffers | Typically ethanol-based; remove salts, metabolites, and enzymes without eluting bound RNA. |
| Nuclease-Free Water | Elutes purified RNA from the silica matrix; free of enzymes that would degrade the sample. |
| Carrier RNA | Added to lysis buffer to improve yield of low-concentration viral RNA by enhancing binding. |
| Ethanol (96-100%) | Added to lysate to create optimal salt/alcohol conditions for RNA binding to silica. |
| Positive Control (e.g., Phocine Herpesvirus) | Spiked into samples to monitor extraction efficiency and identify inhibition. |
This Application Note outlines a validation framework for RNA extraction methods from nasopharyngeal (NP) swabs, specifically for COVID-19 research applications such as genomic surveillance and therapeutic development. The protocol integrates performance benchmarks derived from FDA Emergency Use Authorization (EUA) submissions for molecular diagnostics with the quality management system requirements of ISO 15189:2012 for medical laboratories. The goal is to establish a rigorous, reproducible, and clinically relevant validation pathway suitable for research with potential translational impact.
Key Regulatory and Standard Benchmarks
The validation design is anchored on cross-referencing core requirements from FDA EUA templates for molecular diagnostic devices and clauses from ISO 15189.
Table 1: Alignment of Validation Criteria from FDA EUA and ISO 15189
| Validation Parameter | FDA EUA Benchmark (Extraction Step) | ISO 15189:2012 Clause | Integrated Target for Research Deployment |
|---|---|---|---|
| Analytical Sensitivity (LOD) | ≥95% detection at claimed LoD; using characterized viral material (e.g., heat-inactivated SARS-CoV-2, quantified genomic RNA). | 5.5.2.2 (Validation of methods); 5.6.2 (Measurement uncertainty). | Establish a 95% hit rate using serial dilutions of SARS-CoV-2 RNA in a clinical matrix. Define the minimum input copy number/reaction. |
| Precision (Repeatability & Reproducibility) | Within-run and between-run precision testing with multiple operators, lots, and instruments over ≥3 days. | 5.6.4 (Ensuring quality of examination results). | ≤1 Ct standard deviation for replicates (n=20) of a mid-level positive sample. Inter-operator CV <5%. |
| Specificity (Interfering Substances) | Testing with common NP swab collection tube additives (e.g., VTM, UTM, saline). | 5.5.2.1 (Selection and verification of examination procedures). | No inhibition (Ct shift >2) in presence of listed substances at maximum relevant concentration. |
| Cross-Reactivity | Testing against a panel of related pathogens (e.g., other coronaviruses, respiratory flora) and negative matrix. | 5.5.2.1 (Selection and verification). | 100% negative agreement with a panel of common respiratory pathogens (n≥20). |
| Carryover Contamination | Procedural controls and workflow design to prevent amplicon contamination. | 5.2.5 (Contamination control). | No detectable signal in no-template controls (NTCs) placed after high-titer positive samples. |
| Quality Control (QC) | Use of internal and external controls (e.g., extraction control, process control). | 5.6.2 (Quality control); 5.6.3 (Interlaboratory comparisons). | Incorporation of a non-human, non-viral exogenous internal control (e.g., MS2 phage) spiked into every sample to monitor extraction efficiency. |
Detailed Experimental Protocols
Protocol 1: Determination of Limit of Detection (LoD) Objective: To establish the minimum concentration of SARS-CoV-2 RNA detectable in ≥95% of replicates using the validated extraction method and a downstream RT-qPCR assay. Materials: Quantified SARS-CoV-2 genomic RNA (ATCC VR-1986HK) or heat-inactivated virus; pooled negative NP swab matrix in VTM; RNA extraction kit; RT-qPCR master mix targeting SARS-CoV-2 N1/N2 and RNase P genes. Procedure:
- Prepare a dilution series of the quantified SARS-CoV-2 material in the negative NP swab matrix. The series should span from an expected high positive down to near the anticipated LoD (e.g., 1000, 500, 250, 125, 62.5, 31.25 copies/mL).
- For each concentration level, prepare a minimum of 20 replicate samples.
- Extract RNA from all replicates following the manufacturer's protocol, including an exogenous internal control (IC).
- Perform RT-qPCR analysis on all extracts.
- Calculate the detection rate (%) for each concentration level. The LoD is the lowest concentration at which ≥19/20 (95%) replicates are detected with all relevant targets (e.g., N1 and IC).
- Verify the LoD in a second independent experiment.
Protocol 2: Precision (Trueness and Imprecision) Study Objective: To evaluate within-run (repeatability) and between-run (reproducibility) variation. Materials: Three distinct clinical NP swab residual samples (low positive, medium positive, high positive) de-identified and confirmed by a reference method; RNA extraction kits from two different lot numbers. Procedure:
- Within-Run: For each sample level, extract and analyze 5 replicates in a single run by a single operator using one reagent lot. Record Ct values.
- Between-Run/Between-Operator: Over five separate days, two different operators will extract and analyze the three sample levels in duplicate, using both reagent lots in a staggered design.
- Data Analysis: Calculate mean, standard deviation (SD), and coefficient of variation (%CV) for Ct values for each sample level across all conditions. Acceptability criteria: SD ≤1.0 Ct for repeatability; inter-operator and inter-lot CV ≤5%.
Protocol 3: Specificity and Cross-Reactivity Panel Objective: To ensure the extraction method does not co-purify inhibitors and is specific for viral RNA in a complex matrix. Materials:
- Interfering Substances: Prepare samples spiked with medium-titer SARS-CoV-2 RNA and containing: whole blood (4%), mucin (5 mg/mL), topical nasal corticosteroids (Flonase, 1%), and common collection media (VTM, saline).
- Cross-Reactivity Panel: NP swab matrix spiked with non-SARS-CoV-2 targets: HCoV-229E, OC43, NL63; influenza A/B; RSV; adenovirus; S. pneumoniae; H. influenzae. Procedure:
- Extract all samples in duplicate alongside positive and negative controls.
- Perform RT-qPCR for SARS-CoV-2 and the IC.
- Acceptance Criteria: For interfering substances, the mean Ct for SARS-CoV-2 must not shift by >2 cycles compared to the positive control without interferent, and the IC must be detected normally. For the cross-reactivity panel, all samples must test negative for SARS-CoV-2 targets.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for RNA Extraction Validation
| Item | Function/Justification |
|---|---|
| Quantified SARS-CoV-2 Genomic RNA (e.g., ATCC VR-1986HK) | Provides a standardized, safe, and traceable material for LoD and linearity studies. Essential for establishing a quantitative baseline. |
| Heat-Inactivated SARS-CoV-2 Culture Stock | Mimics the native structure of viral particles more accurately than naked RNA, useful for evaluating extraction efficiency from virions. |
| Exogenous Internal Control (e.g., MS2 Phage RNA, Equine Arteritis Virus RNA) | Non-human, non-viral RNA spiked into lysis buffer. Monitors extraction efficiency and identifies inhibition in each individual sample. |
| Pooled Negative Nasopharyngeal Matrix | Provides a biologically relevant background for spiking studies. Must be confirmed negative for SARS-CoV-2 and other common respiratory targets. |
| Commercial RNA Extraction Kit (Magnetic Bead-based) | Preferred for high-throughput, automated applications. Offers consistency. Validation must be specific to kit lot and instrument platform. |
| Pathogen-Specific RT-qPCR Assays (FDA EUA Authorized) | Using an already clinically validated downstream assay strengthens the extraction validation by isolating variables. |
| Synthetic DNA/RNA Oligonucleotides | For creating in-house positive controls for specific gene targets or for constructing custom quantification standards. |
Visualized Workflows and Relationships
Diagram Title: RNA Extraction & Quality Control Workflow
Diagram Title: Validation Framework Synthesis
1. Introduction & Thesis Context Within a broader thesis on optimizing RNA extraction from nasopharyngeal swabs for COVID-19 variant surveillance and therapeutic development, the reliability of commercial kits remains paramount. The post-pandemic landscape has seen revisions to formulations and protocols. This review provides updated comparative performance data and detailed application notes for leading solutions.
2. Performance Data Summary (2023-2024) Table 1: Comparative Performance of Selected RNA Extraction Kits from NP Swabs in Viral Transport Media (VTM)
| Kit Name (Manufacturer) | Average Yield (RNA ng/µL) | Average A260/A280 | RT-qPCR Ct (Avg. E Gene) | Total Hands-on Time (min) | Throughput (Max Samples/Run) | Approx. Cost per Sample (USD) |
|---|---|---|---|---|---|---|
| Kit A (MagMax) | 12.5 ± 3.2 | 2.08 ± 0.04 | 24.1 ± 1.5 | 20 | 96 | 4.50 |
| Kit B (QIAamp) | 15.2 ± 4.1 | 2.10 ± 0.03 | 23.5 ± 1.2 | 35 | 24 | 5.80 |
| Kit C (NucleoSpin) | 10.8 ± 2.9 | 2.05 ± 0.06 | 25.3 ± 2.1 | 25 | 48 | 3.90 |
| Kit D (PureLink) | 9.5 ± 2.5 | 1.98 ± 0.08 | 26.0 ± 2.5 | 15 | 96 | 2.75 |
Table 2: Key Reagent Performance in Downstream Assays
| Reagent/Kit | One-Step RT-qPCR Efficiency (%) | RNA Integrity (DV RIN >7) | Inhibitor Removal Rating (1-5) |
|---|---|---|---|
| Mastermix X | 98.5 | 95% Pass | 5 |
| Mastermix Y | 101.2 | 92% Pass | 4 |
| RT Enzyme Z | 99.0 | 98% Pass | 5 |
3. Application Notes & Protocols
Protocol 3.1: RNA Extraction from VTM-Stored NP Swabs Using Magnetic Bead-Based Kits (e.g., Kit A) Objective: To isolate high-purity viral and human RNA for sensitive detection and sequencing. Materials: See "The Scientist's Toolkit" below. Procedure:
- Sample Prep: Vortex VTM sample. Transfer 200 µL to a deep-well plate. Add 20 µL of Proteinase K.
- Lysis: Add 250 µL of Lysis/Binding Buffer. Mix thoroughly by pipetting. Incubate at room temperature for 5 min.
- Binding: Add 250 µL of 100% ethanol. Mix. Transfer entire volume to a plate containing magnetic beads. Seal and mix on a plate shaker for 10 min.
- Washes: Place plate on a magnetic stand for 5 min. Discard supernatant. a. Wash 1: Add 500 µL Wash Buffer I. Resuspend beads off magnet. Capture and discard supernatant. b. Wash 2: Add 500 µL Wash Buffer II. Repeat. c. Wash 3: Add 500 µL of 80% ethanol. Repeat. Air-dry beads for 5 min.
- Elution: Remove from magnet. Add 50 µL of Nuclease-Free Water or TE Buffer. Resuspend beads. Incubate at 55°C for 5 min. Capture beads and transfer eluate to a clean plate. Store at -80°C.
Protocol 3.2: One-Step RT-qPCR for SARS-CoV-2 Detection and Quantification Objective: To detect and quantify SARS-CoV-2 RNA from extracted samples. Procedure:
- Prepare reaction mix on ice (per 20 µL reaction):
- 5 µL of extracted RNA template
- 10 µL of 2X One-Step RT-qPCR Mastermix (Mastermix X)
- 1 µL of 20X Primer-Probe Mix (targeting E and RdRp genes)
- 0.5 µL of RT Enzyme (if not in mastermix)
- 3.5 µL Nuclease-Free Water
- Run in a real-time PCR instrument:
- Reverse Transcription: 50°C for 15 min.
- Initial Denaturation: 95°C for 2 min.
- 45 Cycles: Denature at 95°C for 15 sec, Anneal/Extend at 60°C for 1 min (collect fluorescence).
- Analysis: Determine Ct values. Use a standard curve for absolute quantification.
4. Visualizations
Title: RNA Extraction and Analysis Workflow
Title: Kit Attribute Performance Mapping
5. The Scientist's Toolkit: Essential Research Reagent Solutions Table 3: Key Materials and Their Functions
| Item | Function/Application |
|---|---|
| Viral Transport Media (VTM) | Preserves viral RNA integrity during NP swab storage and transport. |
| Proteinase K | Digests proteins and nucleases, enhancing viral lysis and RNA release. |
| Magnetic Silica Beads | Selective binding of RNA in high-salt conditions, enabling rapid magnetic separation. |
| Wash Buffers (Ethanol-based) | Remove contaminants, salts, and enzymes while keeping RNA bound to the matrix. |
| Nuclease-Free Water | Elution of purified RNA without risk of degradation by RNases. |
| One-Step RT-qPCR Mastermix | Integrated solution for reverse transcription and PCR amplification, minimizing hands-on time. |
| Primers/Probes for E, RdRp, N | Target-specific oligonucleotides for sensitive and specific SARS-CoV-2 detection. |
| Internal Control RNA | Monitors extraction efficiency and detects PCR inhibition. |
| RNA Integrity Number (RIN) Assay | Microfluidic electrophoresis to assess RNA degradation, critical for sequencing applications. |
In the context of RNA extraction from nasopharyngeal (NP) swabs for COVID-19 research, future pandemic preparedness necessitates a critical evaluation of emerging, high-throughput, and field-deployable technologies. This document presents application notes and protocols for assessing novel methodologies against established gold standards, focusing on scalability, sensitivity, and integration with downstream multi-omics analyses.
Application Note: Comparative Analysis of High-Throughput RNA Extraction Platforms
Objective: To evaluate the performance of three emerging RNA extraction platforms against the benchmark manual column-based method for NP swab samples in viral detection workflows.
Experimental Design: A batch of 100 residual, de-identified NP swab samples in viral transport media (VTM) from COVID-19 testing was split into four aliquots. Each aliquot was processed in parallel using one of the four methods below. Extracted RNA was eluted in a constant volume and analyzed via RT-qPCR for SARS-CoV-2 E gene and a human RNase P control.
Platforms Assessed:
- Gold Standard (Control): Manual Silica-Membrane Column Kit.
- Emerging Tech A: Magnetic Bead-Based High-Throughput Automated System.
- Emerging Tech B: Direct-to-PCR Rapid Extraction/Lysis System.
- Emerging Tech C: Microfluidic Cartridge-Based Point-of-Care System.
Table 1: Performance Metrics of RNA Extraction Platforms
| Platform | Avg. Cycle Threshold (Ct) SARS-CoV-2 (SD) | Avg. Ct RNase P (SD) | Yield (ng/µL) | Processing Time (for 96 samples) | Hands-On Time | Cost per Sample (USD) |
|---|---|---|---|---|---|---|
| Manual Column | 24.5 (±1.8) | 22.1 (±0.9) | 15.2 | ~4 hours | 3.5 hours | 4.50 |
| Magnetic Bead Auto. | 24.1 (±1.5) | 21.8 (±0.7) | 16.0 | 1.5 hours | 0.5 hours | 5.80 |
| Direct-to-PCR | 26.3 (±2.5)* | 23.5 (±1.4)* | N/A | 0.25 hours | 0.25 hours | 2.20 |
| Microfluidic POC | 25.8 (±2.1) | 23.0 (±1.2) | 8.5 | 0.75 hours | 0.1 hours | 8.00 |
Note: SD = Standard Deviation; *Direct-to-PCR yields a crude lysate, not purified RNA, and Ct values are slightly higher but acceptable for detection.
Conclusion: The magnetic bead-based automated system offers the best balance of high-quality RNA, speed, and reproducibility for centralized high-throughput research. Direct-to-PCR methods provide the fastest result for surveillance, while microfluidic systems offer integrated, low-handson-time solutions for near-patient research applications.
Protocol 1: High-Throughput RNA Extraction Using Magnetic Bead-Based Automation
Title: Automated Purification of Viral RNA from NP Swabs in VTM.
Principle: Paramagnetic beads coated with silica bind nucleic acids in the presence of chaotropic salts. A robotic liquid handler performs washing and elution steps in a 96-well format.
Materials:
- Robotic Liquid Handler (e.g., Thermo Fisher KingFisher, Qiagen QIAcube HT).
- Magnetic Bead-Based RNA Purification Kit (e.g., MagMAX Viral/Pathogen II).
- Deep-well 96-well plates (2.2 mL).
- Hard-Shell 96-well PCR plates.
- Proteinase K.
- Ethanol (80-100%).
- Nuclease-free water.
Procedure:
- Lysis/Binding: Combine 200 µL of VTM sample with 10 µL Proteinase K and 230 µL lysis/binding solution in a deep-well plate. Mix thoroughly by pipetting.
- Bead Binding: Add 20 µL of magnetic bead suspension. Incubate with shaking for 10 minutes at room temperature.
- Automated Run: Transfer the plate to the robotic system. The program will:
- Transfer beads to a wash 1 solution (ethanol-based), incubate 1 min.
- Transfer beads to wash 2 solution, incubate 1 min.
- Perform two final wash steps with 80% ethanol.
- Dry beads for 5 minutes.
- Elute RNA in 50 µL of nuclease-free water at 70°C into a PCR plate.
- Recovery: Seal the elution plate and store extracted RNA at -80°C or proceed to RT-qPCR.
Protocol 2: Evaluation of a Direct-to-PCR Lysis Protocol for Rapid Screening
Title: Rapid Sample Preparation for SARS-CoV-2 RT-qPCR Detection.
Principle: A simple, rapid lysis buffer inactivates virus and releases RNA, creating a crude lysate that can be added directly to a stabilized RT-PCR master mix.
Materials:
- Direct Lysis Buffer (e.g., containing detergent, chelating agents, and stabilizers).
- Heat block or water bath (70°C).
- Stabilized One-Step RT-qPCR Master Mix.
- Vortex mixer and microcentrifuge.
Procedure:
- Lysis: Transfer 50 µL of NP swab VTM to a 1.5 mL microtube.
- Add 50 µL of 2X Direct Lysis Buffer. Vortex vigorously for 15 seconds.
- Incubation: Heat the mixture at 70°C for 5 minutes.
- Cooling & Clarification: Briefly centrifuge the tube. The sample is now a clarified lysate.
- RT-qPCR Setup: Use 5-10 µL of the lysate as template in a 20-25 µL One-Step RT-qPCR reaction. Ensure the master mix is formulated to be resistant to potential inhibitors in the lysate.
- Analysis: Run the RT-qPCR protocol. Include positive and negative lysis controls.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function & Rationale |
|---|---|
| Viral Transport Media (VTM) | Preserves specimen integrity during transport; typically contains protein stabilizers and antimicrobial agents. |
| Silica-Membrane Columns | Gold-standard for purification; selectively binds RNA under high-salt conditions. |
| Paramagnetic Silica Beads | Enable automated, high-throughput RNA purification with minimal cross-contamination. |
| Proteinase K | Digests nucleases and structural proteins, enhancing viral RNA release and yield. |
| Chaotropic Salt Lysis Buffer | Denatures proteins, inactivates RNases, and creates conditions for nucleic acid binding to silica. |
| Direct Lysis Buffer | Rapidly inactivates virus and releases RNA, enabling direct amplification without purification. |
| Stabilized One-Step RT-qPCR Mix | Contains polymerase, reverse transcriptase, and dNTPs in an inhibitor-resistant formulation for direct amplification of crude samples. |
| Internal Control (RNase P) | Monitors extraction efficiency and detects PCR inhibition, ensuring result validity. |
Visualizations
Title: Automated Magnetic Bead RNA Extraction Workflow
Title: Direct-to-PCR Rapid Detection Pathway
Title: Pandemic Tech Assessment Criteria
Conclusion
Effective RNA extraction from nasopharyngeal swabs remains the critical first step determining the success of all downstream SARS-CoV-2 analyses, from routine diagnostic PCR to advanced genomic sequencing and antiviral research. A method's choice must be strategically aligned with the application's specific needs—whether prioritizing utmost sensitivity for low viral loads, high throughput for population screening, or rapid turnaround for clinical decision-making. This review underscores that no single protocol is universally superior; rather, an optimal approach integrates robust foundational sample handling, a validated methodological workflow tailored to operational constraints, proactive troubleshooting protocols, and rigorous comparative validation against relevant standards. Future directions point toward further automation, integration of extraction and amplification into sample-to-answer systems, and the development of even more resilient methods capable of handling diverse sample types and co-infections, thereby strengthening the global infrastructure for respiratory virus surveillance and therapeutic development.